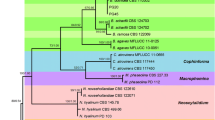
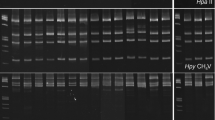
Abstract
Venturia inaequalis isolates (causing apple scab) vary in pathogenicity when infecting common susceptible apple cultivars. This quantitative effect may be due to variation in the pathogen’s virulence factors. South African V. inaequalis isolates were collected in seasons 2012/13 and 2013/14 from various cultivars in the four principal apple growing regions and were characterized using phylogenetic analysis. A partial sequence of the candidate virulence factor ABC2-NB2 gene region was analysed; this gene region was highly polymorphic with six possible haplotypes, and non-synonymous changes were identified. Most of the SA isolates had the amino acid changes V1005I (41%) or S971P (47%) and 5% had two amino acid changes, namely serine to proline (S971P) and arginine to cysteine (R957C). None of the SA isolates were identical to the ABC-NB2 gene Genbank reference sequence. The ABC2-NB2 gene region was found to be under positive/diversifying selection pressure indicating its potential role as virulence factor, while other parts of the gene were under negative selection. The 15 V. inaequalis isolates differed with respect to their conidia production on the media and their germination rate. Isolates LOSL 206-1 and KCL 3-1 produced the most conidia, with 1.5 × 106 and 1 × 106 spores/mL, and they were among those of the highest viability, with germination rates of 92% and 82%, respectively. Pathogenicity testing with conidiospore inoculum from these single spore isolates on common cultivars ‘Fuji’, ‘Golden Delicious’, ‘Royal Gala’ and the rootstock ‘M793’ showed that not all isolates could infect and sporulate on all the cultivars. Some single spore isolates caused chlorotic spots without any sporulation and may have presented a delayed sporulation if incubated for 21 days. A qPCR method was evaluated as a molecular tool to quantify severity of apple scab infection. The amount of fungal DNA in the leaf tissue, indicating variation in virulence, was quantified using qPCR after single spore inoculations during 2015 on ‘Golden Delicious’, ‘Royal Gala’ and ‘M793’ at 7 and 14 days after inoculation. The fungal DNA concentration was lower in inoculations of ‘Golden Delicious’ and ‘Royal Gala’ and higher in ‘M793’. The single spore isolate LOSL 206-1, from ‘Cripps Red’, was the most virulent isolate on ‘Royal Gala’ and isolate OBL 210-1, from ‘Braeburn’, the most virulent on ‘M793’. Isolates that caused chlorosis also gave high amounts of fungal DNA. This study concludes that the single spore isolates differed in their virulence and infection ability with respect to different apple cultivars and the qPCR method reliably detected V. inaequalis DNA in asymptomatic Malus tissue.






Similar content being viewed by others
References
Barbara, D. J., Roberts, A. L., & Xu, X. M. (2008). Virulence characteristics of apple scab (Venturia inaequalis) isolates from monoculture and mixed orchards. Plant Pathology, 57, 552–561. https://doi.org/10.1111/j.1365-3059.2007.01781.x
Bus, V. G. M., Rikkerink, E. H., Caffier, V., Durel, C. E., & Plummer, K. M. (2018). Revision of the nomenclature of the differential host-pathogen interactions of Venturia inaequalis and Malus. Annual Reviews in Phytopathology, 49, 391–413. https://doi.org/10.1146/annurev-phyto-072910-095339
Chevalier, M., Lespinasse, Y., & Renaudin, S. (1991). A microscopic study of the different classes of symptoms coded by the Vf gene in apple for resistance to scab (Venturia inaequalis). Plant Pathology, 40, 249–256. https://doi.org/10.1111/j.1365-3059.1991.tb02374.x
Coleman, J. J., & Mylonakis, E. (2009). Efflux in fungi: La piece de resistance. PLoS Pathogens, 5, e1000486. https://doi.org/10.1371/journal.ppat.1000486
Daniëls, B., De Landtsheer, A., Dreesen, R., Davey, M. W., & Keulemans, J. (2012). Real-time PCR as a promising tool to monitor growth of Venturia spp. in scab-susceptible and -resistant apple leaves. European Journal of Plant Pathology, 134, 821–833. https://doi.org/10.1007/s10658-012-0058-6
