Abstract
Main conclusion
Ambrosia species differ both in their trichome types and in metabolic profiles of leaf volatiles. The current study provides tools for easier taxonomic identification of ragweed species.
Abstract
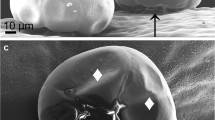
The genus Ambrosia (Asteraceae) includes some of the most noxious allergenic invasive weeds in the world. Due to high polymorphism in this genus, identification of species is often difficult. This study focuses on microscopic investigation of foliar features and GC-MS identification of the main leaf volatile components of three Ambrosia species currently found in Israel—invasive species Ambrosia confertiflora and A. tenuifolia, and transient A. grayi. A. confertiflora and A. tenuifolia have three trichome types: non-glandular trichomes, capitate glandular trichomes and linear glandular trichomes. Their non-glandular trichomes and capitate trichomes have distinct structures and can serve as taxonomic characters. A. grayi (the least successful invader) has only very dense covering trichomes. All three Ambrosia species have secretory structures in their leaf midrib. A. confertiflora, the most problematic invasive plant in Israel, had a ten times higher volatiles content than the other two species. In A. confertiflora, the most abundant volatiles were chrysanthenone (25.5%), borneol (18%), germacrene D and (E)-caryophyllene (both around 12%). In A. tenuifolia, the most abundant volatiles were β-myrcene (32.9%), (2E)-hexenal (13%) and 1,8-cineole (11.7%). In A. grayi, the most abundant volatiles were β-myrcene (17.9%), germacrene D (17.8%) and limonene (14%). The three examined species have distinct trichome types and metabolic profiles. Non-glandular trichomes show structural diversification between species and are a good descriptive character. Considering the anthropocentric significance of this highly problematic genus, the current study provides tools for easier identification of ragweed species.





Similar content being viewed by others
Data availability
The data will be made available on reasonable request.
Abbreviations
- GC–MS:
-
Gas chromatography–Mass spectrometry,
- HS-SPME:
-
Headspace-Solid-phase microextraction
- SEM:
-
Scanning Electron Microscopy
References
Adedeji O, Jewoola OA (2008) Importance of leaf epidermal characters in the Asteraceae family. Not Bot Hort Agrobot Cluj-Napoca. https://doi.org/10.15835/nbha362243
Adkins S, Shabbir A (2014) Biology, ecology and management of the invasive parthenium weed (Parthenium hysterophorus L.). Pest Manag Sci 70:1023–1029. https://doi.org/10.1002/ps.3708
Aschenbrenner AK, Horakh S, Spring O (2013) Linear glandular trichomes of Helianthus (Asteraceae): Morphology, localization, metabolite activity and occurrence. AoB Plants 5:1–9. https://doi.org/10.1093/aobpla/plt028
Bajwa AA, Sadia S, Ali HH et al (2016) Biology and management of two important Conyza weeds: a global review. Environ Sci Pollut Res 23:24694–24710. https://doi.org/10.1007/s11356-016-7794-7
Bajwa A, Matzrafi M, Jugulam M (2021) Editorial: biology and management of weeds and invasive plant species under changing climatic and management regimes. Front Agron. https://doi.org/10.3389/fagro.2021.728144
Bar M, Shtein I (2019) Plant trichomes and the biomechanics of defense in various systems, with Solanaceae as a model. Botany 97:651–660. https://doi.org/10.1139/cjb-2019-0144
Bruce TJA, Birkett MA, Blande J et al (2005) Response of economically important aphids to components of Hemizygia petiolata essential oil. Pest Manag Sci 61:1115–1121. https://doi.org/10.1002/ps.1102
Buters J, Alberternst B, Nawrath S et al (2015) Ambrosia artemisiifolia (ragweed) in Germany – current presence, allergological relevance and containment procedures. Allergo J Int 24:108–120. https://doi.org/10.1007/s40629-015-0060-6
