Abstract
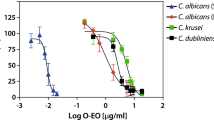
Candida spp. is a significant cause of topical and fungal infections in humans. In addition to Candida albicans, many non-albicans species such as C. krusei, C. glabrata, C. parapsilosis, C. tropicalis, C. guilliermondii cause severe infections. The main antifungal agents belong to three different classes, including azoles, polyenes, and echinocandins. However, resistance to all three categories of drugs has been reported. Therefore, there is an urgent need to search for other alternatives with antifungal activity. Many herbal extracts and compounds from natural sources show excellent antifungal activity. In this study, we used an oil extract from the fruits of Zanthoxylum armatum, which showed significant antifungal activity against various Candida spp. by two different methods—minimum inhibitory concentration (MIC) and agar diffusion. In addition, we attempted to explore the possible mechanism of action in C. albicans. It was found that the antifungal activity of Z. armatum oil is fungicidal and involves a decrease in the level of ergosterol in the cell membrane. The decrease in ergosterol level resulted in increased passive diffusion of a fluorescent molecule, rhodamine6G, across the plasma membrane, indicating increased membrane fluidity. The oil-treated cells showed decreased germ tube formation, an important indicator of C. albicans’ virulence. The fungal cells also exhibited decreased attachment to the buccal epithelium, the first step toward invasion, biofilm formation, and damage to oral epithelial cells. Interestingly, unlike most antifungal agents, in which the generation of reactive oxygen species is responsible for killing, no significant effect was observed in the present study.








Similar content being viewed by others

Data Availability
No separate data is available with authors and is already mentioned after the funding source.
References
Altamirano S, Jackson KM, Nielsen K (2020) The interplay of phenotype and genotype in Cryptococcus neoformans disease. Biosci Rep 40:BSR20190337. https://doi.org/10.1042/BSR20190337
Balouiri M, Sadiki M, Ibnsouda SK (2016) Methods for in-vitro evaluating antimicrobial activity: a review. J Pharm Anal 6:71–79. https://doi.org/10.1016/j.jpha.2015.11.005
Brayman TG, Wilks JW (2003) Sensitive assay for antifungal activity of glucan synthase inhibitors that uses germ tube formation in Candida albicans as an end poinT. Antimicrob Agents Chemother 47:3305–3310. https://doi.org/10.1128/AAC.47.10.3305-3310.2003
Burt S (2004) Essential oils: their antibacterial properties and potential applications in foods—a review. Int J Food Microbiol 94:223–253. https://doi.org/10.1016/j.ijfoodmicro.2004.03.022
Calderone RA, Fonzi WA (2001) Virulence factors of Candida albicans. Trends Microbiol 9:327–335. https://doi.org/10.1016/s0966-842x(01)02094-7
Chebbac K, Moussaoui AE, Bourhia M, Salamatullah AM, Alzahrani A, Guemmouh R (2021) Chemical analysis and antioxidant and antimicrobial activity of essential oils from Artemisia negrei L. against drug-resistant microbes. Evid-Based Complement Altern Med ECAM 2021:5902851. https://doi.org/10.1155/2021/5902851
Dias I, Trajano E, Castro R, Ferreira G, Medeiros H, Gomes D (2017) Antifungal activity of linalool in cases of Candida spp. isolated from individuals with oral candidiasis. Braz J Biol 78. https://doi.org/10.1590/1519-6984.171054
Enjalbert B, Whiteway M (2005) Release from quorum-sensing molecules triggers hyphal formation during Candida albicans resumption of growth. Eukaryot Cell 4:1203–1210. https://doi.org/10.1128/EC.4.7.1203-1210.2005
Franz R, Ruhnke M, Morschhäuser J (1999) Molecular aspects of fluconazole resistance development in Candida albicans. Mycoses 42:453–458. https://doi.org/10.1046/j.1439-0507.1999.00498.x