De Waard, M. A. (1997). Significance of ABC transporters in fungicide sensitivity and resistance. Pesticide Science, 51, 271–275. https://doi.org/10.1002/(SICI)1096-9063(199711)51:3<271::AID-PS642>3.0.CO;2-%23
Degenhardt, J., Al-Masri, A. N., Kürkcüoglu, S., Szankowski, I., & Gau, A. E. (2005). Characterization by suppression subtractive hybridization of transcripts that are differentially expressed in leaves of apple scab-resistant and susceptible cultivars of Malus domestica. Molecular Genetics and Genomics, 273, 326–335. https://doi.org/10.1007/s00438-005-1136-7
Fernández-Fernández, F. (2010). Final report of Defra project GC0140 ‘fingerprinting the national apple and pear collections’. Retrieved February, 20, 2013 from http://randd.defra.gov.uk/
Fleissner, A., Sopalla, C., & Weltring, K. M. (2002). An ATP-binding cassette multidrug-resistance transporter is necessary for tolerance of Gibberella pulicaris to phytoalexins and virulence on potato tubers. Molecular Plant-Microbe Interactions, 15, 102–108. https://doi.org/10.1094/MPMI.2002.15.2.102
Flor, H. H. (1971). Current status of the gene-for-gene concept. Annual Review of Phytopathology, 9, 275–296.
Gessler, C., Patocchi, A., Sansavini, S., Tartarini, S., & Gianfranceschi, L. (2006). Venturia inaequales resistance in apple. Critical Reviews in Plant Sciences, 25, 473–503. https://doi.org/10.1080/07352680601015975
Gusberti, M., Patocchi, A., Gessler, C., & Broggini, G. A. L. (2012). Quantification of Venturia inaequalis growth in Malus x domestica with quantitative real-time polymerase chain reactions. Plant Disease, 12, 1791–1797. https://doi.org/10.1094/PDIS-12-11-1058-RE
Higgins, C. F. (1992). ABC transporters: From microorganisms to man. Annual Review of Cell Biology, 8, 67–113. https://doi.org/10.1146/annurev.cb.08.110192.000435
Hrazdina, G., Borejsza-Wysocki, W., & Lester, C. (1997). Phytoalexin production in an apple cultivar resistant to Venturia inaequalis. Phytopathology, 87, 868–876. https://doi.org/10.1094/PHYTO.1997.87.8.868
Jha, G., Thakur, K., & Thakur, P. (2009). The Venturia-apple pathosystem: Pathogenicity mechanisms and plant defence responses. Journal of Biomedicine and Biotechnology, 2009, 680160. https://doi.org/10.1155/2009/680160
Kearse, M., Moir, R., Wilson, A., Stones-Havas, S., Cheung, M., Sturrock, S., Buxton, S., Cooper, A., Markowitz, S., Duran, C., Thierer, T., Ashton, B., Mentjies, P., & Drummond, A. (2012). Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28, 1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Koch, T., Kellerhals, M., & Gessler, C. (2000). Virulence pattern of Venturia inaequalis field isolates and corresponding differential resistance in Malus x domestica. Journal of Phytopathology, 148, 357–364. https://doi.org/10.1046/j.1439-0434.2000.00523.x
Köller, W., Smith, F. D., & Reynolds, K. L. (1991). Phenotypic instability of flusilazole sensitivity in Venturia inaequalis. Plant Pathology, 40, 608–611. https://doi.org/10.1111/j.1365-3059.1991.tb02425.x
Koopman, T., Meitz-Hopkins, J., Bester-van der Merwe, A. E., Tobutt, K. R., Bester, C., & Lennox, C. L. (2017). Genetic diversity and gene flow of four south African Venturia inaequalis (apple scab) populations. Phytopathology, 107, 455–462. https://doi.org/10.1094/PHYTO-07-16-0279-R
Lemaire, C., De Gracia, M., Leroy, T., Michalecka, M., Lindhard-Pedersen, H., Guerin, F., Gladieux, P., & Le Cam, B. (2016). Emergence of new virulent populations of apple scab from nonagricultural disease reservoirs. New Phytologist, 209, 1220–1229. https://doi.org/10.1111/nph.13658
Li, W. H., Wu, C. I., & Luo, C. C. (1985). A new method for estimating synonymous and nonsynonymous rates of nucleotide substitution considering the relative likelihood of nucleotide and codon changes. Molecular Biology and Evolution, 2, 150–174. https://doi.org/10.1093/oxfordjournals.molbev.a040343
MacHardy, W. E. (1996). Apple scab: Biology, epidemiology and management. The American Phytopathological Society.
