Abstract
Background
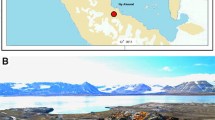
Lichen is a symbiotic association of algae and fungi, recognized as a self-sustaining ecosystem that constitutes an indeterminant number of bacteria, actinomycetes, fungi, and protozoa. We evaluated the endolichenic fungal assemblage given the dearth of knowledge on endolichenic fungi (ELFs), particularly from part of the Central Western Ghats, Karnataka, and conducted a phylogenetic analysis of xylariaceous fungi, the most diversified group of fungi using ITS and ITS+Tub2 gene set.
Results
Out of 17 lichen thalli collected from 5 ecoregions, 42 morphospecies recovered, belong to the class Sordariomycetes, Eurotiomycetes, Dothideomycetes, Leotiomycetes, Saccharomycetes. About 19 and 13 ELF genera have been reported from Parmotrema and Heterodermia thallus. Among the ecoregions EC2 showing highest species diversity (Parmotrema (1-D) = 0.9382, (H) = 2.865, Fisher-α = 8.429, Heterodermia (1-D) = 0.8038, H = 1.894, F-α = 4.57) followed the EC3 and EC1. Xylariales are the predominant colonizer reported from at least one thallus from four ecoregions. The morphotypes ELFX04, ELFX05, ELFX08 and ELFX13 show the highest BLAST similarity (> 99%) with Xylaria psidii, X. feejeensis, X. berteri and Hypoxylon fragiforme respectively. Species delimitation and phylogenetic position reveal the closest relation of Xylariaceous ELFs with plant endophytes.
Conclusions
The observation highlights that the deciduous forest harness a high number of endolichenic fungi, a dominant portion of these fungi are non-sporulating and still exist as cryptic. Overall, 8 ELF species recognized based on phylogenetic analysis, including the two newly reported fungi ELFX03 and ELFX06 which are suspected to be new species based on the present evidence. The study proved, that the lichen being rich source to establish fungal diversity and finding new species. Successful amplification of most phylogenetic markers like RPB2, building of comprehensive taxonomic databases and application of multi-omics data are further needed to understand the complex nature of lichen-fungal symbiosis.







Similar content being viewed by others
Abbreviations
- ELF:
-
Endolichenic fungi
- ITS:
-
Internal transcribed spacer
- Tub2:
-
Beta tubulin subunit 2
- Tef1:
-
α Translation elongation factor 1 alfa
- PDA:
-
Potato dextrose agar
- MYA:
-
Malt yeast extract agar
- SDA:
-
Sabouraud dextrose agar
- CR:
-
Colonization rate
- EC:
-
Ecoregion
- OUT:
-
Operational taxonomic unit
- NMDS:
-
Non metric multidimensional statistical analysis
References
Hawksworth DL, Grube M (2020) Lichens redefined as complex ecosystems. New Phytol 227(5):1281–1283. https://doi.org/10.1111/nph.16630
Bates ST, Donna BL, Lauber CL, Walters WA, Knight R, Fierer N (2012) A preliminary survey of lichen associated eukaryotes using pyrosequencing. Lichenologist 44:137–146. https://doi.org/10.1017/S0024282911000648
Grube M, Berg G (2009) Microbial consortia of bacteria and fungi with focus on the lichen symbiosis. Fungal Biol Rev 23:72–85. https://doi.org/10.1016/j.fbr.2009.10.001
Grube M, Wedin M (2016) Lichenized fungi and the evolution of symbiotic organization. Microbiol Spectr 4(6):4–6. https://doi.org/10.1128/microbiolspec.funk-0011-2016
Muggia L, Grube M (2018) Fungal diversity in lichens: from extremotolerance to interactions with algae. Life 8(2):15
Suryanarayanan TS, Thirunavukkarasu N (2017) Endolichenic fungi: the lesser known fungal associates of lichens. Mycology 8(3):189–196. https://doi.org/10.1080/21501203.2017.1352048
Honegger R, Axe L, Edwards D (2013) Bacterial epibionts and endolichenic actinobacteria and fungi in the lower Devonian lichen Chlorolichenomycites salopensis. Fungal Biol 117:512–518
