Abstract
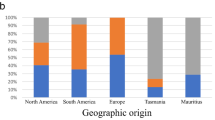
Stereocaulon alpinum is a lichenized fungus commonly found in the Antarctic, circumarctic, alpine, and in mountainous areas of temperate regions. It forms a tripartite lichen symbiosis together with trebouxioid green microalgae, as its primary photobiont, and cyanobacteria of the genus Nostoc inside delimited cephalodia. Previous DNA-based analyses revealed two lineages for this fungus. This research discusses the morphological and chemical circumscription of S. alpinum and its geographic distribution and, to this end, we included new DNA data from the mycobiont and its photosynthetic partners. Phylogenetic reconstructions rejected infraspecific categories and revealed that specimens from the southern hemisphere (maritime Antarctica and South America) formed a well-supported “bipolar clade” with specimens from Finland, Greenland, and Canada. In addition, S. alpinum also formed a “northern clade” with sequences from Europe and North Atlantic islands. In specimens from the southern hemisphere, the identified lineages of phyco- and cyanobionts were already known to be associated with other lichenized fungi. The phycobiont was identified as Asterochloris pseudoirregularis and the cyanobiont as a member of the genus Nostoc. The phenotypic differences among the southern hemisphere specimens examined in this study pointed towards a broad morphological circumscription of S. alpinum. Simultaneously, the available descriptions of S. alpinum collected in the northern hemisphere, including the type specimen, do not provide characters distinguishing them from the southern specimens. These two clades likely represent cryptic species, one new and potentially only recognized using genetic data. Additional integrative studies, including a comprehensive analysis of specimens from the northern hemisphere, are necessary to know the diversity contained in the lichens identified as Stereocaulon alpinum worldwide.





Similar content being viewed by others
Data availability
All specimens studied here are available in the CGMS herbarium (http://cgms.jbrj.gov.br/v2/consulta.php) as well as the sequences generated in the Genbank database (https://www.ncbi.nlm.nih.gov/genbank/). See voucher information and accession numbers in Online Resource 1.
References
Bandelt HJ, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Brodo IM (2016) Keys to lichens of North America: revised and expanded. Yale University Press, New Haven and London
Brodo IM, Duran-Sharnoff S, Sharnoff S (2001) Lichens of North America. Yale University Press, New Haven and London
Chagnon PC, Magain N, Miadlikowska J, Lutzoni F (2019) Species diversification and phylogenetically constrained symbiont switching generated high modularity in the lichen genus Peltigera. J Ecol 107:1645–1661. https://doi.org/10.1111/1365-2745.13207
Colesie C, Büdel B, Hurry V, Green TGA (2018) Can Antarctic lichens acclimatize to changes in temperature? Glob Chang Biol 24:1123–1135. https://doi.org/10.1111/gcb.13984
Dahl E, Krog H (1973) Macrolichens of Denmark, Finland, Norway and Sweden. Universitatesflorlaget, Oslo, Bergen, Tromso
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
Dogde CW (1929) A synopsis of Stereocaulon with notes on some exotic species. Ann Crypt Exot 2:93–153
Ekman S, Tonsberg T (2002) Most species of Lepraria and Leproloma form a monophyletic group closely related to Stereocaulon. Mycol Res 106:1262–1276. https://doi.org/10.1017/S0953756202006718
Elvebakk A, Papaefthimiou D, Robertsen EH, Liaimer A (2008) Phylogenetic patterns among nostoc cyanobionts within bi- and tripartite lichens of the genus Pannaria. J Phycol 44:1049–1059. https://doi.org/10.1111/j.1529-8817.2008.00556.x
Fernández-Mendoza F, Printzen C (2013) Pleistocene expansion of the bipolar lichen Cetraria aculeata into the Southern hemisphere. Mol Ecol 22:1961–1983. https://doi.org/10.1111/mec.12210
