Abstract
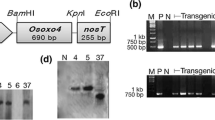
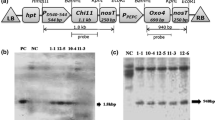
Osmotin, a pathogenesis related (PR) protein belonging to PR-5 protein family is induced in plants exposed to various biotic and abiotic stresses. To evaluate the potential involvement of Osmotin in improving resistance to early blight of potato, transgenic lines of an important processing grade potato cultivar ‘Kufri Chipsona 1’ were developed. The integration of T-DNA was confirmed by amplification of 900 bp DNA fragment of Osmotin (OsmWS) gene. The regenerated microshoots were subjected to GUS assay and eight independent putative transgenic lines showing uniform expression of reporter gene were selected for further assessment. The selected lines (L1–L8) were hardened and transferred to the containment facility. The transgenic plants inoculated with spore suspension of Alternaria solani over-expressed OsmWS gene by about 22 folds after three days. The disease severity also decreased in the transgenic lines as the lesion diameter was significantly reduced in comparison to control plants after 8 days of inoculation. Although, variation in disease severity among the transgenic lines was observed, yet, all the transgenic lines over-expressing OsmWS gene showed decreased disease severity and disease incidence. As a measure of plant health, total chlorophyll content and osmotic potential were also measured. Following infection, higher chlorophyll content (160.2 mg/g FW to 252.32 mg/g FW) was recorded in the transgenic lines in comparison with control plants (114.48 mg/g FW). However, an inverse variation was observed in terms of osmotic potential, where infection causes a significant decrease in osmotic potential in some of the transgenic lines. Thus it seems that over-expression of OsmWS gene in potato have potential to confer certain degree of resistance to early blight.
Key message
Induced over-expression of Osmotin gene cloned from Withania somnifera increased resistance to early blight in Indian potato cultivar ‘Kufri Chipsona 1’.






Similar content being viewed by others
References
Abad LR, D’Urzo MP, Liu D, Narasimhan ML, Reuveni M, Zhu JK, Niu X, Singh NK, Hasegawa PM, Bressan RA (1996) Antifungal activity of tobacco osmotin has specificity and involves plasma membrane permeabilization. Plant Sci 118:11–23. https://doi.org/10.1016/0168-9452(96)04420-2
Ahmed NU, Park JJ, Jung HJ, Kang KK, Lim YP, Hur YK, Nou IS (2013) Molecular characterization of thaumatin family genes related to stresses in Brassica rapa. Sci Hortic 152:26–34. https://doi.org/10.1016/j.scienta.2013.01.007
Ahn IP, Kim S, Kang S, Suh SC, Lee YH (2005) Rice defense mechanisms against Cochliobolus miyabeanus and Magnaporthe grisea are distinct. Phytopathology 95:1248–1255. https://doi.org/10.1094/PHYTO-95-1248
Ali GS, Hu X, Reddy ASN (2018) Overexpression of the Arabidopsis thaumatin-like protein 1 in transgenic potato plants enhances resistance against early and late blights. BioRxiv. https://doi.org/10.1101/621649
Andersen CL, Jensen JL, Orntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250. https://doi.org/10.1158/0008-5472.CAN-04-0496
Ansari MA, Butt TM (2011) Effects of successive subculturing on stability, virulence, conidial yield, germination and shelf-life of entomopathogenic fungi. J Appl Microbiol 110:1460–1469. https://doi.org/10.1111/j.1365-2672.2011.04994.x
