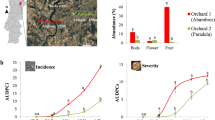
Abstract
The olive tree is one of the most important agricultural plants, affected by several pests and diseases that cause a severe decline in health status leading to crop losses. Olive leaf spot disease caused by the fungus Venturia oleaginea can result in complete tree defoliation and consequently lower yield. The aim of the study was to obtain new knowledge related to plant–pathogen interaction, reveal mechanisms of plant defense against the pathogen, and characterize fungal phyllosphere communities on infected and symptomless leaves that could contribute to the development of new plant breeding strategies and identification of novel biocontrol agents. The highly susceptible olive variety “Istrska Belica”' was selected for a detailed evaluation. Microscopy analyses led to the observation of raphides in the mesophyll and parenchyma cells of infected leaves and gave new insight into the complex V. oleaginea pathogenesis. Culturable and total phyllosphere mycobiota, obtained via metabarcoding approach, highlighted Didymella, Aureobasidium, Cladosporium, and Alternaria species as overlapping between infected and symptomless leaves. Only Venturia and Erythrobasidium in infected and Cladosporium in symptomless samples with higher abundance showed statistically significant differences. Based on the ecological role of identified taxa, it can be suggested that Cladosporium species might have potential antagonistic effects on V. oleaginea.










Similar content being viewed by others
Data Availability
All data supporting the findings of this study are available within the paper and published online. Data were deposited in the BioProject database (NCBI) as PRJNA779983 (under the accessions from SAMN23098178 to SAMN23098184 for infected samples (S01 to S07), and from SAMN23098185 to SAMN23098191 for symptomless samples (S08 to S14).
References
Loumou A, Giourga C (2003) Olive groves: the life and identity of the Mediterranean. Agric Hum Values 20:87–95. https://doi.org/10.1023/A:1022444005336
Şahin S, Bilgin M (2018) Olive tree (Olea europaea L.) leaf as a waste by-product of table olive and olive oil industry: a review. J Sci Food Agric 98:1271–1279. https://doi.org/10.1002/jsfa.8619
Espeso J, Isaza A, Lee JY, Sörensen PM, Jurado P, Avena-Bustillos RDJ, Olaizola M, Arboleya JC (2021) Olive leaf waste management. Front Sustain Food Syst 5:1–13. https://doi.org/10.3389/fsufs.2021.660582
Clodoveo ML, Crupi P, Annunziato A, Corbo F (2022) Innovative extraction technologies for development of functional ingredients based on polyphenols from olive leaves. Foods 11(1):103. https://doi.org/10.3390/foods11010103
Sergeeva V, Tesoriero L, Spooner-Hart R, Nair NG (2005) First report of Macrophomina phaseolina on olives (Olea europaea) in Australia. Plant Pathol 34:273–274. https://doi.org/10.1071/AP05001
Sergeeva V, Nair NG, Spooner-Hart R (2008) First report of flower infection in olives (Olea europaea) by Colletotrichum acutatum and C. gloeosporioides causing anthracnose disease. Australas Plant Dis Notes 3:81–82
Bernès J (1923) Les parasites de l’olivier au congrès oleicole de Nice. Prog Agric Vitic 80:518–524
Graniti A (1993) Olive scab: a review. Bull 23:377–384. https://doi.org/10.1111/j.1365-2338.1993.tb01339.x
González-Lamothe R, Segura R, Trapero A, Baldoni L, Botella MA, Valpuesta V (2002) Phylogeny of the fungus Spilocaea oleagina, the causal agent of peacock leaf spot in olive. FEMS Microbiol Lett 210:149–155. https://doi.org/10.1111/j.1574-6968.2002.tb11174.x