Frey E (1969). In: Poelt J (ed) Bestimmungsschlüssel europäischer Flechten. Verlag, J Cramer, Lehre, pp 1–757
Frey E (1932) Cladoniaceae (unter Ausschluss der Gattung Cladonia) Umbilicariaceae. In: Rabenhorst L, Grunow A (eds) Dr. L. Rabenhorst’s Kryptogamen-Flora von Deutschland, Österreich und der Schweiz. 2nd edn. Bd. 9. Leipzig, pp 1–426
Fries TM (1858) Monographia Stereocaulorum et Pilophororum. Acta Regia Soc Sci Upsal 3:307–380
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Hawksworth DL, Grube M (2020) Lichens redefined as complex ecosystems. New Phytol 227:1–3. https://doi.org/10.1111/nph.16630
Högnabba F (2006) Molecular phylogeny of the genus Stereocaulon (Stereocaulaceae, lichenized ascomycetes). Mycol Res 110:1080–1092. https://doi.org/10.1016/j.mycres.2006.04.013
Högnabba F, Pino-bodas R, Nordin A, Myllys L, Stenroos S (2014) Phylogenetic position of the crustose Stereocaulon species. Lichenologist 46:103–114
Holzmann G, Leuckert C (1990) Applications of negative fast atom bombardment and MS/MS to screening of lichen compounds. Phytochemistry 29:2277–2283. https://doi.org/10.1016/0031-9422(90)83052-3
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755. https://doi.org/10.1093/bioinformatics/17.8.754
Hüneck S, Yoshimura I (1996) Identification of lichen substances. Springer, Berlin
Johnson GT (1938) The Taxonomic Importance and phylogenetic significance of the cephalodia of Stereocaulon. Ann Mo Bot Gard 25:729–768
Kaasalainen U, Olsson S, Rikkinen J (2015) Evolution of the tRNALeu (UAA) intron and congruence of genetic markers in lichen-symbiotic nostoc. PLoS ONE 10:e0131223. https://doi.org/10.1371/journal.pone.0131223
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30:3059–3066. https://doi.org/10.1093/nar/gkf436
Kershaw KA (1960) The genus Stereocaulon Schreb. in the British Isles. Lichenologist 1:184–203
Kim JH, Ahn I-Y, Hong SG, Andreev M, Lim K-M, Oh MJ, Koh YJ, Hur J-S (2006) Lichen flora around the Korean Antarctic scientific station, King George Island, Antarctic. J Microbiol 44:480–491
Kim JI, Kim YJ, Nam SW, So JE, Hong SY, Choi H-G, Shin W (2020) Taxonomic study of three new Antarctic Asterochloris (Trebouxiophyceae) based on morphological and molecular data. Algae 35:17–32. https://doi.org/10.4490/algae.2020.35.2.23
Kistenich S, Timdal E, Bendiksby M, Ekman S (2018) Molecular systematics and character evolution in the lichen family Ramalinaceae (Ascomycota: Lecanorales). Taxon 67:871–904
Kivistö L (1998) Taxonomy of Stereocaulon paschale and allied species in finland. Sauteria 9:25–36
Kosecka M, Krzemińska BG, Černajová I, Škaloud P, Jabłońska A, Kukwa M (2021) New lineages of photobionts in Bolivian lichens expand our knowledge on habitat preferences and distribution of Asterochloris algae. Sci Rep 11:8701. https://doi.org/10.1038/s41598-021-88110-0
Lagostina E, Andreev M, Dal Grande F, Grewe F, Lorenz A, Lumbsch HT, Rozzi R, Ruprecht U, Sancho LG, Søchting U, Scur M, Wirtz N, Printzen C (2021) Effects of dispersal strategy and migration history on genetic diversity and population structure of Antarctic lichens. J Biogeogr 48:1635–1653. https://doi.org/10.1111/jbi.14101
Lamb IM (1951) On the morphology, phylogeny, and taxonomy of the lichen genus Stereocaulon. Can J Bot 29:522–584. https://doi.org/10.1139/b51-045
Lamb IM (1958) La Vegetación Liquénica de los Parques Nacionales Patagónicos. Anales de Parques Nacionales, Buenos Aires
Lamb IM (1977) A conspectus of the lichen genus Stereocaulon (Schreb.) Hoffm. Journ Hattori Bot Lab 43:191–355
Lamb IM (1978) Keys to the species of the lichen genus Stereocaulon (Schreb.) Hoffm. Journ Hattori Bot Lab 44:209–250
Lavoie C, Renaudin M, McMullin RT, Gagnon J, Roy C, Beaulieu M-E, Bellenger JP, Villarreal JC (2020) Extremely low genetic diversity of Stigonema associated with Stereocaulon in eastern Canada. Bryologist 123:188–203. https://doi.org/10.1639/0007-2745-123.2.188
