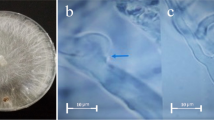
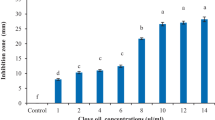
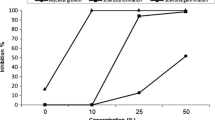
Abstract
In this study, propolis extract (EPE) and oregano essential oil (OEO), both natural products with different bioactive compounds, were evaluated in vitro and in vivo against Sclerotinia sclerotiorum. This phytopathogen is important worldwide as it can contaminate seeds and infect seedlings of many species. Antifungal susceptibility assays on culture media revealed that EPE and OEO were highly active against this phytopathogen, both individually and combined. EPE showed a fungistatic dose-dependent effect, acting mainly through diffusion, while OEO completely prevented mycelial growth at very low concentrations, with a combination of vapor and diffusion activity. Interestingly, OEO was more effective than EPE, as their minimum inhibitory concentrations were 0.39 μl/mL and 5 μl/mL, respectively. Combinations of EPE and OEO showed, depending on the doses, additive or synergistic antifungal effects. In the in vivo trial, dilutions of EPE and OEO made in sterile distilled water (SDW) were tested through a pathosystem with lima bean seeds and S. sclerotiorum. Seeds soaked in formulations with 40% EPE + 12% OEO + 48% SDW, and 20% EPE + 6% OEO + 74% SDW reduced disease incidence of 40% and 60% respectively, significantly decreased severity, with a minimum phytotoxic effect. This study strengthens previous work showing the antifungal action of EPE and OEO against S. sclerotiorum, and, as a main goal, highlights their potential for the development of biofungicides to protect lima bean seeds.




Similar content being viewed by others
Change history
19 July 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10658-022-02543-5
References
Aban, C. L., Taboada, G., Spedaletti, Y., Aparicio, M., Curti, R. N., Casalderrey, N. B., … Galván, M. Z. (2018). Molecular, morphological and pathogenic diversity of Sclerotinia sclerotiorum isolates from common bean (Phaseolus vulgaris) fields in Argentina. Plant Pathology, 67(8), 1740–1748. https://doi.org/10.1111/ppa.12880
Abán, C. L., Taboada, G. M., Casalderrey, N. B., Maggio, M. E., Chocobar, M. O., Spedaletti, Y. A., Gonzalez, M. A. A., Vizgarra, O. V., & Galván, M. Z. (2020). Screening common bean germplasm for resistance to genetically diverse Sclerotinia sclerotiorum isolates from Argentina. Acta Scientiarum. Agronomy, 42, e42786. https://doi.org/10.4025/actasciagron.v42i1.42786
Aban, C. L., Taboada, G. M., Spedaletti, Y., Maita, E., & Galván, M. Z. (2021). Population structure of the fungus Sclerotinia sclerotiorum in common bean fields of Argentina. European Journal of Plant Pathology, 160(4), 841–853. https://doi.org/10.1007/s10658-021-02288-7
Abbey, J. A., Percival, D., Abbey, L., Asiedu, S. K., Prithiviraj, B., & Schilder, A. (2019). Biofungicides as alternative to synthetic fungicide control of grey mould (Botrytis cinerea) - prospects and challenges. Biocontrol Science and Technology, 29(3), 207–228. https://doi.org/10.1080/09583157.2018.1548574
Abo-Elyousr, K. A., Al-Qurashi, A. D., & Almasoudi, N. M. (2021). Evaluation of the synergy between Schwanniomyces vanrijiae and propolis in the control of Penicillium digitatum on lemons. Egyptian Journal of Biological Pest Control, 31(1), 1–10. https://doi.org/10.1186/s41938-021-00415-4