Viruega JR, Roca LF, Moral J, Trapero A (2011) Factors affecting infection and disease development on olive leaves inoculated with Fusicladium oleagineum. Plant Dis 95:1139–1146. https://doi.org/10.1094/PDIS-02-11-0126
Rongai D, Basti C, Di Marco C (2012) A natural product for the control of olive leaf spot caused by Fusicladium oleagineum (Cast.) Ritschel & Braun. Phytopathol Mediterr 51:276–282. https://www.jstor.org/stable/43871734
Mekuria GT, Collins GG, Sedgley M, Lavee S (2001) Identification of genetic markers in olive linked to olive leaf-spot resistance and susceptibility. J Am Soc Hortic Sci 126:305–308. https://doi.org/10.21273/JASHS.126.3.305
Lanza B, Ragnelli AM, Priore M, Aimola P (2017) Morphological and histochemical investigation of the response of Olea europaea leaves to fungal attack by Spilocaea oleagina. Plant Pathol 66:1239–1247. https://doi.org/10.1111/ppa.12671
Rahioui B, Aissam S, Messaouri H, Moukhli A, Khadari B, El Modafar C (2013) Role of phenolic metabolism in the defense of the olive-tree against leaf-spot disease caused by Spilocaea oleagina. Int J Agric Biol 15:273–278
Bosabalidis AM, Kofidis G (2002) Comparative effects of drought stress on leaf anatomy of two olive cultivars. Plant Sci 163:375–379. https://doi.org/10.1016/S0168-9452(02)00135-8
Bacelar EA, Correia CM, Moutinho-Pereira JM, Gonçalves BC, Lopes JI, Torres-Pereira JM (2004) Sclerophylly and leaf anatomical traits of five field-grown olive cultivars growing under drought conditions. Tree Physiol 24:233–239. https://doi.org/10.1093/treephys/24.2.233
Larbi A, Vázquez S, El-Jendoubi H, Msallem M, Abadía J, Abadía A, Morales F (2015) Canopy light heterogeneity drives leaf anatomical, eco-physiological, and photosynthetic changes in olive trees grown in a high-density plantation. Photosynth Res 123:141–155. https://doi.org/10.1007/s11120-014-0052-2
Khalil HA, El-Ansary DO (2020) Morphological, physiological and anatomical responses of two olive cultivars to deficit irrigation and mycorrhizal inoculation. Eur J Hortic Sci 85:51–62. https://doi.org/10.17660/eJHS.2020/85.1.6
Brito C, Dinis LT, Ferreira H, Moutinho-Pereira J, Correia C (2018) The role of nighttime water balance on Olea europaea plants subjected to contrasting water regimes. J Plant Physiol 226:56–63. https://doi.org/10.1016/j.jplph.2018.04.004
Moreno-Alías I, León L, de la Rosa R, Rapoport HF (2009) Morphological and anatomical evaluation of adult and juvenile leaves of olive plants. Trees 23:181–187. https://doi.org/10.1007/s00468-008-0266-z
Gao M, Xiong C, Gao C, Tsui CK, Wang MM, Zhou X, Zhang AM, Cai L (2021) Disease-induced changes in plant microbiome assembly and functional adaptation. Microbiome 9:1–18. https://doi.org/10.1186/s40168-021-01138-2
Buonaurio R, Moretti C, da Silva DP, Cortese C, Ramos C, Venturi V (2015) The olive knot disease as a model to study the role of interspecies bacterial communities in plant disease. Front Plant Sci 6:434. https://doi.org/10.3389/fpls.2015.00434
Vergine M, Meyer JB, Cardinale M, Sabella E, Hartmann M, Cherubini P, De Bellis L, Luvisi A (2020) The Xylella fastidiosa-resistant olive cultivar “Leccino’” has stable endophytic microbiota during the olive quick decline syndrome (OQDS). Pathogens 9:35. https://doi.org/10.3390/pathogens9010035