Cadena-González AL, Sørensen M, Theilade I (2013) Use and valuation of native and introduced medicinal plant species in Campo Hermoso and Zetaquira, Boyacá, Colombia. J Ethnobiol Ethnomed 9:1–34. https://doi.org/10.1186/1746-4269-9-23
Cicció JF, Chaverri C (2015) Essential oil composition of Ambrosia cumanensis (Asteraceae) from Costa Rica. Am J Essent Oils Nat Prod 3:15–21
Dalrymple RR, Rogers JL (1983) Allelopathic effects of western ragweed on seed germination and seedling growth of selected plants. J Chem Ecol 9:1073–1078
Davidovich-Rikanati R, Lewinsohn E, Bar E et al (2008) Overexpression of the lemon basil-zingiberene synthase gene increases both mono- and sesquiterpene contents in tomato fruit. Plant J 56:228–238. https://doi.org/10.1111/j.1365-313X.2008.03599.x
Frank L, Wenig M, Ghirardo A et al (2021) Isoprene and β-caryophyllene confer plant resistance via different plant internal signalling pathways. Plant Cell Environ 44:1151–1164. https://doi.org/10.1111/pce.14010
Genton BJ, Kotanen PM, Cheptou PO et al (2005) Enemy release but no evolutionary loss of defence in a plant invasion: an inter-continental reciprocal transplant experiment. Oecologia 146:404–414. https://doi.org/10.1007/s00442-005-0234-x
Gerber E, Schaffner U, Gassmann A et al (2011) Prospects for biological control of Ambrosia artemisiifolia in Europe: Learning from the past. Weed Res 51:559–573. https://doi.org/10.1111/j.1365-3180.2011.00879.x
Hamaoui-Laguel L, Vautard R, Liu L et al (2015) Effects of climate change and seed dispersal on airborne ragweed pollen loads in Europe. Nat Clim Chang 5:766–771. https://doi.org/10.1038/nclimate2652
Hulme PE (2017) Climate change and biological invasions: evidence, expectations, and response options. Biol Rev 92:1297–1313. https://doi.org/10.1111/brv.12282
Jovanović M, Mimica-Dukić N, Poljački M, Boža P (2003) Erythema multiforme due to contact with weeds: a recurrence after patch testing. Contact Dermatitis 48:17–25. https://doi.org/10.1034/j.1600-0536.2003.480104.x
Kariyat RR, Hardison SB, De Moraes CM, Mescher MC (2017a) Plant spines deter herbivory by restricting caterpillar movement. Biol Lett 13:20170176. https://doi.org/10.1098/rsbl.2017.0176
Kariyat RR, Smith JD, Stephenson AG et al (2017b) Non-glandular trichomes of Solanum carolinense deter feeding by manduca sexta caterpillars and cause damage to the gut peritrophic matrix. Proc R Soc B Biol Sci. https://doi.org/10.1098/rspb.2016.2323
Karrer G, Sirbu C, Oprea A et al (2021) Ambrosia tenuifolia, instead of A. psilostachya in Romania. Sci Ann Danube Delta Inst. https://doi.org/10.7427/DDI.26.02
Kaur S, Aulakh J, Jhala AJ (2016) Growth and seed production of glyphosate-resistant giant ragweed (Ambrosia trifida L.) in response to water stress. Can J Plant Sci 96:828–836. https://doi.org/10.1139/cjps-2015-0309
Kindscher K, Hurlburt DP (1998) Huron Smith’s ethnobotany of the Hocak (Winnebago). Econ Bot 52:352–372. https://doi.org/10.1007/bf02862065
Kono A, Shimizu T (2020) Leaf trichomes as an effective structure for disease resistance: the case of grapevine downy mildew. Japan Agric Res Q 54:293–298. https://doi.org/10.6090/jarq.54.293
Kromer K, Kreitschitz A, Kleinteich T et al (2016) Oil secretory system in vegetative organs of three Arnica taxa: essential oil synthesis, distribution and accumulation. Plant Cell Physiol 57:1020–1037. https://doi.org/10.1093/pcp/pcw040
Kumamoto J, Scora RW, Payne WW (1975) Ambrosial, a dehydrocitral isolated from the leaf oil of Ambrosia confertiflora D.C. (Ragweed). J Agric Food Chem 23:123–124. https://doi.org/10.1021/jf60197a012
Kumar Rai P, Singh JS (2020) Invasive alien plant species: their impact on environment, ecosystem services and human health. Ecol Indic. https://doi.org/10.1016/j.ecolind.2019.106020
Kumari P, Mishra R, Arora N et al (2017) Antifungal and anti-biofilm activity of essential oil active components against Cryptococcus neoformans and Cryptococcus laurentii. Front Microbiol 8:1–14. https://doi.org/10.3389/fmicb.2017.02161