Almasaudi, N. M., Al-Qurashi, A. D., Elsayed, M. I., & Abo-Elyousr, K. A. (2022). Essential oils of oregano and cinnamon as an alternative method for control of gray mold disease of table grapes caused by Botrytis cinerea. Journal of Plant Pathology, 2022, 1–12. https://doi.org/10.1007/s42161-021-01008-8
Aysan, E., & Demir, S. (2009). Using arbuscular mycorrhizal fungi and rhizobium leguminosarum biovar phaseoli against Sclerotinia sclerotiorum (lib.) de Bary in the common bean (Phaseolus vulgaris L.). Plant Pathology Journal, 8(2), 74–78. https://doi.org/10.3923/ppj.2009.74.78
Bankova, V. (2005). Chemical diversity of propolis and the problem of standardization. Journal of Ethnopharmacology, 100(1), 114–117. https://doi.org/10.1016/j.jep.2005.05.004
Bankova, V., Popova, M., & Trusheva, B. (2014). Propolis volatile compounds: Chemical diversity and biological activity: A review. Chemistry Central Journal, 2, 8–28. https://doi.org/10.1186/1752-153X-8-28
Bolton, M. D., Thomma, B. P., & Nelson, B. D. (2006). Sclerotinia sclerotiorum (lib.) de Bary: Biology and molecular traits of a cosmopolitan pathogen. Molecular Plant Pathology, 7(1), 1–16. https://doi.org/10.1111/J.1364-3703.2005.00316.X
Borges, D. F., Lopes, E. A., Moraes, A. R. F., Soares, M. S., Visôtto, L. E., Oliveira, C. R., & Valente, V. M. M. (2018). Formulation of botanicals for the control of plant-pathogens: A review. Crop Protection, 110, 135–140. https://doi.org/10.1016/j.cropro.2018.04.003
Bozin, B., Mimica-Dukic, N., Simin, N., & Anackov, G. (2006). Characterization of the volatile composition of essential oils of some Lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. Journal of Agricultural and Food Chemistry, 54(5), 1822–1828. https://doi.org/10.1021/jf051922u
Chrapačienė, S., Rasiukevičiūtė, N., & Valiuškaitė, A. (2021). Biocontrol of carrot disease-causing pathogens using essential oils. Plants, 10(11), 2231. https://doi.org/10.3390/plants10112231
Cibanal, I. L., Fernández, L. A., Murray, A. P., Pellegrini, C. N., & Gallez, L. M. (2021). Propolis extract and oregano essential oil as biofungicides for garlic seed cloves: In vitro assays and synergistic interaction against Penicillium allii. Journal of Applied Microbiology, 131(4), 1909–1918. https://doi.org/10.1111/jam.15081
Cibanal, I. L., Fernández, L. A., Positano, G. G., Chebataroff, L. B., Garayalde, A. F., Gallez, L. M., & Pérez, E. S. (2020). Chemical characterization and in vitro antimicrobial activity of honeybee propolis and Scaptotrigona jujuyensis geopropolis against tomato pathogenic bacteria. Semina: Ciências Agrárias, 41(5), 1799–1808. https://doi.org/10.5433/1679-0359.2020v41n5p1799
Demirbaş, E., & Karaca, G. H. (2021). Effect of oregano water on Pythium density in soil and damping-off disease on bean plants. International Journal of Agriculture Environment and Food Sciences, 5(3), 333–341. https://doi.org/10.31015/jaefs.2021.3.12
Dudoit, A., Cardinault, N., Mertz, C., Chillet, M., & Brat, P. (2021). Antifungal activities of propolis and its main components with an emphasis against phytopathogenic fungi. Journal of Apicultural Science, 65(1), 5–24. https://doi.org/10.2478/JAS-2021-0013
Fliou, J., Riffi, O., Amechrouq, A., Mohammed, E., & Ghouati, Y. (2020). Comparative study of the chemical composition of the essential oil of Origanum compactum from the seven regions of Morocco and their antimicrobial activity. Journal of microbiology, biotechnology and food Sciences, 10(1), 42–48. https://doi.org/10.15414/jmbfs.2020.10.1.42-48