Arnon DI (1949) Copper enzymes in isolated chloroplasts—polyphenol oxidases in Beta cularis. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Aung K, Jiang Y, He SY (2018) The role of water in plant-microbe interactions. Plant J 93:771–780. https://doi.org/10.1111/tpj.13795
Bolton M (2009) Primary metabolism and plant defense—fuel for the fire. Mol Plant Microbe Interact 22:487–497. https://doi.org/10.1094/MPMI-22-5-0487
Casado-Vela J, Selles S, Martinez RB (2006) Proteomic analysis of tobacco mosaic virus-infected tomato (Lycopersicon esculentum M.) fruits and detection of viral coat protein. Proteomics 6(Suppl 1):S196–S206. https://doi.org/10.1002/pmic.200500317
Chen WP, Chen PD, Liu DJ, Kynast R, Friebe B, Velazhahan R, Muthukrishnan S, Gill BS (1999) Development of wheat scab symptoms is delayed in transgenic wheat plants that constitutively express a rice thaumatin-like protein gene. Theor Appl Genet 99:755–760. https://doi.org/10.1007/s001220051294
Choi DS, Hong JK, Hwang BK (2013) Pepper Osmotin-like protein 1 (CaOSM1) is an essential component for defense response, cell death, and oxidative burst in plants. Planta 238:1113–1124. https://doi.org/10.1007/s00425-013-1956-3
Chowdhury S, Basu A, Kundu S (2015) Cloning, characterization and bacterial over-expression of an osmotin-Like protein gene from Solanum nigrum L. with antifungal activity against three necrotrophic fungi. Mol Biotechnol 57:371–381. https://doi.org/10.1007/s12033-014-9831-4
Chowdhury S, Basu A, Kundu S (2017) Overexpression of a new osmotin-like protein gene (SindOLP) confers tolerance against biotic and abiotic stresses in sesame. Front Plant Sci 8:410. https://doi.org/10.3389/fpls.2017.00410
Coaker GL, Willard B, Kinter M, Stockinger EJ, Francis DM (2004) Proteomic analysis of resistance mediated by Rcm 2.0 and Rcm 5.1, two loci controlling resistance to bacterial canker of tomato. Mol Plant-Microbe Interact 17:1019–1028. https://doi.org/10.1094/MPMI.2004.17.9.1019
Cohen E (2001) Chitin synthesis and inhibition: a revisit. Pest Manag Sci 57:946–950. https://doi.org/10.1002/ps.363
Cornelissen BJC, van HooftHuijsduijnen RAM, Bol JF (1986) A tobacco mosaic virus-induced protein is homologous to the sweet-tasting protein thaumatin. Nature 321:531–532. https://doi.org/10.1038/321531a0
Datta K, Velazhahan R, Oliva N, Ona I, Mew T, Khush GS, Mew T, Khush GS, Muthukrishnan S, Datta SK (1999) Over-expression of the cloned rice thaumatin-like protein (PR-5) gene in transgenic rice plants enhances environmental friendly resistance to Rhizoctonia solani causing sheath blight disease. Theor Appl Genet 98:1138–1145. https://doi.org/10.1007/s001220051178
Dey S, Chakraborty A (2012) Verital reaction against early blight of potato in planes of West Bengal. J Crop Weed 8:181–183
Dolar FS, Tenuta A, Higgins VJ (1994) Detached leaf assay for screening chickpea for resistance to ascochyta blight. Can J Plant Pathol 16:215–220. https://doi.org/10.1080/07060669409500756
Dufera JT (2014) Field, greenhouse and detached-leaf evaluation of tomato (Lycopersicon esculentum Mill.) genotypes for late blight resistance. WJAS 10:76–80. https://doi.org/10.5829/idosi.wjas.2014.10.2.1242
EI-Argawy E, Adss IA, Boyoumi SR (2017) Differential expression of induced resistance genes by abscisic acid (ABE) against potato early blight disease. Potato J 44:16–27
Elvira MI, Galdeano MM, Gilardi P, García-Luque I, Serra MT (2008) Proteomic analysis of pathogenesis related pro-tein (PRs) induced by compatible and incompatible interactions of pepper mild mottle virus (PMMoV) in Capsicum chinense L3 plants. J Exp Bot 59:1253–1265. https://doi.org/10.1093/jxb/ern032
