Abstract
Introduction
Etelcalcetide binds to the extracellular domain of the calcium-sensing receptor (CaSR), while cinacalcet binds to the 7-transmembrane domain of the CaSR; however, it is unknown, whether etelcalcetide has similar effects to cinacalcet on parathyroid hormone (PTH) secretion.
Materials and methods
The PTH–calcium setpoint and maximum and minimum PTH secretion were determined using an ‘in vivo setpoint analyses.’ The PTH–calcium setpoint was obtained in a mouse model of primary hyperparathyroidism (PC) and wild-type (WT) mice, with PC mice divided into two groups. The setpoint was obtained after 7 days of etelcalcetide (3.0 mg/kg BW/day) or vehicle administration via anosmotic pump. After 7 days of crossover administration, the setpoint was obtained again. Parathyroid glands were obtained after crossover administration, and CaSR expression was analyzed by immunohistochemistry.
Results
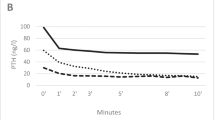
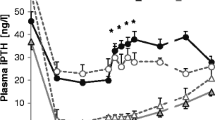
Etelcalcetide administration significantly decreased the setpoint from 9.03 ± 0.56 mg/dL to 6.80 ± 0.28 mg/dL, which was restored to 8.81 ± 0.38 mg/dL after vehicle administration. In the second group of mice, vehicle administration did not alter the setpoint (8.84 ± 0.69 mg/dL to 8.98 ± 0.63 mg/dL), but subsequent etelcalcetide administration significantly decreased it to 7.10 ± 0.72 mg/dL. There was no significant change in maximum and minimum PTH secretion. Expression levels of parathyroid CaSR were lower in PC mice than in WT mice; however, no significant differences were observed between the two mouse groups.
Conclusion
Etelcalcetide decreased the PTH–calcium setpoint without changing maximum and minimum PTH secretion in PC mice, suggesting that like cinacalcet, etelcalcetide has calcimimetic potency.




Similar content being viewed by others
References
Moe S, Drueke T, Cunningham J, Goodman W, Martin K, Olgaard K, Ott S, Sprague S, Lameire N, Eknoyan G (2006) Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 69:1945–1953
Covic A, Vervloet M, Massy ZA, Torres PU, Goldsmith D, Brandenburg V, Mazzaferro S, Evenepoel P, Bover J, Apetrii M, Cozzolino M (2018) Bone and mineral disorders in chronic kidney disease: implications for cardiovascular health and ageing in the general population. Lancet Diabetes Endocrinol 6:319–331
Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, Wang Y, Chung J, Emerick A, Greaser L, Elashoff RM, Salusky IB (2000) Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med 342:1478–1483
Ganesh SK, Stack AG, Levin NW, Hulbert-Shearon T, Port FK (2001) Association of elevated serum PO4, Ca x PO4 product, and parathyroid hormone with cardiac mortality risk in chronic hemodialysis patients. J Am Soc Nephrol 12:2131–2138
Imanishi Y, Nishizawa Y, Inaba M (2013) Chapter 5 Pathogenesis and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder. In: Suzuki H, (ed.) Hemodialysis. InTech: Rijeka, Croatia; 81–100
Kawata T, Imanishi Y, Kobayashi K, Onoda N, Okuno S, Takemoto Y, Komo T, Tahara H, Wada M, Nagano N, Ishimura E, Miki T, Ishikawa T, Inaba M, Nishizawa Y (2006) Direct in vitro evidence of the suppressive effect of cinacalcet HCl on parathyroid hormone secretion in human parathyroid cells with pathologically reduced calcium-sensing receptor levels. J Bone Miner Metab 24:300–306