Gallez, L., Kiehr, M., Fernández, L., Delhey, R., & Stikar, D. (2014). Antifungal activity in vitro of propolis solutions from Argentina against two plant pathogenic fungi: Didymella bryoniae and Rhizotocnia solani. Journal of Apicultural Research, 53(4), 438–440. https://doi.org/10.3896/IBRA.1.53.4.08
Ghisalberti, E. L. (1979). Propolis: A review. Bee World, 60(2), 59–84. https://doi.org/10.1080/0005772X.1979.11097738
Giacometti, J., Kovačević, D. B., Putnik, P., Gabrić, D., Bilušić, T., Krešić, G., … Jambrak, A. R. (2018). Extraction of bioactive compounds and essential oils from mediterranean herbs by conventional and green innovative techniques: A review. Food Research International, 113, 245–262. https://doi.org/10.1016/j.foodres.2018.06.036
Ibáñez, M. D., & Blázquez, M. A. (2018). Phytotoxicity of essential oils on selected weeds: Potential hazard on food crops. Plants, 7(4), 79. https://doi.org/10.3390/plants7040079
Inouye, S., Uchida, K., Maruyama, N., Yamaguchi, H., & Abe, S. (2006). A novel method to estimate the contribution of the vapor activity of essential oils in agar diffusion assay. Nihon Ishinkin Gakkai Zasshi, 47(2), 91–98. https://doi.org/10.3314/jjmm.47.91
Jiang, Z., Jiang, H., & Xie, P. (2013). Antifungal activities against Sclerotinia sclerotiorum by Cinnamomum cassia oil and its main components. Journal of Essential Oil Research, 25(6), 444–451. https://doi.org/10.1080/10412905.2013.782475
Jugreet, B. S., Suroowan, S., Rengasamy, R. K., & Mahomoodally, M. F. (2020). Chemistry, bioactivities, mode of action and industrial applications of essential oils. Trends in Food Science & Technology, 101, 89–105. https://doi.org/10.1016/j.tifs.2020.04.025
Lambert, R. J. W., Skandamis, P. N., Coote, P. J., & Nychas, G. J. (2001). A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. Journal of Applied Microbiology, 91(3), 453–462. https://doi.org/10.1046/j.1365-2672.2001.01428.x
Langeveld, W. T., Veldhuizen, E. J., & Burt, S. A. (2013). Synergy between essential oil components and antibiotics: A review. Critical Reviews in Microbiology, 40(1), 76–94. https://doi.org/10.3109/1040841X.2013.763219
Matny, O. N., Abdul-Karim, E. K., Naemah, R. A., & Al-Ani, R. A. (2014). Activity of propolis and Boswellia sp. resins extract against Sclerotinia sclerotiorum causative agent of white rot disease of Phaseolus vulgaris and Daucus carota under storage conditions. Journal of Experimental Biology and Agricultural Science, 2(1), 65–71. https://doi.org/10.13140/RG.2.2.24334.46406
Miorini, T. J., Kamvar, Z. N., Higgins, R. S., Raetano, C. G., Steadman, J. R., & Everhart, S. E. (2019). Differential aggressiveness of Sclerotinia sclerotiorum isolates from north and South America and partial host resistance in Brazilian soybean and dry bean cultivars. Tropical Plant Pathology, 44(1), 73–81. https://doi.org/10.1007/s40858-018-00273-w
Moody, J. (2004). Synergism testing: Broth microdilution checkerboard and broth macrodilution method. Clinical Microbiology Procedures Handbook, 1–28. https://doi.org/10.1128/9781555818814.ch5.16
Ons, L., Bylemans, D., Thevissen, K., & Cammue, B. (2020). Combining biocontrol agents with chemical fungicides for integrated plant fungal disease control. Microorganisms, 8(12), 1930. https://doi.org/10.3390/microorganisms8121930
Ordóñez, R. M., Zampini, I. C., Moreno, M. N., & Isla, M. I. (2011). Potential application of northern argentine propolis to control some phytopathogenic bacteria. Microbiological Research, 166(7), 578–584. https://doi.org/10.1016/j.micres.2010.11.006