Mark K, Laanisto L, Bueno CG, Niinemets Ü, Keller C, Scheidegger C (2020) Contrasting co-occurrence patterns of photobiont and Cystobasidiomycete yeast associated with common epiphytic lichen species. New Phytol 227(5):1362–1375. https://doi.org/10.1111/nph.16475
Spribille T, Tuovinen V, Resl P, Vanderpool D, Wolinski H, Aime MC, McCutcheon JP (2016) Basidiomycete yeasts in the cortex of ascomycete macrolichens. Science 353(6298):488–492. https://doi.org/10.1126/science.aaf8287
Arnold AE, Miadlikowska J, Higgins KL, Sarvate SD, Gugger P, Way A, Hofstetter V, Kauff F, Lutzoni F (2009) A phylogenetic estimation of trophic transition networks for ascomycetous fungi: are lichens cradles of symbiotrophic fungal diversification? Syst Biol 58:283–297. https://doi.org/10.1093/sysbio/syp001
Girlanda M, Isocrono D, Bianco C, Luppi-Mosca AM (1997) Two foliose lichens as microfungal ecological niches. Mycologia 89:531–536. https://doi.org/10.1080/00275514.1997.12026814
Lagarde A, Millot M, Pinon A, Liagre B, Girardot M, Imbert C, Mambu L (2019) Antiproliferative and antibiofilm potentials of endolichenic fungi associated with the lichen Nephroma laevigatum. J Appl Microbiol 126(4):1044–1058
Petrini O, Hake U, Dreyfuss MM (1990) An analysis of fungal communities isolated from fruticose lichens. Mycologia 82(4):444–451. https://doi.org/10.1080/00275514.1990.12025907
Zhang T, Wei XL, Wei YZ, Liu HY, Yu LY (2016) – Diversity and distribution of cultured endolichenic fungi in the Ny-Ålesund Region, Svalbard (High Arctic). Sci Rep 20:461–470. https://doi.org/10.1007/s00792-016-0836-8
Tan MA, Castro SG, Oliva PMP, Yap PRJ, Nakayama A, Magpantay HD, dela Cruz TEE (2020) Biodiscovery of antibacterial constituents from the endolichenic fungi isolated from Parmotrema rampoddense. 3 Biotech 10:1–7
Yu NH, Park SY, Kim JA, Park CH, Jeong MH, Oh SO, Hur JS (2018) Endophytic and endolichenic fungal diversity in maritime Antarctica based on cultured material and their evolutionary position among Dikarya. Fungal Syst Evol 2(1):263–272. https://doi.org/10.3114/fuse.2018.02.07
Li WC, Zhou J, Guo SY, Guo LD (2007) Endophytic fungi associated with lichens in Baihua mountain of Beijing, China. Fungal Divers 25:69–80
Suryanarayanan TS, Thirunavukkarasu N, Hariharan GN, Balaji P (2005) Occurrence of non-obligate microfungi inside lichen. Sydowia 57(1):120–130
Kannangara BTSDP, Rajapaksha RSCG, Paranagama PA (2009) Nature and bioactivities of endolichenic fungi in Pseudocyphellaria sp., Parmotrema sp. and Usnea sp. at Hakgala montane forest in Sri Lanka. Lett Appl Microbiol 48(2):203–209. https://doi.org/10.1111/j.1472-765X.2008.02512.x
Tripathi M, Joshi Y, Gupta RC (2014) Assessment of endolichenic fungal diversity in some forests of Kumaun Himalaya. Current science 107(5):745–748
Vinayaka KS (2011) Studies on diversity distribution and ecology of Macrolichens occurring in Central western Ghats of Karnataka. Doctoral dissertation to Kuvempu University
Chakarwarti J, Nayaka S, Srivastava S (2020) Diversity of endolichenic fungi–a review. Asian J Mycol 3(1):488–509. https://doi.org/10.5943/ajom/3/1/18
Vinayaka KS, Krishnamurthy YL, Banakar S, Kekuda TP (2016) Association and variation of endophytic fungi among some macrolichens in central Western Ghats, Southern India. Int J Curr Microbiol Appl Sci 5:115–124. https://doi.org/10.20546/ijcmas.2016.506.014
Nelson A, Vandegrift R, Carroll GC, Roy BA (2020) Double lives: transfer of fungal endophytes from leaves to woody substrates. PeerJ 8:e9341
Wijayawardene NN, Hyde KD, Al-Ani LKT, Tedersoo L, Haelewaters D, Rajeshkumar KC, Suija A (2020) Outline of Fungi and fungus-like taxa. Mycosphere Online J Fungal Biol 11(1):1060–1456. https://doi.org/10.5943/mycosphere/11/1/8
Stadler M, Kuhnert E, Peršoh D, Fournier J (2013) The Xylariaceae as model example for a unified nomenclature following the “One Fungus-One Name” (1F1N) concept. Mycology 4(1):5–21