Kidney Disease: Improving Global Outcomes (KDIGO), CKD-MBD Work Group (2017) KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl 7:1–59
Fukagawa M, Yokoyama K, Koiwa F, Taniguchi M, Shoji T et al (2013) Clinical practice guideline for the management of chronic kidney disease-mineral and bone disorder. Ther Apher Dial 17:247–288
Nemeth EF, Steffey ME, Hammerland LG, Hung BC, Van Wagenen BC, DelMar EG, Balandrin MF (1998) Calcimimetics with potent and selective activity on the parathyroid calcium receptor. Proc Natl Acad Sci U S A 95:4040–4045
Nemeth EF (2002) The search for calcium receptor antagonists (calcilytics). J Mol Endocrinol 29:15–21
Nemeth EF, Heaton WH, Miller M, Fox J, Balandrin MF, Van Wagenen BC, Colloton M, Karbon W, Scherrer J, Shatzen E, Rishton G, Scully S, Qi M, Harris R, Lacey D, Martin D (2004) Pharmacodynamics of the type II calcimimetic compound cinacalcet HCl. J Pharmacol Exp Ther 308:627–635
Alexander ST, Hunter T, Walter S, Dong J, Maclean D, Baruch A, Subramanian R, Tomlinson JE (2015) Critical cysteine residues in both the calcium-sensing receptor and the allosteric activator AMG 416 underlie the mechanism of action. Mol Pharmacol 88:853–865
Walter S, Baruch A, Dong J, Tomlinson JE, Alexander ST, Janes J, Hunter T, Yin Q, Maclean D, Bell G, Mendel DB, Johnson RM, Karim F (2013) Pharmacology of AMG 416 (Velcalcetide), a novel peptide agonist of the calcium-sensing receptor, for the treatment of secondary hyperparathyroidism in hemodialysis patients. J Pharmacol Exp Ther 346:229–240
Imanishi Y, Hosokawa Y, Yoshimoto K, Schipani E, Mallya S, Papanikolaou A, Kifor O, Tokura T, Sablosky M, Ledgard F, Gronowicz G, Wang TC, Schmidt EV, Hall C, Brown EM, Bronson R, Arnold A (2001) Primary hyperparathyroidism caused by parathyroid-targeted overexpression of cyclin D1 in transgenic mice. J Clin Invest 107:1093–1102
Imanishi Y, Hall C, Sablosky M, Brown EM, Arnold A (2002) A new method for in vivo analysis of parathyroid hormone-calcium set point in mice. J Bone Miner Res 17:1656–1661
Brown EM (1983) Four-parameter model of the sigmoidal relationship between parathyroid hormone release and extracellular calcium concentration in normal and abnormal parathyroid tissue. J Clin Endocrinol Metab 56:572–581
Imanishi Y, Inaba M, Kawata T, Nishizawa Y (2009) Animal models of hyperfunctioning parathyroid diseases for drug development. Expert Opin Drug Discov 4:727–740
Kawata T, Imanishi Y, Kobayashi K, Kenko T, Wada M, Ishimura E, Miki T, Nagano N, Inaba M, Arnold A, Nishizawa Y (2005) Relationship between parathyroid calcium-sensing receptor expression and potency of the calcimimetic, cinacalcet, in suppressing parathyroid hormone secretion in an in vivo murine model of primary hyperparathyroidism. Eur J Endocrinol 153:587–594
Imanishi Y, Kawata T, Kenko T, Wada M, Nagano N, Miki T, Arnold A, Inaba M (2011) Cinacalcet HCl suppresses Cyclin D1 oncogene-derived parathyroid cell proliferation in a murine model for primary hyperparathyroidism. Calcif Tissue Int 89:29–35
Harada K, Fujioka A, Konno M, Inoue A, Yamada H, Hirota Y (2019) Pharmacology of Parsabiv((R)) (etelcalcetide, ONO-5163/AMG 416), a novel allosteric modulator of the calcium-sensing receptor, for secondary hyperparathyroidism in hemodialysis patients. Eur J Pharmacol 842:139–145