Orhan, G., Bayram, A., Zer, Y., & Balci, I. (2005). Synergy tests by E test and checkerboard methods of antimicrobial combinations against Brucella melitensis. Journal of Clinical Microbiology, 43(1), 140–143. https://doi.org/10.1128/JCM.43.1.140-143.2005
Parekh, J., & Chanda, S. (2007). Antibacterial and phytochemical studies on twelve species of Indian medicinal plants. African Journal of Biomedical Research, 10(2), 175–181. https://doi.org/10.4314/ajbr.v10i2.50624
Pazin, W. M., Santos, S. N., Queiroz, S. C., Bagatolli, L. A., Soares, A. E., Melo, I. S., & Ito, A. S. (2019). Bioactivity and action mechanism of green propolis against Pythium aphanidermatum. Anais da Academia Brasileira de Ciências, 91(2), 1–9. https://doi.org/10.1590/0001-3765201920180598
Peng, L., Yang, S., Cheng, Y. J., Chen, F., Pan, S., & Fan, G. (2012). Antifungal activity and action mode of pinocembrin from propolis against Penicillium italicum. Food Science and Biotechnology, 21(6), 1533–1539. https://doi.org/10.1007/s10068-012-0204-0
Portella, J., Orlandi, R. C., Almeida, J., Koefender, J., Schoffel, A., & Camera, J. N. (2021). Óleos essenciais no controle in vitro de Sclerotinia sclerotiorum. Revista Thema, 19(3), 615–622. https://doi.org/10.15536/thema.V19.2021.615-622.2367
Quiroga, E. N., Sampietro, D. A., Soberon, J. R., Sgariglia, M. A., & Vattuone, M. A. (2006). Propolis from the northwest of Argentina as a source of antifungal principles. Journal of Applied Microbiology, 101(1), 103–110. https://doi.org/10.1111/j.1365-2672.2006.02904
Radünz, M., Camargo, T. M., Dos Santos Hackbart, H. C., Alves, P. I. C., Radünz, A. L., Gandra, E. A., & da Rosa Zavareze, E. (2021). Chemical composition and in vitro antioxidant and antihyperglycemic activities of clove, thyme, oregano, and sweet orange essential oils. LWT, 138, 110632. https://doi.org/10.1016/j.lwt.2020.110632
Reyes-Jurado, F., Franco-Vega, A., Ramírez-Corona, N., Palou, E., & López-Malo, A. (2015). Essential oils: Antimicrobial activities, extraction methods, and their modeling. Food Engineering Reviews, 7(3), 275–297. https://doi.org/10.1007/s12393-014-9099-2
Rienth, M., Crovadore, J., Ghaffari, S., & Lefort, F. (2019). Oregano essential oil vapour prevents Plasmopara viticola infection in grapevine (Vitis Vinifera) and primes plant immunity mechanisms. PLoS One, 14(9), 1–29. https://doi.org/10.1371/journal.pone.0222854
Rodriguez-Garcia, I., Silva-Espinoza, B. A., Ortega-Ramirez, L. A., Leyva, J. M., Siddiqui, M. W., Cruz-Valenzuela, M. R., Gonzalez-Aguilar, G. A., & Ayala-Zavala, J. F. (2015). Oregano essential oil as an antimicrobial and antioxidant additive in food products. Critical Reviews in Food Science and Nutrition, 56(10), 1717–1727. https://doi.org/10.1080/10408398.2013.800832
Sawaya, A. C. H. F., Palma, A. M., Caetano, F. M., Marcucci, M. C., Da Silva Cunha, I. B., Araujo, C. E. P., & Shimizu, M. T. (2002). Comparative study of in vitro methods used to analyse the activity of propolis extracts with different compositions against species of Candida. Letters in Applied Microbiology, 35(3), 203–207. https://doi.org/10.1046/j.1472-765X.2002.01169.x
Sforcin, J. M. (2016). Biological properties and therapeutic applications of propolis. Phytotherapy Research, 30(6), 894–905. https://doi.org/10.1002/ptr.5605