Makizumi, Y., Takeda, S., Matsuzaki, Y., Nakaune, R., Hamamoto, H., Akutsu, K., & Hibi, T. (2002). Cloning and selective toxicant-induced expression of BMR1 and BMR3, novel ABC transporter genes in Botrytis cinerea. Journal of General Plant Pathology, 68, 338–341. https://doi.org/10.1007/PL00013100
Meitz-Hopkins, J. C., von Diest, S. G., Koopman, T. A., Bahramisharif, A., & Lennox, C. L. (2014). A method to monitor airborne Venturia inaequalis ascospores using volumetric spore traps and quantitative PCR. European Plant Pathology, 140, 527–541. https://doi.org/10.1007/s10658-014-0486-6
Mhelembe, K.G. (2015). Molecular characterisation of ARC pome fruit collections in South Africa. MSc Thesis. Stellenbosch University.
Nakajima, M., Suzuki, J., Hosaka, T., Hibi, T., & Akutsu, K. (2001). Functional analysis of an ATP-binding cassette transporter gene in Botrytis cinerea by gene disruption. Journal of General Plant Pathology, 67, 212–214. https://doi.org/10.1007/PL00013014
Ortega, S. F., Prencipe, S., Gullino, M. L., & Spadaro, D. (2020). New molecular tool for a quick and easy detection of apple scab in the field. Agronomy, 10, 581. https://doi.org/10.3390/agronomy10040581
Parker, D. M., Hilber, U. W., Bodmer, M., Smith, F. D., Yao, C., & Köller, W. (1995). Production and transformation of conidia of Venturia inaequalis. Phytopathology, 85, 87–91. https://doi.org/10.1094/Phyto-85-87
Rees, D. J. G., Celton, J. M., Sargent, D. J., Xu, X., & Cristoffels, A. (2010). Whole-genome SNP sequencing of selected progeny individuals allows anchoring of draft genome sequence to a linkage map for the apple scab pathogen Venturia inaequalis. Acta Horticulturae, 859, 295–300. https://doi.org/10.17660/ActaHortic.2010.859.34
Rozas, J., & Rozas, R. (1999). DnaSP version 3: An integrated program for molecular population genetics and molecular evolution analysis. Bioinformatics, 19, 2496–2497. https://doi.org/10.1093/bioinformatics/15.2.174
Rozen, S., & Skaletsky, H. J. (2000). Primer 3 on the WWW for general users and for biologist programmers. In S. Krawetz & S. Misener (Eds.), Bioinformatics methods and protocols: Methods in molecular biology (pp. 365–386). Humana Press.
Schena, L., Nigro, F., Ippolito, A., & Gallitelli, D. (2004). Real-time quantitative PCR: A new technology to detect and study phytopathogenic and antagonistic fungi. European Journal of Plant Pathology, 110, 893–908. https://doi.org/10.1007/s10658-004-4842-9
Schnabel, G., & Jones, A. L. (2001). The 14 alpha-demethylase (CYP51A1) gene is overexpressed in Venturia inaequalis strains resistant to myclobutanil. Phytopathology, 91, 102–110. https://doi.org/10.1094/PHYTO.2001.91.1.102
Schnabel, G., Schnabel, E., & Jones, A. L. (2002). Genes encoding multidrug resistance like proteins in Venturia inaequalis. In H. W. Dehne, U. Gisi, K. H. Kuck, P. E. Russell, & H. Lyr (Eds.), Modern fungicides and antifungal compounds III. Proceedings of the13th International Reinhardsbrunn Symposium, May 14–18 (pp. 297–305).