Kuhnert E, Sir EB, Lambert C, Hyde KD, Hladki AI, Romero AI, Stadler M (2017) Phylogenetic and chemotaxonomic resolution of the genus Annulohypoxylon (Xylariaceae) including four new species. Fungal Diversity 85:1–43
Lambert C, Wendt L, Hladki AI, Stadler M, Sir EB (2019) Hypomontagnella (Hypoxylaceae): a new genus segregated from Hypoxylon by a polyphasic taxonomic approach. Mycol Prog 18:187–201
Maharachchikumbura SSN, Chen Y, Ariyawansa HA (2021) Integrative approaches for species delimitation in Ascomycota. Fungal Diversity 109:155–179. https://doi.org/10.1007/s13225-021-00486-6
Davis EC, Franklin JB, Shaw AJ, Vilgalys R (2003) Endophytic Xylaria (Xylariaceae) among liverworts and angiosperms: phylogenetics, distribution, and symbiosis. Am J Bot 90(11):1661–1667
Ma X, Chomnunti P, Doilom M, Daranagama DA, Kang J (2022) Multigene phylogeny reveals endophytic Xylariales novelties from dendrobium species from Southwestern China and Northern Thailand. Journal of Fungi 8(3):248
U’Ren JM, Miadlikowska J, Zimmerman NB, Lutzoni F, Stajich JE, Arnold AE (2016) Contributions of North American endophytes to the phylogeny, ecology, and taxonomy of Xylariaceae (Sordariomycetes, Ascomycota). Mol Phylogenet Evol 98:210–232
Awasthi DD (2007) A Compendium of the Macrolichens from India, Nepal and ShriLanka, Bishen Singh Mahendra Pal Singh, Dehra Dun, India ISBN: 978-81211-0600-9
Orange A, James PW, White FJ (2001) Microchemical methods for the identification of lichens. British Lichen Society, London
Seifert KG, Morgan-Joan W, Gams B, Kendrick (2011) The genera of Hyphomycetes. CBS-KNAW Fungal Biodiversity Centre. ISBN:978-90-70351-85-4
Rogers SO, Bendich AJ (1994) Extraction of total cellular DNA from plants algae and fungi. Plant molecular biology manual. Springer, Dordrecht, pp 183–190
Zhang J, Kapli P, Pavlidis P, Stamatakis A (2013) A general species delimitation method with applications to phylogenetic placements. Bioinformatics 29(22):2869–2876. https://doi.org/10.1093/bioinformatics/btt499
Roswell M, Dushoff J, Winfree R (2021) A conceptual guide to measuring species diversity. Oikos 130(3):321–338. https://doi.org/10.1111/oik.07202
Thompson GG, Thompson SA (2007) Using species accumulation curves to estimate trapping effort in fauna surveys and species richness. Austral Ecol 32(5):564–569. https://doi.org/10.1111/j.1442-9993.2007.01728.x
Wang Y, Zheng Y, Wang X, Wei X, Wei J (2016) Lichen-associated fungal community in Hypogymnia hypotrypa (Parmeliaceae, Ascomycota) affected by geographic distribution and altitude. Front Microbiol 7:1231. https://doi.org/10.3389/fmicb.2016.01231
Chakarwarti J, Nayaka S, Srivastava S (2023) Diversity of endolichenic fungi within lichen genus Parmotrema from India. Turk J Bot 47(4):291–306. https://doi.org/10.55730/1300-008X.2767
Maduranga K, Attanayake RN, Santhirasegaram S, Weerakoon G, Paranagama PA (2018) Molecular phylogeny and bioprospecting of Endolichenic Fungi (ELF) inhabiting in the lichens collected from a mangrove ecosystem in Sri Lanka. PLoS ONE 13(8):e0200711. https://doi.org/10.1371/journal.pone.0200711
U’Ren JM, Lutzoni F, Miadlikowska J, Laetsch AD, Arnold AE (2012) Host and geographic structure of endophytic and endolichenic fungi at a continental scale. Am J Bot 99(5):898–914. https://doi.org/10.3732/ajb.1100459
Oh SY, Yang JH, Woo JJ, Oh SO, Hur JS (2020) Diversity and distribution patterns of endolichenic fungi in Jeju Island. South Korea Sustain 12(9):3769. https://doi.org/10.3390/su12093769
U’Ren JM, Lutzoni F, Miadlikowska J, Arnold AE (2010) Community analysis reveals close affinities between endophytic and endolichenic fungi in mosses and lichens. Microb Ecol 60:340–353. https://doi.org/10.1007/s00248-010-9698-2