Lee JR, Raymond B, Bracegirdle TJ, Chadès I, Fuller RA, Shaw JD, Terauds A (2017) Climate change drives expansion of Antarctic ice-free habitat. Nature 547:49–54. https://doi.org/10.1038/nature22996
Lewis-Smith RI (1984) Terrestrial plant biology of the sub-Antarctic and Antarctic. In: Laws RM (ed) Antarctic ecology, vol 1. Academic Press, London, pp 61–162
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Lindsay DC (1974) The macrolichens of South Georgia. Brit Antarct Survey Sci Repts 89:1–91
Lohtander K, Oksanen I, Rikkinen J (2003) Genetic diversity of green algal and cyanobacterial photobionts in Nephroma (Peltigerales). Lichenologist 35:325–339. https://doi.org/10.1016/S0024-2829(03)00051-3
Lücking R, Lawrey JD, Sikaroodi M, Gillevet PM, Chaves JL, Sipman HJM, Bungartz F (2009) Do lichens domesticate photobionts like farmers domesticate crops? Evidence from a previously unrecognized lineage of filamentous cyanobacteria. Am J Bot 96:1409–1418. https://doi.org/10.3732/ajb.0800258
Lücking R, Dal Forno M, Sikaroodi M, Gillevet PM, Bungartz F, Moncada B, Yánez-Ayabaca A, Coca LF, Chaves JD, Lawrey JD (2014) A single macrolichen constitutes hundreds of unrecognized species. Proc Natl Acad Sci USA 111:11091–11096
Lutzoni F, Miadlikowska J (2009) Lichens. Curr Biol 19:R502–R503. https://doi.org/10.1016/j.cub.2009.04.034
Magain N, Miadlikowska J, Goffinet B, Sérusiaux E, Lutzoni F (2017) Macroevolution of specificity in cyanolichens of the genus Peltigera Section Polydactylon (Lecanoromycetes, Ascomycota). Syst Biol 66:74–99. https://doi.org/10.1093/sysbio/syw065
Magain N, Truong C, Goward T, Niu D, Goffinet B, Sérusiaux E, Lutzoni F, Miadlikowska J (2018) Species delimitation at a global scale reveals high species richness with complex biogeography and patterns of symbiont association in Peltigera section Peltigera (lichenized Ascomycota: Lecanoromycetes). Taxon 67:836–870
Magnusson AH (1926) Studies on boreal Stereocaula. Göteborgs Kungl. Vetenskaps- Och Vitterhets-Samhälles Handlingar Handl 30:1–89
McCune B, Di Meglio E, Tønsberg T, Yahr R (2019) Five new crustose Stereocaulon species in western North America. Bryologist 122:197–218. https://doi.org/10.1639/0007-2745-122.2.197
Mercado-Díaz JA, Lücking R, Moncada B, Widhelm TJ, Lumbsch HT (2020) Elucidating species richness in lichen fungi: the genus Sticta (Ascomycota: Peltigeraceae) in Puerto Rico. Taxon 69:851–891. https://doi.org/10.1002/tax.12320
Miller MA, Pfeiffer WT, Schwartz T (2010) Creating the CIPRES science gateway for inference of large phylogenetic trees. In: Gateway computing environments workshop (GCE). pp 1–8. https://doi.org/10.1109/GCE.2010.5676129
Musharraf SG, Kanwal N, Thadhani VM, Choudlary ML (2015) Rapid identification of lichen compounds based on the structure–fragmentation relationship using ESI-MS/MS analysis. Anal Methods 7:6066–6076. https://doi.org/10.1039/C5AY01091H
Myllys L, Lohtander K, Tehler A (2001) β-tubulin, ITS and group I intron sequences challenge the species pair concept in Physcia aipolia and P. caesia. Mycologia 93:335–343. https://doi.org/10.2307/3761655
Nelsen MP, Gargas A (2006) Actin type I introns offer potential for increasing phylogenetic resolution in Asterochloris (Chlorophyta: Trebouxiophyceae). Lichenologist 38:435–440. https://doi.org/10.1017/S0024282906005779
Nelsen MP, Gargas A (2008) Dissociation and horizontal transmission of codispersing lichen symbionts in the genus Lepraria (Lecanorales: Stereocaulaceae). New Phytol 177:264–275. https://doi.org/10.1111/j.1469-8137.2007.02241.x
O’Brien HE, Miadlikowska J, Lutzoni F (2005) Assessing host specialization in symbiotic cyanobacteria associated with four closely related species of the lichen fungus Peltigera. Eur J Phycol 40:363–378. https://doi.org/10.1080/09670260500342647
Oset M (2014) The lichen genus Stereocaulon (Schreb.) Hoffm. in Poland.—a taxonomic and ecological study. Monogr Bot 104:1–81. https://doi.org/10.5586/mb.2014.001