Boutaj H, Chakhchar A, Meddich A, Wahbi S, Alaoui-Talibi E, Douira A et al (2020) Bioprotection of olive tree from Verticillium wilt by autochthonous endomycorrhizal fungi. J Plant Dis 127:349–357. https://doi.org/10.1007/s41348-020-00323-z
Ogrin D (2004) Modern climate change in Slovenia. In: Orožen Adamič M (ed) Slovenia: a geographical overview. Association of the Geographical Societies of Slovenia, Ljubljana, pp 45–50
Environmental Agency of the Republic of Slovenia (2006) Podnebne razmere v Sloveniji (obdobje 1971–2000) [Climate conditions in Slovenia (period 1971–2000)]. Ljubljana, https://meteo.arso.gov.si/uploads/probase/www/climate/text/sl/publications/podnebne_razmere_v_sloveniji_71_00.pdf. Accessed 17 Aug 2022
Bandelj D, Darovec D, Kastelic M, Valenčič V (2012) Following olive footprints in Slovenia. In: El-Kholy M (ed) Following olive footprints (Olea europaea L.): cultivation and culture, folklore and history, tradition and uses, Scripta Horticulturae, N. 13. AARINENA, Cairo; IOC, Madrid; ISHS, Leuven, pp 339–350
Lazović B, Klepo T, Adakalić M, Šatović Z, Arbeiter AB, Hladnik M, Strikić F, Liber Z, Bandelj D (2018) Intra-varietal variability and genetic relationships among the homonymic East Adriatic olive (Olea europaea L.) varieties. Sci Hortic 236:175–185. https://doi.org/10.1016/j.scienta.2018.02.053
Ruzin SE (1999) Plant microtechnique and microscopy. Oxford University Press, Oxford
Wolf L (1950) Mikroskopická technika. Státní zdravotnické nakladatelství, Praha
Pearse AGE (1980) Histochemistry theoretical and applied, 4th edn. Longman Group Limited, London
Al-Khatib M, Alhussaen K, El-Banna N, Zyadeh M (2010) Biological control of olive leaf spot (peacock spot disease) caused by Cycloconium oleaginum (Spilocea oleaginea). J Microbiol Antimicrob 2:64–67. https://doi.org/10.5897/JMA.9000018
Salman M, Jawabreh M, Abu Rumaileh B (2014) The effect of local fungicides on conidial germination of Spilocaea oleagina in Palestine. PTURJ 2:26–28. https://doi.org/10.53671/pturj.v2i1.25
Pitt JI (1979) The genus Penicillium and its teleomorphic state Eupenicillium and Talaromyces. Academic Press Inc., Ltd, London
Ellis MB, Ellis PJ (1997) Microfungi on land plants. An identification handbook. The Richmond Publishing Co., Ltd, Oxford
Samson RA, Houbraken J, Thrane U, Frisvad JC, Andersen B (2010) Food and indoor fungi. CBS-KNAW Fungal Biodiversity Centre, Utrecht
Bensch K, Braun U, Groenewald JZ, Crous PW (2012) The genus Cladosporium. Stud Mycol 72:1–401. https://doi.org/10.3114/sim0003
Woudenberg JHC, Groenewald JZ, Binder M, Crous PW (2013) Alternaria redefined. Stud Mycol 75:171–212. https://doi.org/10.3114/sim0015
White TJ, Bruns T, Lee SJWT, Taylor JL (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols - a guide to methods and applications. Academic Press, London, p 482
Unković N, Erić S, Šarić K, Stupar M, Savković Ž, Stanković S, Stanojević O et al (2017) Biogenesis of secondary mycogenic minerals related to wall paintings deterioration process. Micron 100:1–9. https://doi.org/10.1016/j.micron.2017.04.004
Janakiev T, Dimkić I, Unković N, Ljaljević Grbić M, Opsenica D, Gašić U, Stanković S, Berić T (2019) Phyllosphere fungal communities of plum and antifungal activity of indigenous phenazine-producing Pseudomonas synxantha against Monilinia laxa. Front Microbiol 10:2287. https://doi.org/10.3389/fmicb.2019.02287