Gao S, Gao J, Zhu X, Song Y, Li Z, Ren G, Zhou X, Kuai B (2016) ABF2, ABF3, and ABF4 promote ABA-mediated chlorophyll degradation and leaf senescence by transcriptional activation of chlorophyll catabolic genes and senescence associated genes in Arabidopsis. Mol Plant 9:1272–1285. https://doi.org/10.1016/j.molp.2016.06.006
Garyali S, Kumar A, Reddy MS (2013) Taxol Production by an Endophytic Fungus, Fusarium redolens, Isolated from Himalayan Yew. J Microbiol Biotechnol 23:1372–1380. https://doi.org/10.4014/jmb.1305.05070
Gherbawy YA (2005) Genetic variation among isolates of Alternaria spp. From selected Egyptian crops. Arch Phytopath Plant Protec 38:77–89. https://doi.org/10.1080/03235400400027390
Goth RW, Keane J (1997) A detached-leaf method to evaluate late blight resistance in potato and tomato. Am Potato J 74:347–352. https://doi.org/10.1007/BF02851579
Grenier J, Potvin C, Trudel J, Asselin A (1999) Some thaumatin-like proteins hydrolyse polymeric beta-1,3-glucans. Plant J 19:473–480. https://doi.org/10.1046/j.1365-313x.1999.00551.x
Hein I, Gilroy EM, Armstrong MR, Birch PRJ (2009) The zig-zag-zig in oomycete-plant interactions. Mol Plant Pathol 10:547–562. https://doi.org/10.1111/j.1364-3703.2009.00547.x
Holsters M, De Waele D, Depicker A, Messens E, Montagu MV, Schell J (1978) Transfection and transformation of Agrobacterium tumefaciens. Mol Gen Genet 163:181–187. https://doi.org/10.1007/BF00267408
Hon WC, Griffith M, Mlynarz A, Kwok YC, Yang DSC (1995) Antifreeze proteins in winter rye are similar to pathogenesis-related proteins. Plant Physiol 109:879–889. https://doi.org/10.1104/pp.109.3.879
Hood EE, Gelvin SB, Melchers LS, Hoekema A (1993) New Agrobacterium helper plasmids for gene transfer to plants. Transgenic Res 2:208–218. https://doi.org/10.1007/BF01977351
Houterman PM, Speijer D, Dekker HL, DE Koster CG, Cornelissen BJ, Rep M (2007) The mixed xylems approteome of Fusarium oxysporum-infected tomato plants. Mol Plant Pathol 8:215–221. https://doi.org/10.1111/j.1364-3703.2007.00384.x
Hu X, Reddy ASN (1997) Cloning and expression of a PR5 like protein from Arabidopsis: inhibition of fungal growth by bacterially expressed protein. Plant Mol Biol 34:949 –949 59. https://doi.org/10.1023/a:1005893119263()
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Katam R, Chibanguza K, Latinwo LM, Smith D (2015) Proteome biomarkers in xylem reveal pierce’s disease tolerance in grape. J Proteomics Bioinform 8:217–224. https://doi.org/10.4172/jpb.1000372
Kaur A, Reddy MS, Kumar A (2017) Efficient, one step and cultivar independent shoot organogenesis of potato. Physiol Mol Biol Plants 23:461–469. https://doi.org/10.1007/s12298-017-0418-y
Kaur A, Guleria S, Reddy MS, Kumar A (2020) A robust genetic transformation protocol to obtain transgenic shoots of Solanum tuberosum L. cultivar ‘Kufri Chipsona 1’. Physiol Mol Biol Plants 26:367–377. https://doi.org/10.1007/s12298-019-0074-7-4
Khan RS, Sjahril R, Nakamura I, Mii M (2008) Production of transgenic potato exhibiting enhanced resistance to fungal infections and herbicide applications. Plant Biotechnol Rep 2:13–20. https://doi.org/10.1007/s11816-008-0043-x
Khan A, Ahmad I, Shah A (2013) Amelioration of salinity stress in wheat (Triticum aestivum L) by foliar application of phosphorus. FYTON 82:281–287
