Abstract
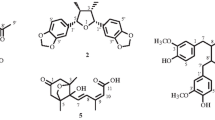
Six novel compounds, 3-nonadec-14′-(Z)-enyl phenol (1a); 4,5-dihydroxy-4,2′-epoxy-5-[16′-Z-18′-E-heneicosenyldiene]-cyclohex-2-enone (2), 2,4,5-trihydroxy-2-[16′-Z-heneicosenyl]-cyclohexanone (3); 4S,6R-dihydroxy-6-[12′-Z-heptadecenyl]-cyclohex-2-enone (4a); 4S,6R-dihydroxy-6-[14′-Z-nonadecenyl]-cyclohex-2-enone (4b); and 1,2,4-trihydroxy-4-[16′-Z-heneicosenyl]-cyclohexane (5) were identified from the roots and stems of Lannea rivae in addition to the known cardanols, 3-heptadec-12′-Z-enyl phenol (1b), 3-pentadec-10′-Z-enyl phenol (1c) and 3-pentadecyl phenol (1d), sitosterol (6), sitosterol glucoside (7), taraxerone (8), taraxerol (9), E-lutein (10), myricetin (11), myricetin-3-O-α-rhamnopyranoside (12), myricetin-3-O-β-galactopyranoside (13) and (-)-epicatechin-3-O-gallate (14). The ketones 4a and 4b were isolated as a mixture and were qualitatively separated and identified by GCMS. Myricetin (11) and epicatechin gallate (14) displayed over 90 % DPPH radical-scavenging activity at 50 μg mL−1, while its glycosides (12 and 13) showed percentages of over 70 % in the same assay. The same compounds 11 and 14 showed antibacterial activity similar to erythromycin and vancomycin against Gram-positive bacteria and were also active against Gram-negative bacteria, but not as much as the cefuroxime, ciprofloxacin and nalidixic acid standards. Compounds 1a–d, 4a–b and 5 were all relatively non-toxic, while 2 (the epoxy cyclohex-2-enone) and 3 (the trihydroxy cyclohexanone) showed more toxicity than the others. These two toxic compounds, 2 and 3 also showed antiplasmodial activity with IC50 values between 0.48 and 2.05 μg mL−1. The mixture of dihydroxy cyclohex-2-enones 4a and 4b, which was far less toxic than 2 and 3, also showed promising antiplasmodial activity and may be a possible lead for further investigation as an antiplasmodial drug.

Similar content being viewed by others
References
Akala HM, Eyase FL, Cheruiyot AC, Omondi AA, Ogutu BR, Waters NC, Johnson JD, Polhemus ME, Schnabel DC, Walsh DS (2011) Antimalarial drug sensitivity profile of western Kenya Plasmodium falciparum field isolates determined by a SYBR Green I in vitro assay and molecular analysis. Am J Trop Med Hyg 85:34–41
Akhtar MN, Lam KW, Abas F, Maulidiani F, Ahmad S, Shah SAA, Choudhary MI, Atta-ur-Rahman TI, Lajis NH (2011) New class of acetylcholinesterase inhibitors from the stem bark of Knema laurina and their structural insights. Bioorg Med Chem Lett 21:4097–4103
Alam B, Hossain S, Habib R, Rea J, Islam A (2012) Antioxidant and analgesic activities of Lannea coromandelica Linn. bark extract. Int J Pharm 8:224–233
Anderson JC, Headley C, Stapleton PD, Taylor PW (2005) Synthesis and antibacterial activity of hydrolytically stable (−)-epicatechin gallate analogues for the modulation of beta-lactam resistance in Staphylococcus aureus. Bioorg Med Chem Lett 15:2633–2635
Anderson JC, McCarthy RA, Paulin S, Taylor PW (2011) Anti-staphylococcal activity and β-lactam resistance attenuating capacity of structural analogues of (−)-epicatechin gallate. Bioorg Med Chem Lett 21:6996–7000