Gautam M, Rolta R, Kumar V, Sourirajan A, Sharma DR, Dev K (2020) Comparative antimicrobial and synergistic potential of extracts of leaves of Zanthoxylum armatum Dc. from different geographical regions of Himachal Pradesh. Plant Arch 20:8127–8135
Hammer KA, Carson CF, Riley TV (2000) Melaleuca alternifolia (tea tree) oil inhibits germ tube formation by Candida albicans. Med Mycol 38:354–362. https://doi.org/10.1080/mmy.38.5.354.361
Hsu C-C, Lai W-L, Chuang K-C, Lee M-H, Tsai Y-C (2013) The inhibitory activity of linalool against the filamentous growth and biofilm formation in Candida albicans. Med Mycol 51:473–482. https://doi.org/10.3109/13693786.2012.743051
Kainz K, Bauer MA, Madeo F, Carmona-Gutierrez D (2020) Fungal infections in humans: the silent crisis. Microb Cell Graz Austria 7:143–145. https://doi.org/10.15698/mic2020.06.718
Khan A, Ahmad A, Akhtar F, Yousuf S, Xess I, Khan LA et al (2010) Ocimum sanctum essential oil and its active principles exert their antifungal activity by disrupting ergosterol biosynthesis and membrane integrity. Res Microbiol 161:816–823. https://doi.org/10.1016/j.resmic.2010.09.008
Kontoyiannis DP (2017) Antifungal resistance: an emerging reality and a global challenge. J Infect Dis 216:S431–S435. https://doi.org/10.1093/infdis/jix179
Kundu D, Hameed S, Fatima Z, Pasrija R (2020) Phospholipid biosynthesis disruption renders the yeast cells sensitive to antifungals. Folia Microbiol (Praha) 65:121–131. https://doi.org/10.1007/s12223-019-00713-3
Li Y, Gao Y, Niu X, Wu Y, Du Y, Yang Y et al (2020) A 5-year review of invasive fungal infection at an academic medical center. Front Cell Infect Microbiol 10. Available at: https://www.frontiersin.org/articles/10.3389/fcimb.2020.553648. Accessed February 23, 2023
Lima TC, Ferreira AR, Silva DF, Lima EO, de Sousa DP (2018) Antifungal activity of cinnamic acid and benzoic acid esters against Candida albicans strains. Nat Prod Res 32:572–575. https://doi.org/10.1080/14786419.2017.1317776
Lo HJ, Köhler JR, DiDomenico B, Loebenberg D, Cacciapuoti A, Fink GR (1997) Nonfilamentous Candida albicans mutants are avirulent. Cell 90:939–949. https://doi.org/10.1016/s0092-8674(00)80358-x
Lopes G, Pinto E, Andrade PB, Valentão P (2013) Antifungal activity of phlorotannins against dermatophytes and yeasts: approaches to the mechanism of action and influence on Candida albicans virulence factor. PLoS ONE 8:e72203. https://doi.org/10.1371/journal.pone.0072203
Manoharan RK, Lee J-H, Kim Y-G, Kim S-I, Lee J (2017) Inhibitory effects of the essential oils α-longipinene and linalool on biofilm formation and hyphal growth of Candida albicans. Biofouling 33:143–155. https://doi.org/10.1080/08927014.2017.1280731
Mishra NN, Prasad T, Sharma N, Lattif AA, Prasad R, Gupta DK (2007) Membrane fluidity and lipid composition in clinical isolates of Candida albicans isolated from AIDS/HIV patients. Acta Microbiol Immunol Hung 54:367–377. https://doi.org/10.1556/AMicr.54.2007.4.4
Mukhopadhyay K, Prasad T, Saini P, Pucadyil TJ, Chattopadhyay A, Prasad R (2004) Membrane sphingolipid-ergosterol interactions are important determinants of multidrug resistance in Candida albicans. Antimicrob Agents Chemother 48:1778–1787. https://doi.org/10.1128/AAC.48.5.1778-1787.2004
Nazzaro F, Fratianni F, Coppola R, Feo VD (2017) Essential oils and antifungal activity. Pharmaceuticals 10:86. https://doi.org/10.3390/ph10040086
Paiva CN, Bozza MT (2014) Are reactive oxygen species always detrimental to pathogens? Antioxid Redox Signal 20:1000–1037. https://doi.org/10.1089/ars.2013.5447
Pappas PG, Lionakis MS, Arendrup MC, Ostrosky-Zeichner L, Kullberg BJ (2018) Invasive candidiasis. Nat Rev Dis Primer 4:1–20. https://doi.org/10.1038/nrdp.2018.26
Pasrija R, Naime M (2022) Resolving the equation between mucormycosis and COVID-19 disease. Mol Biol Rep 1–8. https://doi.org/10.1007/s11033-021-07085-3