Kim ST, Kim SG, Hwang DH, Kang SY, Kim HJ, Lee BH, Lee JJ, Kang KY (2004) Proteomic analysis of pathogen-responsive proteins from rice leaves induced by rice blast fungus, Magnaporthe grisea. Proteomics 4:3569–3578. https://doi.org/10.1002/pmic.200400999
Koiwa H, Kato H, Nakatsu T, Oda J, Yamada Y, Sato F (1997) Purification and characterisation of tobacco pathogenesis-related protein PR-5d, an anti-fungal thaumatin-like protein. Plant Cell Physiol 38:783–791. https://doi.org/10.1093/oxfordjournals.pcp.a029236
Kumar A, Palni LMS, Nandi SK (2003) The effect of light source and gelling agent on micropropagation of Rosa damascene Mill. and Rhychostylis retusa (L.) Bl. J Hortic Sci Biotechnol 78:786–792. https://doi.org/10.1080/14620316.2003.11511700
Lamb CJ, Ryals JA, Ward ER, Dixon RA (1992) Emerging strategies for enhancing crop resistance to microbial pathogens. Biotechnology 10:1436–1445. https://doi.org/10.1038/nbt1192-1436
Lei Y, Zhang ZY, Liu X, Wang WF, An XM, Lin SZ, Zheng HQ (2012) Cloning and expression of three pathogenesis related protein genes from Populus tomeutosa. J Northeast Agric Univ 40:67–75
Li X-Y, Gao L, Zhang W-H, Liu J-K, Zhang Y-J, Wang H-Y, Liu D-Q (2015) Characteristic expression of wheat PR5 gene in response to infection by the leaf rust pathogen, Puccinia triticina. J Plant Interact 10:132–141. https://doi.org/10.1080/17429145.2015.1036140
Liu D, Rhodes D, D’Urzo P, Xu Y, Narasimhan ML, Hasegarua PM, Bressan RA, Abad L (1996) In vivo and in vitro activity of truncated Osmotin that is secreated into the extracellular matrix. Plant Sci 121:123–131.
Mackintosh CA, Lewis J, Radmer LE, Shin S, Heinen SJ, Smith LA, Wyckoff MN, Dill-Macky R, Evans CK, Kravchenko S, Baldridge GD, Zeyen RJ, Muehlbauer GJ (2007) Overexpression of defense response genes in transgenic wheat enhances resistance to Fusarium head blight. Plant Cell Rep 26:479–488. https://doi.org/10.1007/s00299-006-0265-8
Mahmood T, Jan A, Kakishima M, Komatsu S (2006) Proteomic analysis of bacterial-blight defence-responsive proteins in rice leaf blades. Proteomics 6:6053–6065. https://doi.org/10.1002/pmic.200600470
Malcolmson JF, Black W (1966) New R genes in Solanum demissum Lindl. and their complementary races of Phytophthora infestans (Mont.) de Bary. Euphytica 15:199–203. https://doi.org/10.1007/BF00022324
Meena M, Swapnil P, Upadhyay RS (2017) Isolation, characterization and toxicological potential of Alternaria-mycotoxins (TeA, AOH and AME) in different Alternaria species from various regions of India. Sci Rep 7:8777. https://doi.org/10.1038/s41598-017-09138-9
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Namukwaya B (2015) Evaluation of transgenic bananas expressing anti-apoptotic genes for resistance against fusarium wilt. Ph.D. Thesis, Centre for tropical crops and biocommodities, Queensland University of Technology, Australia
Nielsen KK, Mikkelsen JD, Kragh KM, Bojsen K (1993) An acidic class III chitinase in sugar beet induced by cercospora beticola: characterization and expression in transgenic tobacco plants. Mol Plant-Microbe Interact 6:495–506. https://doi.org/10.1094/mpmi-6-495
Pandey AK, Reddy MS, Suryanarayanan TS (2003) ITS-RFLP and ITS sequence analysis of a foliar endophytic Phyllosticta from different tropical trees. Mycol Res 107:439–444. https://doi.org/10.1017/S0953756203007494
Pandey P, Irulappan V, Bagavathiannan MV, Senthil-Kumar M (2017) Impact of combined abiotic and biotic stresses on plant growth and avenues for crop improvement by exploiting physio-morphological traits. Front Plant Sci 8:537. https://doi.org/10.3389/fpls.2017.00537