Braca A, Politi M, Sanogo R, Sanou H, Morelli I, Pizza C, de Tommasi N (2003) Chemical composition and antioxidant activity of phenolic compounds from wild and cultivated Sclerocarya birrea (Anacardiaceae) leaves. J Agric Food Chem 51:6689–6695
Caturla N, Vera-Samper E, Villalain J, Mateo CR, Micole V (2003) The relationship between the antioxidant and the antibacterial properties of galloylated catechins and the structure of phospholipid membranes. Free Radical Biol Med 34:648–662
Chaabia M, Beghidja N, Benayache S, Lobstein A (2008) Activity-guided isolation of antioxidant principles from Limoniastrum feei (Girard) Batt. Z Naturforsch C J Biosci 63:801–807
Chaturvedula VSP, Schilling JK, Miller JS, Andriantsiferana R, Rasamison VE, Kingston GI (2004) New cytotoxic terpenoids from the wood of Vepris punctata from the Madagascar rainforest. J Nat Prod 67:895–898
Ciu Y, Oh YJ, Lim J, Youn M, Lee I, Pak HK, Park W, Jo W, Park S (2012) AFM study of differential inhibitory effects of green tea polyphenol (−)-epicatechin-3-gallate (EGGC) against Gram positive and Gram negative bacteria. Food Microbiol 29:80–87
Clarkson C, Maharaj VJ, Crouch NR, Grace WM, Pillay P, Matsabisa MG, Bhagwandin N, Smith PJ, Folb PI (2004) In vitro antiplasmodial activity of medicinal plants native to or naturalised in South Africa. J Ethnopharmacol 92:177–191
Croft KD (1998) The chemistry and biological effects of flavonoids and phenolic acids. Ann N Y Acad Sci 854:435–442
Cuendet M, Hostettmann K, Potterat O, Dyatmiko W (1997) Iridoid glucosides with free radical scavenging properties from Fagraea blumei. Helv Chim Acta 80:1144–1152
David JM, Chávez JP, Chai H-B, Pezzuto JM, Cordell GA (1998) Two new cytotoxic compounds from Tapirira guianensis. J Nat Prod 61:287–289
Davids D, Blouws T, Aboyade O, Gibson D, De Jong JT, Van’t Klooster C, Hughes G (2014) Traditional health practitioners’ perceptions, herbal treatment and management of HIV and related opportunistic infections. J Ethnobiol Ethnomed 10:77
de Jesus Correia S, David JM, David JP, Chai H-B, Pezzuto JM, Cordell GA (2001) Alkyl phenols and derivatives from Tapirira obtusa. Phytochemistry 56:781–784
Deharo E, Bourdy G, Quenevo C, Munoz V, Ruiz G, Sauvain M (2001) A search for bioactive natural compounds in Bolivia through a multidisciplinary approach. Part V. Evaluation of the antimalarial activity of plants used by the Tacana Indians. J Ethnopharmacol 77:91–98
Deji-Agboola AM, Olajubu FA (2010) Antimicrobial activity of Lannea welwitschii stem bark against wound pathogens. Ethiopian Pharm J 28:110–116
Desjardins RE, Canfield CJ, Haynes JD, Chulay JD (1979) Quantitative assessment of antimalarial activity in vitro by a semiautomated microdilution technique. Antimicrob Agents Chemother 16:710–718
D’Souza L, Wahidullah S, Prabha D (2010) Antibacterial phenolics from the mangrove Lumnitzera racemosa. Indian J Mar Sci 39:294–298
Faizi S, Ali M, Saleem R, Irfanullah Bibi S (2001) Complete 1H and 13C NMR assignments of stigma-5-en-3-O-beta-glucoside and its acetyl derivative. Magn Reson Chem 39:399–405
Gachet MS, Kunert O, Kaiser M, Brun R, Zehl M, Keller W, Munoz RA, Bauer R, Schuehly W (2011) Antiparasitic compounds from Cupania cinerea with activities against Plasmodium falciparum and Trypanosoma brucei rhodesiense. J Nat Prod 74:559–566