Langmaier M, Lapin K (2020) A systematic review of the impact of invasive alien plants on forest regeneration in European temperate forests. Front Plant Sci 11:1–15. https://doi.org/10.3389/fpls.2020.524969
Lersten NR, Curtis JD (1988) Secretory reservoirs (ducts) of two kinds in giant gagweed (Ambrosia trifida; Asteraceae). Am J Bot 75:1313–1323
Levin DADA (1973) The role of trichomes in plant defense. Q Rev Biol 48:3–15
Levine JM, Vilà M, D’Antonio CM et al (2003) Mechanisms underlying the impacts of exotic plant invasions. Proc R Soc B Biol Sci 270:775–781. https://doi.org/10.1098/rspb.2003.2327
Manetas Y (2003) The importance of being hairy: The adverse effects of hair removal on stem photosynthesis of Verbascum speciosum are due to solar UV-B radiation. New Phytol 158:503–508. https://doi.org/10.1046/j.1469-8137.2003.00768.x
Marei GIK, Abdel Rasoul MA, Abdelgaleil SAM (2012) Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pestic Biochem Physiol 103:56–61. https://doi.org/10.1016/j.pestbp.2012.03.004
Martin MD, Quiroz-Claros E, Brush GS, Zimmer EA (2018) Herbarium collection-based phylogenetics of the ragweeds (Ambrosia, Asteraceae). Mol Phylogenet Evol 120:335–341. https://doi.org/10.1016/j.ympev.2017.12.023
Matzrafi M, Preston C, Brunharo CA (2021) Review: evolutionary drivers of agricultural adaptation in Lolium spp. Pest Manag Sci 77:2209–2218. https://doi.org/10.1002/ps.6219
Metcalfe CRCR, Chalk L (1957) Anatomy of the dicotyledons. Oxford University Press, London
Molinaro F, Monterumici CM, Ferrero A et al (2016) Bioherbicidal activity of a germacranolide sesquiterpene dilactone from Ambrosia artemisiifolia L. J Environ Sci Heal - Part B 51:847–852. https://doi.org/10.1080/03601234.2016.1208466
Möller H, Spirén A, Svensson Å et al (2002) Contact allergy to the Asteraceae plant Ambrosia artemisiifolia L. (ragweed) in sesquiterpene lactone-sensitive patients in southern Sweden. Contact Dermatitis 47:157–160. https://doi.org/10.1034/j.1600-0536.2002.470306.x
Montagnani C, Gentili R, Smith M et al (2017) The worldwide spread, success, and impact of ragweed (Ambrosia spp.). CRC Crit Rev Plant Sci 36:139–178. https://doi.org/10.1080/07352689.2017.1360112
Onen H, Farooq S, Gunal H et al (2017) Higher tolerance to abiotic stresses and soil types may accelerate common ragweed (Ambrosia artemisiifolia) invasion. Weed Sci 65:115–127. https://doi.org/10.1614/ws-d-16-00011.1
Payne WW (1964) A re-evaluation of the genus Ambrosia (Compositae). J Arnold Arbor 45:401–438
Payne WW (1966) Notes on the ragweeds of south America with the description of two new species: Ambrosia pannosa and A. parvifolia (Compositae). Brittonia 18:28–37. https://doi.org/10.2307/2805107
Payne WW (1976) Biochemistry and species problems in Ambrosia (Asteraceae-Ambrosieae). Plant Syst Evol 125:169–178. https://doi.org/10.1007/BF00986149
Payne WW (1978) A glossary of plant hair terminology. Brittonia 30:239–255
Peters K, Breitsameter L, Gerowitt B (2014) Impact of climate change on weeds in agriculture: a review. Agron Sustain Dev 34:707–721. https://doi.org/10.1007/s13593-014-0245-2
Pillemer EA, Tingey WM (1976) Hooked trichomes : a physical plant barrier to a major agricultural pest. Science 80(193):482–484
Pimentel D, Zuniga R, Morrison D (2005) Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol Econ 52:273–288. https://doi.org/10.1016/j.ecolecon.2004.10.002
Riddick EW, Simmons AM (2014) Do plant trichomes cause more harm than good to predatory insects? Pest Manag Sci 70:1655–1665. https://doi.org/10.1002/ps.3772
Røstelien T, Borg-Karlson A-K, Fäldt J et al (2000) The plant sesquiterpene germacrene D specifically activates a major type of antennal receptor neuron of the tobacco budworm moth Heliothis virescens. Chem Senses 25:141–148. https://doi.org/10.1093/chemse/25.2.141
Ruzin S (1999) Plant microtechnique and microscopy. Oxford University Press, New York