Chagnon PL, U’Ren JM, Miadlikowska J, Lutzoni F, Elizabeth Arnold A (2016) Interaction type influences ecological network structure more than local abiotic conditions: evidence from endophytic and endolichenic fungi at a continental scale. Oecologia 180:181–191. https://doi.org/10.1007/s00442-015-3457-5
Yang JH, Oh SY, Kim W, Woo JJ, Kim H, Hur JS (2021) Effect of isolation conditions on diversity of endolichenic fungal communities from a foliose lichen Parmotrema tinctorum. J Fungi 7(5):335. https://doi.org/10.3390/jof7050335
Muggia L, Kopun T, Grube M (2017) Effects of growth media on the diversity of culturable fungi from lichens. Molecules 22(5):824. https://doi.org/10.3390/molecules22050824
Yang JH, Oh SY, Kim W, Hur JS (2022) Endolichenic fungal community analysis by pure culture isolation and metabarcoding: a case study of Parmotrema tinctorum. Mycobiology 50(1):55–65. https://doi.org/10.1080/12298093.2022.2040112
Santiago KAA, dela Cruz TEE, Ting ASY (2021) Diversity and bioactivity of endolichenic fungi in Usnea lichens of the Philippines. Czech Mycol. 73(1):1–19
Govindarajulu MB, Thirunavukkarasu N, Kumar SS, Kaur T, Reddy MS, Suryanarayanan TS (2020) Endolichenic fungal diversity associated with some lichens of the Western Ghats. Planta Med 86(13/14):960–966. https://doi.org/10.1055/a-1045-1989
Govindarajulu MB, Thirunavukkarasu N, Babu AG, Aggarwal A, Suryanarayanan TS, Reddy MS (2013) Endophytic Xylariaceae from the forests of Western Ghats, southern India: distribution and biological activities. Mycology 4(1):29–37
Suryanarayanan TS, Govindarajulu MB, Rajamani T, Tripathi M, Joshi Y (2017) Endolichenic fungi in lichens of Champawat district, Uttarakhand, northern India. Mycol Prog 16:205–211. https://doi.org/10.1007/s11557-016-1268-7
Konta S, Hyde KD, Phookamsak R, Xu JC, Maharachchikumbura SSN, Daranagama DA, Lu YZ (2020) Polyphyletic genera in Xylariaceae (Xylariales): Neoxylaria gen. nov. and Stilbohypoxylon. Mycosphere 11(1):2629–2651
Maha A, Rukachaisirikul V, Phongpaichit S, Poonsuwan W, Sakayaroj J (2016) Dimeric chromanone, cyclohexenone and benzamide derivatives from the endophytic fungus Xylaria sp. PSU-H182. Tetrahedron 72(22):2874–2879
Wendt L, Sir EB, Kuhnert E, Heitkämper S, Lambert C, Hladki AI, Stadler M (2018) Resurrection and emendation of the Hypoxylaceae, recognised from a multigene phylogeny of the Xylariales. Mycol Prog 17:115–154
Greenhalgh GN, Chesters CGC (1968) Conidiophore morphology in some British members of the Xylariaceae. Trans Brit Mycol Soc 51(1):57-IN6
Tang AMC, Jeewon R, Hyde KD (2009) A re-evaluation of the evolutionary relationships within the Xylariaceae based on ribosomal and protein-coding gene sequences. Fungal Divers 34(1):127–155
Acknowledgements
We acknowledge Dr. Sanjeeva Nayaka, Principal scientist, CSIR-National Botanical Research Institute, for his assistance during lichen identification. We thanks Dr. K. Manjunath lecturer, Department of PG Studies and Research in Applied Botany, Kuvempu University, Shankaraghatta and reviewer molecular biology reports for providing insightful comments on an earlier version of this manuscript. We extend our thank to Barcode Biosciences Private Limited Bengaluru for providing the sequencing service.
Funding
This work was financially supported by University Grant Commission, India under NET JRF program (Reference No: 201610056738 dated: 4/2/202).
Author information
Authors and Affiliations
Contributions
DP generated, analyses the data and prepare draft, YLK prepare the outline of the study, analyze the data, and finalized the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pushpavathi, D., Krishnamurthy, Y.L. Study on endolichenic fungal assemblage in Parmotrema and Heterodermia lichens of Shivamoga, Karnataka. Mol Biol Rep 51, 549 (2024). https://doi.org/10.1007/s11033-024-09497-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09497-3