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Gilbert JA, Jansson JK, Knight R (2018) Earth microbiome project and global systems biology. MSystems 3:e00217-e317. https://doi.org/10.1128/mSystems.00217-17
Hannon GJ (2010) FASTX-Toolkit: FASTQ/A short-reads pre-processing tools, http://hannonlab.cshl.edu/fastx_toolkit/. Accessed June 2021
Abarenkov K, Zirk A, Piirmann T, Pöhönen R, Ivanov F, Nilsson RH, Kõljalg U (2020) UNITE QIIME release for Fungi. UNITE Community. https://doi.org/10.15156/BIO/786385. Accessed June 2021
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC, Al-Ghalith GA et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, Blondel M et al (2011) Scikit-learn: machine learning in python. J Mach Learn Res 12:2825–2830
McMurdie PJ, Holmes S (2013) phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8:e61217
Xu S, Yu G (2021) MicrobiotaProcess: an R package for analysis, visualization and biomarker discovery of microbiome. R package version 1.6.2, https://github.com/YuLab-SMU/MicrobiotaProcess/. Accessed June 2021
Anderson MJ (2005) Permutational multivariate analysis of variance. Department of Statistics, University of Auckland, Auckland, 26:32–46
Gu Z, Eils R, Schlesner M (2016) Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 32:2847–2849. https://doi.org/10.1093/bioinformatics/btw313
Mallick H, Rahnavard A, McIver LJ, Ma S, Zhang Y, Nguyen LH, Tickle TL et al (2021) Multivariable association discovery in population-scale meta-omics studies. PLoS Comput Biol 17:e1009442. https://doi.org/10.1371/journal.pcbi.1009442
Lakušić B, Popov V, Runjaić-Antić D (2007) Morpho-anatomical characteristics of the raw material of the herbal drug Olivae folium and its counterfeits. Arch Biol Sci 59:187–192. https://doi.org/10.2298/ABS0703187L
Roka L, Koudounas K, Daras G, Zoidakis J, Vlahou A, Kalaitzis P, Hatzopoulos P (2018) Proteome of olive non-glandular trichomes reveals protective protein network against (a) biotic challenge. J Plant Physiol 231:210–218. https://doi.org/10.1016/j.jplph.2018.09.016
Tyree EL (1994) Phytolith analysis of olive oil and wine sediments for possible identification in archaeology. Can J Bot 72:499–504. https://doi.org/10.1139/b94-067
Lersten NR, Horner HT (2009) Crystal diversity and macropatterns in leaves of Oleaceae. Plant Syst Evol 282:87–102. https://doi.org/10.1007/s00606-009-0209-1
Alquati P (1906) Studi anatomici e morfologici sull’ulivo (Olea europaea). Atti della Societa` Lingustica di Scienze Naturali e Geografiche. Genova 17:124–148
Schneider G, Löbenberg L (1972) Zur Morphologie und anatomie der Olivenblätter. Planta Med 22:117–121. https://doi.org/10.1055/s-0028-1099592
Christodoulakis NS (1992) Structural diversity and adaptations in some Mediterranean evergreen sclerophyllous species. Environ Exp Bot 32:295–305. https://doi.org/10.1016/0098-8472(92)90012-Q
Stefi AL, Vassilacopoulou D, Routsi E, Stathopoulos P, Argyropoulou A, Skaltsounis AL, Christodoulakis NS (2021) The combined environmental stress on the leaves of Olea europaea L. and the relief mechanism through biosynthesis of certain secondary metabolites. J Plant Growth Regul 40:1044–1059. https://doi.org/10.1007/s00344-020-10162-9
Christodoulakis NS, Koutsogeorgopoulou L (1991) Air pollution effects on the leaf structure of two injury resistant species: Eucalyptus camaldulensis and Olea europaea L. Bull Environ Contam Toxicol 47:433–439