Pasrija R, Krishnamurthy S, Prasad T, Ernst JF, Prasad R (2005) Squalene epoxidase encoded by ERG1 affects morphogenesis and drug susceptibilities of Candida albicans. J Antimicrob Chemother 55:905–913. https://doi.org/10.1093/jac/dki112
Prajapati N, Ojha P, Karki TB (2015) Antifungal property of essential oil extracted from Zanthoxylum armatum (Timur). J Nutr Health Food Eng 3:266–270. https://doi.org/10.15406/jnhfe.2015.03.00096
Prasad T, Saini P, Gaur NA, Vishwakarma RA, Khan LA, Haq QMR et al (2005) Functional analysis of CaIPT1, a sphingolipid biosynthetic gene involved in multidrug resistance and morphogenesis of Candida albicans. Antimicrob Agents Chemother 49:3442–3452. https://doi.org/10.1128/AAC.49.8.3442-3452.2005
Revie NM, Iyer KR, Robbins N, Cowen LE (2018) Antifungal drug resistance: evolution, mechanisms and impact. Curr Opin Microbiol 45:70–76. https://doi.org/10.1016/j.mib.2018.02.005
Robbins N, Caplan T, Cowen LE (2017) Molecular evolution of antifungal drug resistance. Annu Rev Microbiol 71:753–775. https://doi.org/10.1146/annurev-micro-030117-020345
Selmecki A, Forche A, Berman J (2006) Aneuploidy and isochromosome formation in drug-resistant Candida albicans. Science 313:367–370. https://doi.org/10.1126/science.1128242
Singh S, Fatima Z, Ahmad K, Hameed S (2018) Fungicidal action of geraniol against Candida albicans is potentiated by abrogated CaCdr1p drug efflux and fluconazole synergism. PLOS ONE 13:e0203079. https://doi.org/10.1371/journal.pone.0203079
Spivak ES, Hanson KE (2018) Candida auris: an emerging fungal pathogen. J Clin Microbiol 56:e01588-e1617. https://doi.org/10.1128/JCM.01588-17
Tiwary M, Naik SN, Tewary DK, Mittal PK, Yadav S (2007) Chemical composition and larvicidal activities of the essential oil of Zanthoxylum armatum DC (Rutaceae) against three mosquito vectors. J Vector Borne Dis 44:198–204
Turner SA, Butler G (2014) The Candida pathogenic species complex. Cold Spring Harb Perspect Med 4:a019778. https://doi.org/10.1101/cshperspect.a019778
Verma KK, Kumar B, Raj H, Sharma A (2021) A review on chemical constituents, traditional uses, pharmacological studies of Zanthoxylum armatum(Rutaceae). J Drug Deliv Ther 11:136–142. https://doi.org/10.22270/jddt.v11i2-S.4786
Yu D, Wang J, Shao X, Xu F, Wang H (2015) Antifungal modes of action of tea tree oil and its two characteristic components against Botrytis cinerea. J Appl Microbiol 119:1253–1262. https://doi.org/10.1111/jam.12939
Acknowledgements
We are thankful to Prof Joachim Morschhäuser (Würzburg, Germany) for providing the C. albicans fluconazole-resistant cultures. We also acknowledge Mamta Saini for helping in completion of this study.
Funding
R. Pasrija received funding from Science & Engineering Research Board (SERB) (CRG/2020/004986) under the Department of Science and Technology and Maharshi Dayanand University, Radhakrishnan (R.K.Fund) minor research project (Sanctioned amount: Rs. 50,000).
Author information
Authors and Affiliations
Contributions
R.Pasrija conceived and designed the experiments. D. Kumari and S. Wadhwan conducted the experiments and analyzed the data. R.Pasrija, and V. Mittal wrote the manuscript. R.Pasrija, A. K. Chhillar, and V. Mittal analyzed the data.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Consent to publish has been received from all authors.
Competing interests
The authors declare no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pasrija, R., Kumari, D., Wadhwan, S. et al. The Zanthoxylum armatum fruit’s oil exterminates Candida cells by inhibiting ergosterol biosynthesis without generating reactive oxygen species. Int Microbiol 27, 423–434 (2024). https://doi.org/10.1007/s10123-023-00401-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-023-00401-w