Abstract
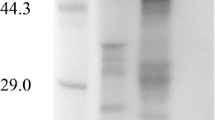
A highly efficient β-1,4-glucosidase (BGL) secreting strain, Stereum hirsutum SKU512, was isolated and identified based on morphological features and sequence analysis of internal transcribed spacer rDNA. A BGL containing a carbohydrate moiety was purified to homogeneity from S. hirsutum culture supernatants using only a single chromatography step on a gel filtration column. The relative molecular weight of S. hirsutum BGL was determined as 98 kDa by sodium dodecyl sulfate polyacrylamide gel electrophoresis or 780 kDa by size exclusion chromatography, indicating that the enzyme is an octamer. S. hirsutum BGL showed the highest activity toward p-nitrophenyl-β-D-glucopyranoside (V max = 3,028 U mg-protein−1, k cat = 4,945 s−1) ever reported. The enzyme also showed good stability at an acidic pH ranging from 3.0 to 5.5. The BGL was able to promote transglycosylation with an activity of 42.9 U mg-protein−1 using methanol as an acceptor and glucose as a donor. The internal amino acid sequences of the isolated enzyme showed significant homology with hydrolases from glycoside hydrolase family 1 (GH1), indicating that the S. hirsutum BGL is a member of GH1 family. The characteristics of S. hirsutum BGL could prove to be of interest in several potential applications, especially in enhancing flavor release during the wine fermentation process.







Similar content being viewed by others
References
Amouri B, Gargouri A (2006) Characterization of a novel β-glucosidase from a Stachybotrys strain. Biochem Eng J 32:191–197
Baldrian P, Valaskova V (2008) Degradation of cellulose by basidiomycetous fungi. FEMS Microbiol Rev 32:501–521
Barbagallo RN, Spagna G, Palmeri R, Restuccia C, Giudici P (2004) Selection, characterization and comparison of β-glucosidase from mould and yeasts employable for enological applications. Enzyme Microb Technol 35:58–66
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cao W, Crawford DL (1993) Purification and some properties of β-glucosidase from the ectomycorrhizal fungus Pisolithus tinctorius strain SMF. Can J Microbiol 39:125–129
Christakopoulos P, Goodenough PW, Kekos D, Macris BJ, Claeyssens M, Bhat MK (1994) Purification and characterisation of an extracellular β-glucosidase with transglycosylation and exo-glucosidase activities from Fusarium oxysporum. Eur J Biochem 224:379–385
Christine RJ-MS, Marie-Jose V, Ziya G, Pierre B (1998) Purification, characterization, and substrate specificity of a novel highly glucose-tolerant β-glucosidase from Aspergillus oryzae. Appl Environ Microbiol 64:3607–3614
Gunata YS, Bayonove CL, Tapiero C, Cordonnier RE (1990) Hydrolysis of grape monterpenyl β-D-glucosides by various β-glucosidases. J Agric Food Chem 38:1232–1236
Joo AR, Jeya M, Lee KM, Sim WI, Kim JS, Kim IW, Kim YS, Oh DK, Gunasekaran P, Lee JK (2009) Purification and characterization of a β-1, 4-glucosidase from a newly isolated strain of Fomitopsis pinicola. Appl Microbiol Biotechnol 83:285–294
Kaur J, Chadha BS, Kumar BA, Ghatora SK, Saini HS (2007) Purification and characterization of β-glucosidase from Melanocarpus sp. MTCC 3922. Electron J Biotechnol 10:260–270
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Leite RSR, Gomes E, Da Silva R (2007) Characterization and comparison of thermostability of purified β-glucosidases from a mesophilic Aureobasidium pullulans and a thermophilic Thermoascus aurantiacus. Proc Biochem 42:1101–1106
Lin J, Ndlovu LM, Singh S, Pillay B (1999) Purification and biochemical characteristics of β-D-xylanase from a thermophilic fungus, Thermomyces lanuginosus-SSBP. Biotechnol Appl Biochem 30:73–79
Magalhaes PO, Ferraz A, Milagres AFM (2006) Enzymatic properties of two β-glucosidases from Ceriporiopsis subvermispora produced in biopulping conditions. J Appl Microbiol 101:480–486
McHale A, Coughlan MP (1982) Properties of the β-glucosidases of Talaromyces emersonii. Microbiology 128:2327
Miller GL (1959) Use of dinitrosalycilic acid for determining reducing sugars. Anal Chem 31:426–428
O'Farrell PH (1975) High resolution two-dimensional electrophoresis of proteins. J Biol Chem 250:4007–4021
Parry NJ, Beever DE, Owen E, Vandenberghe I, Van Beeumen J, Bhat MK (2001) Biochemical characterization and mechanism of action of a thermostable β-glucosidase purified from Thermoascus aurantiacus. Biochem J 353:117
Pemberton M, Brown R Jr, Emert G (1980) Role of β-glucosidase in the bioconversion of cellulose to ethanol. Can J Chem Eng 58:723–729
Pérez-Pons JA, Rebordosa X, Querol E (1995) Properties of a novel glucose-enhanced β-glucosidase purified from Streptomyces sp. ATCC 11238. Biochim Biophys Acta 1251:145–153
Plant AR, Oliver JE, Patchett ML, Daniel RM, Morgan HW (1988) Stability and substrate specificity of a β-glucosidase from the thermophilic bacterium Tp8 cloned into Escherichia coli. Arch Biochem Biophys 262:181
Riou C, Salmon JM, Vallier MJ, Gunata Z, Barre P (1998) Purification, characterization and substrate specificity of a novel highly glucose-tolerant β-glucosidase from Aspergillus oryzae. Appl Environ Microbiol 64:3607–3614
Rojas A, Arola L, Romeu A (1995) β-glucosidase families revealed by computer analysis of protein sequences. Biochem Mol Biol Int 35:1223
Saha BC, Bothast RJ (1996) Production, purification, and characterization of a highly glucose-tolerant novel β-glucosidase from Candida peltata. Appl Environ Microbiol 62:3165–3170
Valaskova V, Baldrian P (2006) Degradation of cellulose and hemicelluloses by the brown rot fungus Piptoporus betulinus-production of extracellular enzymes and characterization of the major cellulases. Microbiology 152:3613
Wei DL, Kirimura K, Usami S, Lin TH (1996) Purification and characterization of an extracellular β-glucosidase from the wood-grown fungus Xylaria regalis. Curr Microbiol 33:297–301
White TJ, Bruns T, Lee S, Taylor J, Innis MA, Gelfand DH, Sninsky JJ (1990) PCR protocols: a guide to methods and applications. PCR protocols: a guide to methods and applications 42
Workman WE, Day DF (1982) Purification and properties of β-glucosidase from Aspergillus terreus. Appl Environ Microbiol 44:1289
Yan TR, Lin CL (1997) Purification and characterization of a glucose-tolerant β-glucosidase from Aspergillus niger CCRC 31494. Biosci Biotechnol Biochem 61:965–970
Yoon JJ, Kim KY, Cha CJ (2008) Purification and characterization of thermostable β-glucosidase from the brown-rot basidiomycete Fomitopsis palustris grown on microcrystalline cellulose. J Microbiol 46:51–55
Acknowledgment
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST; 220-2009-1-D00033). This work was also supported by a grant a grant (S210707L010120) from Korea Forest Service, Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nguyen, NPT., Lee, KM., Lee, KM. et al. One-step purification and characterization of a β-1,4-glucosidase from a newly isolated strain of Stereum hirsutum . Appl Microbiol Biotechnol 87, 2107–2116 (2010). https://doi.org/10.1007/s00253-010-2668-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2668-2