de Oliveira CG, Macêdo JNA, Santos TB, Alemanno L, da Silva GA, Micheli F, Mariano AC, Peres Gramacho K, da Costa SD, Meinhardt L, Mazzafera P, Guimaraes Pereira GA, de Mattos Cascardo JC (2007) Involvement of calcium oxalate degradation during programmed cell death in Theobroma cacao tissues triggered by the hemibiotrophic fungus Moniliophthora perniciosa. Plant Sci 173:106–117. https://doi.org/10.1016/j.plantsci.2007.04.006
Hu X, Bidney DL, Yalpani N, Duvick JP, Crasta O, Folkerts O, Lu G (2003) Overexpression of a gene encoding hydrogen peroxide-generating oxalate oxidase evokes defense responses in sunflower. Plant Physiol 133:170–181. https://doi.org/10.1104/pp.103.024026
Jennings JM, Apel-Birkhold PC, Mock NM, Baker CJ, Anderson JD, Bailey BA (2001) Induction of defense responses in tobacco by the protein Nep 1 from Fusarium oxysporum. Plant Sci 161:891–899. https://doi.org/10.1016/S0168-9452(01)00483-6
Novelli S, Gismondi A, Di Marco G, Canuti L, Nanni V, Canini A (2019) Plant defense factors involved in Olea europaea resistance against Xylella fastidiosa infection. J Plant Res 132:439–455. https://doi.org/10.1007/s10265-019-01108-8
Báidez AG, Gómez P, Del Río JA, Ortuño A (2007) Dysfunctionality of the xylem in Olea europaea L. plants associated with the infection process by Verticillium dahliae Kleb. Role of phenolic compounds in plant defense mechanism. J Agric Food Chem 55:3373–3377. https://doi.org/10.1021/jf063166d
Viruega JR, Moral J, Roca LF, Navarro N, Trapero A (2013) Spilocaea oleagina in olive groves of southern Spain: survival, inoculum production, and dispersal. Plant Dis 97:1549–1556. https://doi.org/10.1094/PDIS-12-12-1206-RE
Gomes T, Pereira JA, Benhadi J, Lino-Neto T, Baptista P (2018) Endophytic and epiphytic phyllosphere fungal communities are shaped by different environmental factors in a Mediterranean ecosystem. Microb Ecol 76:668–679. https://doi.org/10.1007/s00248-018-1161-9
Scibetta S, Agosteo GE, Abdelfattah A, Li Destri Nicosia MG, Cacciola SO, Schena L (2020) Development and application of a quantitative PCR detection method to quantify Venturia oleaginea in asymptomatic olive (Olea europaea) leaves. Phytopathology 110:547–555. https://doi.org/10.1094/PHYTO-07-19-0227-R
Abdelfattah A, Li Destri Nicosia MG, Cacciola SO, Droby S, Schena L (2015) Metabarcoding analysis of fungal diversity in the phyllosphere and carposphere of olive (Olea europaea). PLoS ONE 10:e0131069. https://doi.org/10.1371/journal.pone.0131069
Varanda CM, Materatski P, Landum M, Campos MD, Félix MDR (2019) Fungal communities associated with peacock and cercospora leaf spots in olive. Plants 8:169. https://doi.org/10.3390/plants8060169
Hudson HJ (1971) The development of the saprophytic fungal flora as leaves senesce and fall. In: Preece TF, Dickinson CH (eds) Ecology of leaf surface microorganisms. Academic Press, London, pp 447–455
Chliyeh M, Rhimini Y, Selmaoui K, Ouazzani Touhami A, Filali-Maltouf A, El Modafar CC, Moukhli A, Oukabli A, Benkirane R, Douira A (2014) Survey of the fungal species associated to olive-tree (Olea europaea L.) in Morocco. Int J Biotechnol 2:15–32
Landum MC, Félix MR, Alho J, Garcia R, Cabrita MJ, Rei F, Varanda CMR (2016) Antagonistic activity of fungi of Olea europaea L. against Colletotrichum acutatum. Microbiol Res 183:100–108. https://doi.org/10.1016/j.micres.2015.12.001
Sanei SJ, Razavi SE (2012) Survey of olive fungal disease in North of Iran. Annu. Res. Rev. Biol 2:27–36. https://journalarrb.com/index.php/ARRB/article/view/26473