Gathirwa JW, Rukunga GM, Njagi ENM, Omar SA, Guantai AN, Muthaura CN, Mwitari PG, Kimani CW, Kirira PG, Tolo FM, Ndunda TN, Ndiege IO (2007) In vitro anti-plasmodial and in vivo anti-malarial activity of some plants traditionally used for the treatment of malaria by the Meru community in Kenya. J Nat Med 61:261–268
Gathirwa JW, Rukunga GM, Njagi ENM, Omar SA, Mwitari PG, Guantai AN, Tolo FM, Kimani CW, Muthaura CN, Kirira PG, Ndunda TN, Amalemba G, Mungai GM, Ndiege IO (2008) The in vitro anti-plasmodial and in vivo anti-malarial efficacy of combinations of some medicinal plants used traditionally for treatment of malaria by the Meru community in Kenya. J Ethnopharmacol 115:223–231
Gathirwa JW, Rukunga GM, Mwitari PG, Mwikwabe NM, Kimani CW, Muthaura CN, Kiboi DM, Nyangacha RM, Omar SA (2011) Traditional herbal antimalarial therapy in Kilifi district, Kenya. J Ethnopharmacol 134:434–442
Gibbons S (2005) Plants as a source of bacterial resistance modulators and anti-infective agents. Phytochem Rev 4:63–78
Gibbons S, Moser E, Kaatz GW (2004) Catechin gallates inhibit multidrug resistance (MDR) in Staphylococcus aureus. Planta Med 70:1240–1242
Griep MA, Blood S, Larson MA, Koepsell SA, Hinrichs SH (2007) Myricetin inhibits Escherichia coli DnaB helicase but not primase. Bioorg Med Chem 15:7203–7208
Groweiss A, Cardellina JH, Pannell LK, Uyakul D, Kashman Y, Boyd MR (1997) Novel cytotoxic, alkylated hydroquinones from Lannea welwitschii. J Nat Prod 60:116–121
Guo Q, Zhao B, Shen S, Hou J, Hu J, Xin W (1999) ESR study on the structure–antioxidant activity relationship of tea catechins and their epimers. Biochim Biophys Acta 1427:13–23
Hamilton-Miller JMT, Shah S (2000) Activity of the tea component epicatechin gallate and analogues against methicillin-resistant Staphylococcus aureus. J Antimicrob Chemother 46:847–863
Islam MT, Ito T, Sakasai M, Tahara S (2002) Zoosporicidal activity of polyflavonoid tannin identified in Lannea coromandelica stem bark against phytopathogenic oomycete Aphanomyces cochlioides. J Agric Food Chem 50:6697–6703
Jayaraman P, Sakharkar MK, Lim CS, Tang TH, Sakharkar KR (2010) Activity and interactions of antibiotic and phytochemical combinations against Pseudomonas aeruginosa in vitro. Int J Biol Sci 6:556–568
Johnson JD, Dennull RA, Gerena L, Lopez-Sanchez M, Roncal NE, Waters NC (2007) Assessment and continued validation of the malaria SYBR Green I-based fluorescence assay for use in malaria drug screening. Antimicrob Agents Chemother 51:1926–1933
Kapche GDWF, Laatsch H, Fotso S, Kouam SF, Wafo P, Ngadjui BT, Abegaz BM (2007) Lanneanol: a new cytotoxic dihydroalkylcyclohexenol and phenolic compounds from Lannea nigritana (Sc. Ell.) Keay. Biochem Syst Ecol 35:539–543
Khachik F, Chang A-N (2009) Total synthesis of (3R,3’R,6’R)-Lutein and its stereoisomers. J Org Chem 74:3875–3885
Kokwaro JO (2009) Medicinal plants of east Africa. University of Nairobi Press, Nairobi
Koné WM, Soro D, Dro B, Yao K, Kamanzi K (2011) Chemical composition, antioxidant, antimicrobial and acetylcholinesterase inhibitory properties of Lannea barteri (Anacardiaceae). Aust J Basic Appl Sci 5:1516–1523