Brown EM, Gardner DG, Brennan MF, Marx SJ, Spiegel AM, Attie MF, Downs RW Jr, Doppman JL, Aurbach CD (1979) Calcium-regulated parathyroid hormone release in primary hyperparathyroidism: studies in vitro with dispersed parathyroid cells. Am J Med 66:923–931
Brown EM, Wilson RE, Eastman RC, Pallotta J, Marynick SP (1982) Abnormal regulation of parathyroid hormone release by calcium in secondary hyperparathyroidism due to chronic renal failure. J Clin Endocrinol Metab 54:172–179
Mun HC, Franks AH, Culverston EL, Krapcho K, Nemeth EF, Conigrave AD (2004) The Venus Fly Trap domain of the extracellular Ca2+ -sensing receptor is required for l-amino acid sensing. J Biol Chem 279:51739–51744
Broadhead GK, Mun HC, Avlani VA, Jourdon O, Church WB, Christopoulos A, Delbridge L, Conigrave AD (2011) Allosteric modulation of the calcium-sensing receptor by gamma-glutamyl peptides: inhibition of PTH secretion, suppression of intracellular cAMP levels, and a common mechanism of action with l-amino acids. J Biol Chem 286:8786–8797
Petrel C, Kessler A, Dauban P, Dodd RH, Rognan D, Ruat M (2004) Positive and negative allosteric modulators of the Ca2+-sensing receptor interact within overlapping but not identical binding sites in the transmembrane domain. J Biol Chem 279:18990–18997
Yu L, Tomlinson JE, Alexander ST, Hensley K, Han CY, Dwyer D, Stolina M, Dean C Jr, Goodman WG, Richards WG, Li X (2017) Etelcalcetide, a novel calcimimetic, prevents vascular calcification in a rat model of renal insufficiency with secondary hyperparathyroidism. Calcif Tissue Int 101:641–653
Colloton M, Shatzen E, Miller G, Stehman-Breen C, Wada M, Lacey D, Martin D (2005) Cinacalcet HCl attenuates parathyroid hyperplasia in a rat model of secondary hyperparathyroidism. Kidney Int 67:467–476
Imanishi Y, Inaba M, Seki H, Koyama H, Nishizawa Y, Morii H, Otani S (1999) Increased biological potency of hexafluorinated analogs of 1,25-dihydroxyvitamin D3 on bovine parathyroid cells. J Steroid Biochem Mol Biol 70:243–248
Acknowledgements
We gratefully thank Prof. Andrew Arnold (University of Connecticut School of Medicine) for providing us with PC mice and for engaging in in-depth discussions. We thank Ms. Emi Donoue and Mr. Keisuke Inoue (Research Support Platform of Osaka City University Graduate School of Medicine) for technical support.
Funding
This work was supported in part by a research Grant from ONO Pharmaceutical, Co., Ltd., Osaka, Japan.
Author information
Authors and Affiliations
Contributions
YI designed the study and wrote the main manuscript. NH and TH contributed to the setpoint analysis, and TH and IK contributed to the immunohistochemistry experiments. TH, TT, DM, YN, and TM contributed to the acquisition, analysis, and interpretation of the data. KM, AI, KH, ME, and MI reviewed the manuscript critically for intellectual content. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
YI, KM, ME, and MI received lecture fees from ONO Pharmaceutical Co., Ltd., Osaka, Japan. AI and KH are employees of ONO Pharmaceutical Co., Ltd., Osaka, Japan. NH, TH, IK, TT, DM, YN, and TM declare no conflicts of interest.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Hayashi, N., Imanishi, Y., Hirakawa, T. et al. Etelcalcetide decreases the PTH–calcium setpoint without changing maximum and minimum PTH secretion in mice with primary hyperparathyroidism. J Bone Miner Metab 39, 430–438 (2021). https://doi.org/10.1007/s00774-020-01169-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-020-01169-z