Sarić-Krsmanović M, Umiljendić JG, Radivojević L et al (2020) Chemical composition of Ambrosia trifida essential oil and phytotoxic effect on other plants. Chem Biodivers. https://doi.org/10.1002/cbdv.201900508
Shemluck M (1982) Medicinal and other uses of the Compositae by Indians in the United States and Canada. J Ethnopharmacol 5:303–358. https://doi.org/10.1016/0378-8741(82)90016-2
Shimono Y, Takiguchi Y, Konuma A (2010) Contamination of internationally traded wheat by herbicide-resistant Lolium rigidum. Weed Biol Manag 10:219–228. https://doi.org/10.1111/j.1445-6664.2010.00387.x
Spring O, Rodon U, Macias FA (1992) Sesquiterpenes from noncapitate glandular trichomes of Helianthus annuus. Phytochemistry 31:1541–1544
Steckel LE (2007) The dioecious Amaranthus spp.: Here to stay. Weed Technol 21:567–570. https://doi.org/10.1614/WT-06-045.1
Taglialatela-Scafati O, Pollastro F, Minassi A et al (2012) Sesquiterpenoids from common ragweed (Ambrosia artemisiifolia L.), an invasive biological polluter. European J Org Chem 27:5162–5170. https://doi.org/10.1002/ejoc.201200650
Tahvanainen JO, Root RB (1972) Influence of vegetational diversity on the population ecology of a specialized herbivore, Phyllotreta cruciferae (Coleoptera: Chrysomelidae). Oecologia 10:321–346
Talbot MJ, White RG (2013) Methanol fixation of plant tissue for scanning electron microscopy improves preservation of tissue morphology and dimensions. Plant Methods 9:36. https://doi.org/10.1186/1746-4811-9-36
Vidotto F, Tesio F, Ferrero A (2013) Allelopathic effects of Ambrosia artemisiifolia L. in the invasive process. Crop Prot 54:161–167. https://doi.org/10.1016/j.cropro.2013.08.009
Wagner WH, Beals TF (1958) Perennial ragweeds (Ambrosia) in Michigan, with the description of a new, intermediate taxon. Rhodora - J New Engl Bot Club 60:177–204
Waisel Y, Eshel A, Keynan N, Langgut D (2008) Ambrosia : a new impending disaster for the israeli allergic population. Isr Med Assoc J 10:856–857
Wang P, Chui HK, Chao XZ (2006) Chemical composition and antimicrobial activity of the essential oil from Ambrosia trifida L. Molecules 11:549–555. https://doi.org/10.3390/11070549
Watt GC (1987) Ragweed dermatitis and the distribution of ragweed in Australia. Australas J Dermatol 28:27–29. https://doi.org/10.1111/j.1440-0960.1987.tb00325.x
Wopfner N, Gadermaier G, Egger M et al (2005) The spectrum of allergens in ragweed and mugwort pollen. Int Arch Allergy Immunol 138:337–346. https://doi.org/10.1159/000089188
Xu L, Shi Z, Wang B et al (2016) Pine defensive monoterpene α-pinene influences the feeding behavior of dendroctonus valens and its gut bacterial community structure. Int J Mol Sci 17:173. https://doi.org/10.3390/ijms17111734
Yair Y, Sibony M, Rubin B (2017) Four Ambrosia species in Israel: invasive, naturalized and casual alien plants. Isr J Plant Sci 9978:1–6. https://doi.org/10.1080/07929978.2017.1288399
Yamasaki T, Sato M, Sakoguchi H (1997) (–)-Germacrene D: Masking substance of attractants for the cerambycid beetle, Monochamus elternatus (Hope. Appl Enthomology Zool 32:423–429
Acknowledgements
The study was supported by a grant N 3-15745 from the Israel Ministry of Science. We wish to thank Danielle Neta for growing plant material of A. grayi and Jotham Ziffer-Berger for his insights into plant taxonomy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
425_2023_4113_MOESM2_ESM.tif
Supplementary file2 S2 Larger magnification of secretory cavities in A. confertiflora (a), A. tenuifolia (b) and A. grayi (c) leaf veins (black arrows). Light microscopy, Safranin-Alcian Blue stain (TIF 9490 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matzrafi, M., Wolberg, S., Abu-Nassar, J. et al. Distinctive foliar features and volatile profiles in three Ambrosia species (Asteraceae). Planta 257, 79 (2023). https://doi.org/10.1007/s00425-023-04113-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04113-3