Kovganko NV, Kashkan ZN, Borisov EV, Batura EV (1999) C-13 NMR spectra of β-sitosterol derivatives with oxidized rings A and B. Chem Nat Compd 35:646–649
Kumawat RS, Mruthunjaya K, Gupta MK (2012) Preparation, characterization and antioxidant activities of gallic acid-phospholipid complex. Int J Res Pharm Sci 2:138–148
Lee JI, Kong C-S, Jung ME, Hong JW, Lim SY, Seo Y (2011) Antioxidant activity of the Halophyte Limonium tetragonum and its major active components. Biotechnol Bioprocess Eng 16:992–999
Lehane AM, Saliba KJ (2008) Common dietary flavonoids inhibit the growth of the intra erythrocytic malaria parasite. BMC Res Notes 1:26
Liu Y, Abreu PJM (2006) Long chain alkyl and alkenyl phenols from the roots of Ozoroa insignis. J Braz Chem Soc 17:527–532
Liu M, Matsuzaki S (1995) Antibacterial activity of flavonoids against methicillin-resistant Staphylococcus aureus (MRSA). Dokkyo J Med Sci 22:253–261
Liu Z, Jiang W, Deng ZW, Lin WH (2010) Assignment of the absolute stereochemistry of an unusual diterpenoid from the mangrove plant Excoecaria agallocha L. J Chin Pharm Sci 19:387–392
Liu Q, Wang Y-q, Xu P-r, Zhang H-m, Zhao Y-f, Lu Y-n (2011) Study on the antibacterial activity of catechins against the heterogeneous vancomycin resistant Staphylococcus. Zhongguo Kangshengsu Zazhi 36:557-S1
Lomonaco D, Pinheiro Santiago GM, Ferreira YS, Campos Arriaga AM, Mazzetto SE, Mele G, Vasapollo G (2009) Study of technical CNSL and its main components as new green larvicides. Green Chem 11:31–33
Maiga A, Malterud KE, Diallo D, Paulsen BS (2006) Antioxidant and 15-lipoxygenase inhibitory activities of the Malian medicinal plants Diospyros abyssinica (Hiern) F. White (Ebenaceae), Lannea velutina A. Rich (Anacardiaceae) and Crossopteryx febrifuga (Aftel) Benth. (Rubiaceae). J Ethnopharmacol 104:132–137
Maiga A, Malterud KE, Mathisen GH, Paulsen RE, Thomas-Oates J, Bergstrom E, Reubsaet L, Diallo D, Paulsen BS (2007) Cell protective antioxidants from the root bark of Lannea velutina A. Rich., a Malian medicinal plant. J Med Plants Res 1:66–79
Maregesi SM, Pieters L, Ngassapa OD, Apers S, Vingerhoets R, Cos P, van den Berghe DA, Vlietinck AJ (2008) Screening of some Tanzanian medicinal plants from Bunda district for antibacterial, antifungal and antiviral activities. J Ethnopharmacol 119:58–66
Maregesi S, van Miert S, Pannecouque C, Haddad MHF, Hermans N, Wright CW, Vlietinck AJ, Apers S, Pieters L (2010) Screening of Tanzanian Medicinal Plants against Plasmodium falciparum and Human Immunodeficiency Virus. Planta Med 76:195–201
Maundu PM, Ngugi GW, Kabuye CHS (1999) Traditional food plants of Kenya. National Museums of Kenya, Nairobi
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Muregi FW, Chhabra SC, Njagi ENM, Lang’at-Thoruwa CC, Njue WM, Orago ASS, Omar SA, Ndiege IO (2003) In vitro antiplasmodial activity of some plants used in Kisii, Kenya against malaria and their chloroquine potentiation effects. J Ethnopharmacol 84:235–239
Nanjo F, Goto K, Seto R, Suzuki M, Sakai M, Hara Y (1996) Scavenging effects of tea catechins and their derivatives on 1,1-diphenyl-2-picrylhydrazyl radical. Free Radical Biol Med 21:895–902
