Abstract
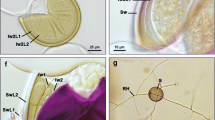
The mycorrhizal fungi in the roots of achlorophyllous Sciaphila japonica and S. tosaensis (Triuridaceae) were identified by molecular methods. The habitats of S. japonica were in a tree plantation of Japanese cypress, Chamaecyparis obtusa, and bamboo forests, and those of S. tosaensis were in a camellia forest and a bamboo forest. In the root cortical cells of both plants, aseptate hyphal coils were observed, which suggested the Paris-type arbuscular mycorrhiza (AM). A phylogenetic analysis based on a partial sequence of an AM fungal nuclear small subunit ribosomal RNA gene showed that the fungal DNA sequences of S. japonica were separated into three closely related clades. Those of S. tosaensis were separated into two clades, which were also closely related to each other. The AM fungi of S. japonica and S. tosaensis were completely separated in the phylogenetic tree even among those found in the same habitat, which suggests the high specificities in the plant-fungal partnerships. All the detected AM fungi in these plants belonged to Glomus-group A. Even though the habitats are in quite common environments, both plant species are known as endangered species in Japan. Such a definite specificity in AM symbioses seems to restrict the distribution of the myco-heterotrophic plants.



Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Angiosperm Phylogeny Group (2003) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants. Bot J Linn Soc 141:399–436
Bidartondo MI, Redecker D, Hijri I, Wiemken A, Bruns TD, Domínguez L, Sérsic A, Leake JR, Read DJ (2002) Epiparasitic plants specialized on arbuscular mycorrhizal fungi. Nature 419:389–392
Brundrett M, Bougher N, Dell B, Grove T, Malajczuk N (1996) Working with mycorrhizas in forestry and agriculture. Australian Center for international Agriculture Research, Canberra
Chase MW, Soltis DE, Soltis PS, Rudall PJ, Fay MF, Hahn WJ, Sullivan S, Joseph J, Givnish T, Sytsma KJ, Price C (2000) A combined analysis of multiple datasets and a new phylogenetic classification of the monocotyledons. In: Wilson KL, Morrison DA (eds) Monocots: systematic and evolution. Proceedings of the second international conference on the comparative biology of the monocotyledons. CSIRO Publishing, Melbourne, pp 3–16
Cronquist A (1988) The evolution and classification of flowering plants, 2nd edn. New York Botanical Garden, New York
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Franke T, Beenken L, Döring M, Kocyan A, Agerer R (2006) Arbuscular mycorrhizal fungi of the Glomus-group A lineage (Glomerales; Glomeromycota) detected in myco-heterotrophic plants from tropical Africa. Mycol Progress 5:24–31
Imhof S (1998) Subterranean structures and mycotrophy of the achlorophyllous Triuris hyaline (Triuridaceae). Can J Bot 76:2011–2019
Imhof S (1999) Root morphology, anatomy and mycotrophy of the achlorophyllous Voyria aphylla (Jacq.) Pers. (Gentianaceae). Mycorrhiza 9:33–39
Imhof S (2003) A dorsivental mycorrhizal root in the achlorophyllous Sciaphila polygyna (Triuridaceae). Mycorrhiza 13:327–332
Imhof S (2007) Specialized mycorrhizal colonization pattern in achlorophyllous Epirixanthes spp. (Polygalaceae). Plant Biol 9:786–792
Kitamura S, Murata G, Koyama T (2008) Colored illustrations of herbaceous plants of Japan vol. III (Monocotyledoneae), 55th edn. Hoikusha, Osaka
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Leake JR (1994) The biology of myco-heterotrophic (‘saprophytic’) plants. New Phytol 127:171–216
Leake JR, Cameron DD (2010) Physiological ecology of mycoheterotrophy. New Phytol 185:601–605
Maas-van de Kamer H (1995) Triuridiflorae: Gardner’s delight? In: Rudall PJ, Cribb PJ, Cutler DF, Humphries CJ (eds) Monocotyledons: systematic and evolution. Royal Botanic Gardens, Kew, pp 287–301
Mejstrik V (1974) The frequency of vesicular-arbuscular mycorrhizae in the roots of Camellia japonica L. from different sites in New Zealand. Pac Sci 28:73–77
Merckx V, Bidartondo MI (2008) Breakdown and delayed cospection in the arbuscular mycorrhizal mutualism. Proc R Soc B 275:1029–1035
Muthukumar T, Udaiyan K (2006) Growth of nursery-grown bamboo inoculated with arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria in two tropical soil types with and without fertilizer application. New For 31:469–485
Page RDM (1996) An application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Russell J, Bulman S (2005) The liverwort Marchantia foliacea forms a specialized symbiosis with arbuscular mycorrhizal fungi in the genus Glomus. New Phytol 165:567–579
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:404–425
Schüßler A, Schwarzott D, Walker C (2001) A new fungal phylum, the Glomaromycota: phylogeny and evolution. Mycol Res 105:1413–1421
Schwarzott D, Schüßler A (2001) A simple and reliable method for SSU rRNA gene DNA extraction, amplification, and cloning from single AM fungal spores. Mycorrhiza 10:203–207
Simon L, Lalonde M, Bruns TD (1992) Specific amplification of 18S fungal ribosomal genes from vesicular-arbuscular endomycorrhizal fungi colonizing roots. Appl Environ Microbiol 58:291–295
Simon L, Lévesque RC, Lalonde M (1993) Identification of endomycorrhizal fungi colonizing roots by fluorescent single-strand conformation polymorphism-polymerase chain reaction. Appl Environ Microbiol 59:4211–4215
Smith SE, Read DJ (1997) Mycorrhizal symbiosis, 2nd edn. Academic Press, San Diego
Takhtajan A (1997) Diversity and classification of flowering plants. Columbia Univ Press, New York
Thorne RF (1992) Classification and geography of the flowering plants. Bot Rev 58:225–348
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Winther JL, Friedman WE (2007) Arbuscular mycorrhizal symbionts in Botrychium (Ophioglossaceae). Am J Bot 94:1248–1255
Winther JL, Friedman WE (2008) Arbuscular mycorrhizal associations in Lycopodiaceae. New Phytol 177:790–801
Winther JL, Friedman WE (2009) Phylogenetic affinity of arbuscular mycorrhizal symbionts in Psilotum nudum. J Plant Res 122:485–496
Yamato M (2001) Identification of a mycorrhizal fungus in the roots of achlorophyllous Sciaphila tosaensis Makino (Triuridaceae). Mycorrhiza 11:83–88
Yamato M (2002) Morphological types of arbuscular mycorrhizal fungi in roots of understory plants in Japanese deciduous broadleaved forests. Mycorrhiza 12:291–296
Acknowledgments
We thank Dr. Tsutomu Teramine in Kochi Gakuen College, Mr. Hiroshi Fukuhara in The Kochi Prefectural Makino Botanical Garden, and Yasuo Katayama, Takeki Tabuchi and Hisanori Takeuchi, plant fanciers in Tokushima and Kochi Pref., for their kind help in the plant sampling. This study was supported by the Global COE Program “Advanced utilization of fungus/mushroom resources for sustainable society in harmony with nature” from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Yamato, M., Yagame, T. & Iwase, K. Arbuscular mycorrhizal fungi in roots of non-photosynthetic plants, Sciaphila japonica and Sciaphila tosaensis (Triuridaceae). Mycoscience 52, 217–223 (2011). https://doi.org/10.1007/s10267-010-0084-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10267-010-0084-1