Schnabel, G., Dait, Q., & Paradkar, M. R. (2003). Cloning and expression analysis of the ATP-binding cassette transporter gene MFABC1 and the alternative oxidase gene MfAOX1 from Monilinia fructicola. Pest Management Science, 59, 1143–1151. https://doi.org/10.1002/ps.744
Schoonbeek, H. J., Del Sorbo, G., & De Waard, M. A. (2001). The ABC transporter BcatrB affects the sensitivity of Botrytis cinerea to the phytoalexin resveratrol and the fungicide fenpiclonil. Molecular Plant-Microbe Interactions, 14, 562–571. https://doi.org/10.1094/MPMI.2001.14.4.562
Soufflet-Freslon, V., Gianfranceschi, L., Patocchi, A., & Durel, C. E. (2008). Inheritance studies of apple scab resistance and identification of Rvi14, a new major gene that acts together with other broad-spectrum QTL. Genome, 51, 657–667. https://doi.org/10.1139/G08-046
Stergiopoulos, I., Zwiers, L. H., & De Waard, M. A. (2002). Secretion of natural and synthetic toxic compounds from filamentous fungi by membrane transporters of the ATP-binding cassette and major facilitator superfamily. European Journal of Plant Pathology, 108, 719–734. https://doi.org/10.1023/A:1020604716500
Swofford, D. L. (1998). PAUP. Phylogenetic analysis using parsimony, version 4.0b10. Sinauer Association, Inc..
Tajima, F. (1989). Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics, 123, 585–595. https://doi.org/10.1093/genetics/123.3.585
Urban, M., Bhargava, T., & Hamer, J. E. (1999). An ATP-driven efflux pump is a novel pathogenicity factor in rice blast disease. EMBO Journal, 18, 512–521. https://doi.org/10.1093/emboj/18.3.512
Vela-Corcía, D., Srivastava, D. A., Dafa-Berger, A., Rotem, N., Barda, O., & Levy, M. (2019). MFS transporter from Botrytis cinerea provides tolerance to glucosinolate-breakdown products and is required for pathogenicity. Nature Communications, 10, 2886. https://doi.org/10.3389/fpls.2021.651716
VINQUEST initiative (2022). http://www.vinquest.ch/monitoring/publication.htm
Waalwijk, C., Van der Heide, R., De Vries, I., Van der Lee, T., Schoen, C., Costrel-de Corainville, G., Häuser-Hahn, I., Kastelein, P., Köhl, J., Lonnet, P., Dermarquet, T., & Kema, G. H. J. (2004). Quantitative detection of fusarium species in wheat using TaqMan. European Journal of Plant Pathology, 110, 481–494. https://doi.org/10.1023/B:EJPP.0000032387.52385.13
Westrick, N. M., Smith, D. L., & Kabbage, M. (2021). Disarming the host: Detoxification of plant defense compounds during fungal necrotrophy. Frontiers in Plant Science, 12, 651716. https://doi.org/10.3389/fpls.2021.651716
Acknowledgements
We would like to thank the producers that participated in this study and Hortgro and the national Research Foundation (NRF) for incentive and THRIP funding.
Funding
The project was funded as part of the Hortgro Science project 2100-33 and NRF THRIP project TP1208035615.
Author information
Authors and Affiliations
Contributions
Trevor A. Koopman: Funding Acquisition, Investigation, Data collection and curation, Methodology, Formal Analysis and investigation, Writing – original draft, Julia C. Meitz-Hopkins: Conceptualization, Funding Acquisition, Methodology, Writing – review and editing, Visualization, Supervision, Ken Tobutt: Conceptualization, Funding Acquisition, Methodology, Writing – review and editing, Visualization, Supervision, Cecilia Bester: Resources, Writing – review and editing, Cheryl L. Lennox: Resources, Writing – review and editing, Supervision.
Corresponding author
Ethics declarations
Conflicts of interests/competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
About this article
Cite this article
Koopman, T.A., Meitz-Hopkins, J.C., Tobutt, K.R. et al. Pathogenicity and virulence of south African isolates of Venturia inaequalis. Eur J Plant Pathol 164, 45–58 (2022). https://doi.org/10.1007/s10658-022-02537-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-022-02537-3