Okoth DA, Koorbanally NA (2015) Cardanols, long chain cyclohexenones and cyclohexenols from Lannea schimperi (Anacardiaceae). Nat Prod Commun 10(1):103–106
Okoth DA, Chenia HY, Koorbanally NA (2013) Antibacterial and antioxidant activities of flavonoids from Lannea alata (Engl.) Engl. (Anacardiaceae). Phytochem Lett 6:476–481
Ouattara L, Koudou J, Karou DS, Giaco L, Capelli G, Simpore J, Fraziano M, Colizzi V, Traore AS (2011) In vitro anti Mycobacterium tuberculosis H37Rv activity of Lannea acida A. Rich from Burkina Faso. Pak J Biol Sci 14:47–52
Park KD, Park YS, Choo SJ, Sun WS, Kim SH, Jung DH, Kim JH (2004) Antimicrobial activity of 3-O-acyl-(-)-epicatechin and 3-O-acyl-(+)-catechin derivatives. Planta Med 70:272–276
Picerno P, Mencherini T, Della Loggia R, Meloni M, Sanogo R, Aquino RP (2006) An extract of Lannea microcarpa: composition activity and evaluation of cutaneous irritation in cell cultures and reconstituted human epidermis. J Pharm Pharmacol 58:981–988
Qin R, Xiao K, Li B, Jiang W, Peng W, Zheng J, Zhou H (2013) The combination of catechin and epicatechin gallate from Fructus Crataegi potentiates β-lactam antibiotics against methicillin-resistant Staphylococcus aureus (MRSA) in vitro and in vivo. Int J Mol Sci 14:1802–1821
Queiroz EF, Kuhl C, Terreaux C, Mavi S, Hostettmann K (2003) New dihydroalkylhexenones from Lannea edulis. J Nat Prod 66:578–580
Rice-Evans CA, Miller NJ, Paganga G (1996) Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biol Med 20:933–956
Roumy V, Fabre N, Portet B, Bourdy G, Acebey L, Vigor C, Valentin A, Moulis C (2009) Four anti-protozoal and anti-bacterial compounds from Tapirira guianensis. Phytochemistry 70:305–311
Rubinstein LV, Shoemaker RH, Paul KD, Simon RM, Tosini S, Skehan P, Scudiero DA, Monks A, Boyd MR (1990) Comparison of in vitro anticancer-drug-screening data generated with a tetrazolium assay against a diverse panel of human tumor cell lines. J Natl Cancer Inst 82:1113–1118
Sakanaka S, Juneja LR, Taniguchi M (2000) Antimicrobial effects of green tea polyphenols on thermophillic spore-forming bacteria. J Biosci Bioeng 90:81–85
Sakurai N, Yaguchi Y, Inoue T (1987) Constituents of Myrica rubra. 2. Triterpenoids from Myrica rubra. Phytochemistry 26:217–219
Sannella AR, Messori L, Casini A, Vincieri FF, Bilia AR, Majori G, Severini C (2007) Antimalarial properties of green tea. Biochem Biophys Res Commun 353:177–181
Shah S, Stapleton PD, Taylor PW (2008) The polyphenol (−)-epicatechin gallate disrupts the secretion of virulence-related proteins by Staphylococcus aureus. Lett Appl Microbiol 46:181–185
Shiota S, Shimizu M, Mizushima T, Ito H, Hatano T, Yoshida T, Tsuchiya T (1999) Marked reduction in the minimum inhibitory concentration (MIC) of beta-lactams in methicillin-resistant Staphylococcus aureus produced by epicatechin gallate, an ingredient of green tea (Camellia sinensis). Biol Pharm Bull 22:1388–1390
Smilkstein M, Sriwilaijaroen N, Kelly JX, Wilairat P, Riscoe M (2004) Simple and inexpensive fluorescence-based technique for high-throughput antimalarial drug screening. Antimicrob Agents Chemother 48:1803–1806
Stapleton PD, Shah S, Anderson JC, Hara Y, Hamilton-Miller JMT, Taylor PW (2004) Modulation of β-lactam resistance in Staphylococcus aureus by catechins and gallates. Int J Antimicrob Agents 23:462–467