Moral J, de la Rosa R, León L, Barranco D, Michailides TJ, Trapero A (2008) High susceptibility of the olive cultivar FS-17 to Alternaria alternata in southern Spain. Plant Dis 92:1252. https://doi.org/10.1094/PDIS-92-8-1252A
Lagogianni CS, Tjamos EC, Antoniou PP, Tsitsigiannis DI (2017) First report of Alternaria alternata as the causal agent of Alternaria bud and blossom blight of olives. Plant Dis 101:2151. https://doi.org/10.1094/PDIS-04-17-0527-PDN
Bottalico A, Logrieco A (2008) Mycotoxins in Alternaria alternata infected olive fruits and their possible transfer into oil. Bull 23:473–479. https://doi.org/10.1111/j.1365-2338.1993.tb01355.x
Yamagishi N, Nishikawa J, Oshima Y, Eguchi N (2009) Black spot disease of alstroemeria caused by Alternaria alstroemeriae in Japan. J Gen Plant Pathol 75:401–403. https://doi.org/10.1007/s10327-009-0182-0
Valdés I, Rodríguez J, Portela A, Jiménez P (2014) First report of Alternaria alstroemeriae on Alstroemeria sp. in Colombia. New Dis Rep 29:21–21. https://doi.org/10.5197/j.2044-0588.2014.029.021
Malacrinò A, Schena L, Campolo O, Laudani F, Mosca S, Giunti G, Strano CP, Palmeri V (2017) A metabarcoding survey on the fungal microbiota associated to the olive fruit fly. Microb Ecol 73:677–684. https://doi.org/10.1007/s00248-016-0864-z
Preto G, Martins F, Pereira JA, Baptista P (2017) Fungal community in olive fruits of cultivars with different susceptibilities to anthracnose and selection of isolates to be used as biocontrol agents. Biol Control 110:1–9. https://doi.org/10.1016/j.biocontrol.2017.03.011
Gomes T, Pereira JA, Lino-Neto T, Bennett AE, Baptista P (2019) Bacterial disease induced changes in fungal communities of olive tree twigs depend on host genotype. Sci Rep 9:1–10. https://doi.org/10.1038/s41598-019-42391-8
Chliyeh M, Achbani E, Rhimini Y, Selmaoui K, Touhami AO, Filali-Maltouf A, El Modafar C, Moukhli A, Oukabli A, Benkirane R, Douira A (2014) Pathogenicity of four fungal species on fruits and leaves of the olive tree (Olea europaea L.). Int J Pure App Biosci 2:1–9
Prior R, Feige A, Begerow D (2017) Antagonistic activity of the phyllosphere fungal community. Sydowia 69:183–198. https://doi.org/10.12905/0380.sydowia69-2017-0183
Răut I, Călin M, Capră L, Gurban AM, Doni M, Radu N, Jecu L (2021) Cladosporium sp. isolate as fungal plant growth promoting agent. Agronomy 11:392. https://doi.org/10.3390/agronomy11020392
Chaudhry V, Runge P, Sengupta P, Doehlemann G, Parker JE, Kemen E (2021) Shaping the leaf microbiota: plant–microbe–microbe interactions. J Exp Bot 72:36–56. https://doi.org/10.1093/jxb/eraa417
Zalar P, Gostinčar C, De Hoog GS, Uršič V, Sudhadham M, Gunde-Cimerman N (2008) Redefinition of Aureobasidium pullulans and its varieties. Stud Mycol 61:21–38. https://doi.org/10.3114/sim.2008.61.02
Materatski P, Varanda C, Carvalho T, Dias AB, Campos MD, Rei F, do Rosário Félix M, (2019) Spatial and temporal variation of fungal endophytic richness and diversity associated to the phyllosphere of olive cultivars. Fungal Ecol 123:66–76. https://doi.org/10.1016/j.funbio.2018.11.004
Nicoletti R, Di Vaio C, Cirillo C (2020) Endophytic fungi of olive tree. Microorganisms 8:1321. https://doi.org/10.3390/microorganisms8091321
Pihet M, Vandeputte P, Tronchin G, Renier G, Saulnier P, Georgeault S, Mallet R, Chabasse D, Symoens F, Bouchara JP (2009) Melanin is an essential component for the integrity of the cell wall of Aspergillus fumigatus conidia. BMC Microbiol 9:1–11. https://doi.org/10.1186/1471-2180-9-177