Øvstedal DO, Lewis-Smith RI (2001) Lichens of Antarctica and South Georgia: a guide to their identification and ecology. Cambridge
Pardo-De la Hoz CJ, Magain N, Lutzoni F, Goward T, Restrepo S, Miadlikowska J (2018) Contrasting symbiotic patterns in two closely related lineages of trimembered lichens of the genus peltigera. Front Microbiol 9:1–14. https://doi.org/10.3389/fmicb.2018.02770
Park JS, Park CH, Park SY, Oh SO, Jayalal U, Hur JS (2018) Revision of the lichen genus Stereocaulon (Stereocaulaceae, Ascomycota) in South Korea. Mycobiology 46:101–113. https://doi.org/10.1080/12298093.2018.1461314
Paudel B, Bhattarai HD, Lee JS, Hong SG, Shin HW, Yim JH (2008) Antioxidant activity of polar lichens from King George Island (Antarctica). Polar Biol 31:605–608. https://doi.org/10.1007/s00300-007-0394-8
Paulsrud P, Rikkinen J, Lindblad P (2000) Spatial patterns of photobiont diversity in some Nostoc-containing lichens. New Phytol 146:291–299. https://doi.org/10.1046/j.1469-8137.2000.00647.x
Peat HJ, Clarke A, Convey P (2007) Diversity and biogeography of the Antarctic flora. J Biogeogr 34:132–146. https://doi.org/10.1111/j.1365-2699.2006.01565.x
Peksa O, Škaloud P (2011) Do photobionts influence the ecology of lichens? A case study of environmental preferences in symbiotic green alga Asterochloris (Trebouxiophyceae). Mol Ecol 20:3936–3948. https://doi.org/10.1111/j.1365-294X.2011.05168.x
Quilhot W, Cuellar M, Díaz R, Riquelme F, Rubio C (2012) Lichens of aisen, Southern Chile. Gayana Bot 69:57–87
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using tracer 1.7. Syst Biol. https://doi.org/10.1093/sysbio/syy032
Riddle LW (1910) The North American species of Stereocaulon. Bot Gaz 50:285–304. https://doi.org/10.1086/330363
Rikkinen J, Oksanen I, Lohtander K (2002) Lichen guilds share related cyanobacterial symbionts. Science 297:357. https://doi.org/10.1126/science.1072961
Ruprecht U, Fernández-Mendoza F, Türk R, Fryday AM (2020) High levels of endemism and local differentiation in the fungal and algal symbionts of saxicolous lecideoid lichens along a latitudi-nal gradient in southern South America. Lichenologist 52:287–303. https://doi.org/10.1017/S0024282920000225
Sancho LG, Pintado A, Green A (2019) Antarctic studies show lichens to be excellent biomonitors of climate change. Diversity 11:1–14. https://doi.org/10.3390/d11030042
Singh SM, Olech M, Cannone N, Convey P (2015) Contrasting patterns in lichen diversity in the continental and maritime Antarctic. Polar Sci 9:311–318. https://doi.org/10.1016/j.polar.2015.07.001
Škaloud P, Peksa O (2010) Evolutionary inferences based on ITS rDNA and actin sequences reveal extensive diversity of the common lichen alga Asterochloris (Trebouxiophyceae, Chlorophyta). Mol Phylogenet Evol 54:36–46. https://doi.org/10.1016/j.ympev.2009.09.035
Søchting U, Castello M (2012) The polar lichens Caloplaca darbishirei and C. soropelta highlight the direction of bipolar migration. Polar Biol 35:1143–1149. https://doi.org/10.1007/s00300-012-1161-z
Spielmann AA, Pereira AB (2012) Lichens on the maritime Antarctica (a small field guide for some common species). Glalia 4:1–28
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313. https://doi.org/10.1093/bioinformatics/btu033
Stenroos S, Högnabba F, Myllys L, Hyvönen J, Thell A (2006) High selectivity in symbiotic associations of lichenized ascomycetes and cyanobacteria. Cladistics 22:230–238. https://doi.org/10.1111/j.1096-0031.2006.00101.x
Talavera G, Castresana J (2007) Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst Biol 56:564–577. https://doi.org/10.1080/10635150701472164
Thomson JW (1984) American arctic lichens: the macrolichenes. Columbia University Press, New York
Vančurová L, Muggia L, Peksa O, Řídká T, Škaloud P (2018) The complexity of symbiotic interactions influences the ecological amplitude of the host: a case study in Stereocaulon (lichenized Ascomycota). Mol Ecol 27:3016–3033. https://doi.org/10.1111/mec.14764