Stapleton PD, Shah S, Hara Y, Taylor PW (2006) Potentiation of catechin gallate mediated sensitization of Staphylococcus aureus to oxacillin by nongalloylated catechins. Antimicrob Agents Chemother 50:752–755
Stapleton PD, Shah S, Ehlert K, Hara Y, Taylor PW (2007) The beta-lactam resistance modifier (−)-epicatechin gallate alters the architecture of the cell wall of Staphylococcus aureus. Microbiol Sgm 153:2093–2103
Sultana S, Ilyas M (1986) A flavanone from Lannea acida. Phytochemistry 25:963–964
Sun Y, Tang C, Wu X, Pan Z, Wang L (2012) Characterization of alkylphenol components in Ginkgo biloba Sarcotesta by thermochemolysis–gas chromatography/mass spectrometry in the presence of trimethylsulfonium hydroxide. Chromatographia 75:387–395
Tasdemir D, Lack G, Brun R, Ruedi P, Scapozza L, Perozzo R (2006) Inhibition of Plasmodium falciparum fatty acid biosynthesis: evaluation of FabG, FabZ, and FabI as drug targets for flavonoids. J Med Chem 49:3345–3353
Thomford NE, Dzobo K, Chopera D, Wonkam A, Skelton M, Blackhurst D, Chirikure S, Dandara C (2015) Pharmacogenomics implications of using herbal medicinal plants on African populations in Health transition. Pharmaceuticals 8:637–663
Trager W, Jensen JB (1976) Human malaria parasites in continuous culture. Science 193:673–675
Vitoria M, Granich R, Gilks CF, Gunneberg C, Hosseini M, Were W, Raviglione M, De Cock KM (2009) The global fight against HIV/AIDS, Tuberculosis, and Malaria: current status and future perspectives. Am J Clin Pathol 131:844–848
World Health Organization (WHO) (2008) Traditional medicine fact sheet number 134. www.who.int/mediacentre/factsheets/fs134/en/. Accessed 7th July 2015
Xiao K-k, Qin R-x, Li B, Li X-l, Peng W, Zhou H (2012) The enhanced anti-MRSA effect by catechins on beta-lactam antibiotics. Zhongguo Kangshengsu Zazhi 37:397-S394
Xu HX, Lee SF (2001) Activity of plant flavonoids against antibiotic-resistant bacteria. Phytother Res 15:39–43
Xu ZJ, Yeung SYV, Chang Q, Huang Y, Chen Z-Y (2004) Comparison of antioxidant activity and bioavailability of tea epicatechins and their epimers. Br J Nutr 91:873–881
Yoda Y, Hu ZQ, Zhao WH, Shimamura T (2004) Different susceptibilities of Staphylococcus and Gram-negative rods to epigallocatechin gallate. J Infect Chemother 10:55–58
Acknowledgments
The authors are grateful to TWOWS for financial assistance. We also thank Mr. Philip Apamo and Mr. Charles Maasai of the Center for Disease Control, Kisian, Kenya, for technical assistance with antibacterial assays. We thank Prof. Peter Smith of Stellenbosch University for conducting cytotoxicity assays. Lastly, we thank Ms. Agnes Cheruiyot, Mr. Charles Okudo and Ms. Redemptah Yeda for Plasmodium falciparum culture and sensitivity testing.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Okoth, D.A., Akala, H.M., Johnson, J.D. et al. Alkyl phenols, alkenyl cyclohexenones and other phytochemical constituents from Lannea rivae (chiov) Sacleux (Anacardiaceae) and their bioactivity. Med Chem Res 25, 690–703 (2016). https://doi.org/10.1007/s00044-016-1521-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1521-2