Janisiewicz WJ, Jurick WM, Peter KA, Kurtzman CP, Buyer JS (2014) Yeasts associated with plums and their potential for controlling brown rot after harvest. Yeast 31:207–218. https://doi.org/10.1002/yea.3009
Kunz S (2004) Development of “Blossom-Protect” - a yeast preparation for the reduction of blossom infections by fire blight. In: Boos M (ed) Ecofruit - 11th International Conference on Cultivation Technique and Phytopathological Problems in Organic Fruit-Growing. Weinsberg, Germany, pp 108–112
de Jong H, Reglinski T, Elmer PA, Wurms K, Vanneste JL, Guo LF, Alavi M (2019) Integrated use of Aureobasidium pullulans strain CG163 and acibenzolar-S-methyl for management of bacterial canker in kiwifruit. Plants 8:287. https://doi.org/10.3390/plants8080287
de Freitas SE, Marcon J, Mazzer Luvizotto D, Quecine MC, Tsui S, Pereira JO, Pizzirani-Kleiner AA, Azevedo JL (2013) Endophytic fungi from the Amazonian plant Paullinia cupana and from Olea europaea isolated using cassava as an alternative starch media source. Springerplus 2:579. https://doi.org/10.1186/2193-1801-2-579
Giampetruzzi A, Baptista P, Morelli M, Cameirão C, Lino Neto T, Costa D, D’Attoma G, Abou Kubaa R, Altamura G, Saponari M, Pereira JA, Saldarelli P (2020) Differences in the endophytic microbiome of olive cultivars infected by Xylella fastidiosa across seasons. Pathogens 9:723. https://doi.org/10.3390/pathogens9090723
Arroyo-López FN, Querol A, Bautista-Gallego J, Garrido-Fernández A (2008) Role of yeasts in table olive production. Int J Food Microbiol 128:189–196. https://doi.org/10.1016/j.ijfoodmicro.2008.08.018
Dzoyem JP, Melong R, Tsamo AT, Maffo T, Kapche DG, Ngadjui BT, McGaw LJ, Eloff JN (2017) Cytotoxicity, antioxidant and antibacterial activity of four compounds produced by an endophytic fungus Epicoccum nigrum associated with Entada abyssinica. Rev Bras Farmacogn 27:251–253. https://doi.org/10.1016/j.bjp.2016.08.011
Jouda JB, Mbazoa CD, Sarkar P, Bag PK, Wandji J (2016) Anticancer and antibacterial secondary metabolites from the endophytic fungus Penicillium sp. CAM64 against multi-drug resistant Gram-negative bacteria. Afr Health Sci 16:734–743. https://doi.org/10.4314/ahs.v16i3.13
Funding
This work was supported by the Ministry of Education, Science, and Technological developments of the Republic of Serbia and by the Slovenian Research Agency, as well as by bilateral scientific-research cooperation between SRB and RS [Contracts Nos.: 451–03-68/2022–14/200178; P1-0386; SLO-SRB-2018/19-OAN].
Author information
Authors and Affiliations
Contributions
MH, DB, and ID—conceptualization of idea and data curation; MH, NU, TJ, ABA, MLG, PJ, MG, DR, and ID—methodology and formal analysis; DB, SS, and ID—funding acquisition; MH, and ID—supervision; MH, NU, TJ, MLG, PJ, MG, and ID—writing the original draft; MH, NU, TJ, MLG, ABA, SS, PJ, MG, DR, DB, and ID—review, editing, and approval of the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
A study was not required ethics approval.
Conflict of Interests
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hladnik, M., Unković, N., Janakiev, T. et al. An Insight into an Olive Scab on the “Istrska Belica” Variety: Host‐Pathogen Interactions and Phyllosphere Mycobiome. Microb Ecol 86, 1343–1363 (2023). https://doi.org/10.1007/s00248-022-02131-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-022-02131-4