Vančurová L, Kalníková V, Peksa O, Škvorová Z, Malíček J, Moya P, Chytrý K, Černajová I, Škaloud P (2020) Symbiosis between river and dry lands: phycobiont dynamics on river gravel bars. Algal Res 51:102062. https://doi.org/10.1016/j.algal.2020.102062
Vančurová L, Malíček J, Steinová J, Škaloud P (2021) Choosing the right life partner: ecological drivers of lichen symbiosis. Front Microbiol 12:769304. https://doi.org/10.3389/fmicb.2021.769304
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S Ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
White TJ, Bruns TD, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, London, pp 315–322
Wirtz N, Lumbsch HT, Allan Green TG, Türk R, Pintado A, Sancho L, Schroeter B (2003) Lichen fungi have low cyanobiont selectivity in maritime Antarctica. New Phytol 160:177–183. https://doi.org/10.1046/j.1469-8137.2003.00859
Zúñiga C, Leiva D, Carú M, Orlando J (2017) Substrates of Peltigera lichens as a potential source of cyanobionts. Microb Ecol 74:561–569. https://doi.org/10.1007/s00248-017-0969-z
Acknowledgements
The authors thank Dr. Laurens Sparrius for sending a specimen of S. alpinum from Finland and Dr. Andre Aptroot for arranging this collaboration. We also want to thank to the CGMS curator Luciana Canêz who quickly attended our requests. Finally thanks to Dra. Mayara Scur and to Drs. Marcos Kitaura and Wellington Fava for collecting fresh specimens.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. V. Torres received a Master fellowship (CAPES), and J.-M. Torres and A. Rodrigues received a Doctoral fellowship (CAPES). This was also supported by the Brazilian “Evolution and Dispersal of Antarctic Bipolar Species of Mosses and Lichens” project (MCTI/CNPq/FNDCT—Ação Transversal n° 64/2013).
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology and validation, APL and JMT; field work, AAS and APL; morphological analyses, JMT; chemical analyses, AG, ACM and NKH; molecular analyses, APL and JMT; writing of original draft preparation, JMT, APL, AR and VT; writing, review, and editing, JMT, APL and AAS; funding acquisition, AAS and APL All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest at any level for the publication of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
300_2023_3168_MOESM1_ESM.xlsx
Supplementary file1 (XLSX 13 KB)Online Resources 1 Information about the specimens of Stereocaulon alpinum analysed in this study
300_2023_3168_MOESM2_ESM.xlsx
Supplementary file2 (XLSX 1029 KB)Online Resource 2 Information about nuITS, β-tubulin, Actin and 16s sequences analysed in this study
300_2023_3168_MOESM3_ESM.tif
Supplementary file3 (TIF 4019 KB) Online Resource 3 Maximum clade credibility tree from Bayesian inference of nuITS sequences of Stereocaulon. Thickened branches correspond to high support values from bayesian inference (a priori probability ≥ 0.9) and from maximum likelihood (bootstrap ≥ 70) respectively. Red branches corresponding to the clades of Stereocaulon alpinum. The sequences here generated of S. alpinum are in the bipolar clade and are marked in bold
300_2023_3168_MOESM4_ESM.tif
Supplementary file4 (TIF 2214 KB)Online Resource 4 Maximum clade credibility tree from Bayesian inference of the concatenated date set of nuITS and β-tubulin sequences of Stereocaulon. Thickened branches correspond to high support values from bayesian inference (a priori probability ≥ 0.9) and from maximum likelihood (bootstrap ≥ 70) respectively. Red branches corresponding to the clades of Stereocaulon alpinum. The sequences here generated of S. alpinum are in the bipolar clade and are marked in bold
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Torres, J.M., Torres, V.O., Rodrigues, A.S. et al. Lineages of the lichen-forming fungus Stereocaulon alpinum and their photobionts in southern South America and maritime Antarctica. Polar Biol 46, 865–879 (2023). https://doi.org/10.1007/s00300-023-03168-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-023-03168-y