Smolińska, U., & Kowalska, B. (2018). Biological control of the soil-borne fungal pathogen Sclerotinia sclerotiorum - a review. Journal of Plant Pathology, 100(1), 1–12. https://doi.org/10.1007/s42161-018-0023-0
Soylu, S., Yigitbas, H., Soylu, E. M., & Kurt, Ş. (2007). Antifungal effects of essential oils from oregano and fennel on Sclerotinia sclerotiorum. Journal of Applied Microbiology, 103(4), 1021–1030. https://doi.org/10.1111/j.1365-2672.2007.03310.x
Suárez, G. A. P., Galindo, N. J. P., & Cuervo, O. H. P. (2022). Obtaining Colombian propolis extracts using modern methods: A determination of its antioxidant capacity and the identification of its bioactive compounds. The Journal of Supercritical Fluids, 182(2022), 105538. https://doi.org/10.1016/j.supflu.2022.105538
Tarlanović, J., Grahovac, M., Milić, B., Keserović, Z., Magazin, N., Petreš, M., & Miodragović, M. (2017). Effect of storage conditions on virulence of fusarium avenaceum and Alternaria alternata on apple fruits. Journal of Phytopathology, 165(9), 595–601. https://doi.org/10.1111/jph.12597
Türkmen, M., Kara, M., Maral, H., & Soylu, S. (2021). Determination of chemical component of essential oil of Origanum dubium plants grown at different altitudes and antifungal activity against Sclerotinia sclerotiorum. Journal of Food Processing and Preservation, 2021, 1–10. https://doi.org/10.1111/jfpp.15787
Werrie, P. Y., Durenne, B., Delaplace, P., & Fauconnier, M. L. (2020). Phytotoxicity of essential oils: Opportunities and constraints for the development of biopesticides. A review. Foods, 9(9), 1291. https://doi.org/10.3390/foods9091291
Willbur, J., McCaghey, M., Kabbage, M., & Smith, D. L. (2019). An overview of the Sclerotinia sclerotiorum pathosystem in soybean: Impact, fungal biology, and current management strategies. Tropical Plant Pathology, 44(1), 3–11. https://doi.org/10.1007/s40858-018-0250-0
Acknowledgments
The authors are particularly thankful to CIC (Comisión de Investigaciones Científicas, Argentina), and to CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas) for financial support. Authors would also like to thank Ing. Agr. Antonela Gil and Dr. Ana Paula Murray for their technical support, and Dr. Antonio Garayalde for his statistical assistance.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Irene Cibanal performed the experiments, analyzed the results and wrote the manuscript; Leticia Andrea Fernández contributed to the implementation of the research, the analysis of the results and the writing of the manuscript; Silvana Andrea Rodríguez performed part of the chemical characterization; Cecilia Pellegrini made substantial contributions revising the manuscript; Liliana Gallez critically revised the manuscript for important intellectual content.
Corresponding author
Ethics declarations
Research involving human participants and/or animals
The authors confirm there was no research on any humans or animals.
Consent to participate
All authors have reviewed the manuscript and consented the submission to the European Journal of Plant Pathology.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Rights and permissions
About this article
Cite this article
Cibanal, I.L., Fernández, L.A., Rodriguez, S.A. et al. Propolis extract combined with oregano essential oil applied to lima bean seeds against Sclerotinia sclerotiorum. Eur J Plant Pathol 164, 33–43 (2022). https://doi.org/10.1007/s10658-022-02536-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-022-02536-4