Pourazari F, Andersson M, Weih M (2018) Altered tuber yield in genetically modified high-amylose and oil potato lines is associated with changed whole-plant nitrogen economy. Front Plant Sci 9:342. https://doi.org/10.3389/fpls.2018.00342
Rosyara UR, Duveiller E, Pant K, Sharma RC (2007) Variation in chlorophyll content, anatomical traits and agronomic performance of wheat genotypes differing in spot blotch resistance under natural epiphytotic conditions. Aust Plant Pathol 36:245–251. https://doi.org/10.1071/AP07014
Singh V (2017) Biotechnological interventions against leaf spot disease of Withania somnifera (L.) Dunal. Ph.D. Thesis, Guru Nanak Dev University, Amritsar
Stintzi A, Heitz T, Kauffman S, Legrand M, Fritig B (1991) Identification of a basic pathogenesis related, thau-matin like protein of virus infected tobacco as osmotin. Physiol Mol Plant Pathol 38:137–146. https://doi.org/10.1016/S0885-5765(05)80131-6
Trudel J, Grenier J, Potvin C, Asselin A (1998) Several thaumatin like proteins bind to beta 1,3 glucans. Plant Physiol. https://doi.org/10.1104/pp.118.4.1431
van der Vossen E, Sikkema A, Hekkert Bt, Gros J, Stevens P, Muskens M, Wouters D, Pereira A, Stiekema W, Allefs S (2003) An ancient R gene from the wild potato species Solanum bulbocastanum confers broad-spectrum resistance to Phytophthora infestans in cultivated potato and tomato. Plant J 36:867–882. https://doi.org/10.1046/j.1365-313x.2003.01934.x
van der Vossen EAG, Gros J, Sikkema A, Muskens M, Wouters D, Wolters P, Pereira A, Allefs S (2005) The Rpi-blb2 gene from Solanum bulbocastanum is an Mi-1 gene homolog conferring broad-spectrum late blight resistance in potato. Plant J 44:208–222. https://doi.org/10.1111/j.1365-313X.2005.02527.x
Van der Walls JE, Korsten L, Slippers B (2004) Genetic diversity among Alternaria solani isolates from potatoes in South Africa. Plant Dis 88:959–964. https://doi.org/10.1094/PDIS.2004.88.9.959
Van Loon LC (1985) Pathogenesis-related proteins. Plant Mol Biol 4:111–116. https://doi.org/10.1007/BF02418757
Van Loon LC, Van-Strein EA (1999) The families of pathogenesis-related proteins, their activities,and comparative analysis of PR-1 type proteins. Physiol Mol Plant Pathol 55:85–97. https://doi.org/10.1006/pmpp.1999.0213
Velazhahan R, Datta SK, Muthukrishnan S (1999) The PR-5 family: thaumatin-like proteins in plants. In: Datta SK, Muthukrishnan S (eds) Pathogenesis-related proteins in plants. CRC Press, Boca Raton, pp 107–129
Vigers AJ, Wiedemann S, Roberts WK, Legrand M, Claude PS, Fritig B (1992) Thaumatin-like pathogenesis-related proteins are antifungal. Plant Sci 83:155–161. https://doi.org/10.1016/0168-9452(92)90074-V
Wanderley-Nogueira AC, Belarmino LC, Soares-Cavalcanti Nda M, Bezerra-Neto JP, Kido EA, Pandolfi V, Abdelnoor RV, Binneck E, Carazzole MF, Benko-Iseppon AM (2012) An overall evaluation of the Resistance (R) and Pathogenesis-Related (PR) superfamilies in soybean, as compared with Medicago and Arabidopsis. Genet Mol Biol 35:260–271. https://doi.org/10.1590/S1415-47572012000200007
Wang L, Yang L, Zhang JX, Dong J, Yu J, Zhou J, Zhuge Q (2013) Cloning and characterization of a thaumatin-like protein gene PeTLP in Populus deltoids X P. euramericana cv. ‘Nanlin895.’ Acta Physiol Plant 35:2985–2998. https://doi.org/10.1007/s11738-013-1330-x)
Weber RL, Wiebke-Strohm B, Bredemeier C, Margis-Pinheiro M, de Brito GG, Rechenmacher C, Bertagnolli PF, de Sa ME, Campos Mde A, de Amorim RM, Beneventi MA, Margis R, Grossi-de-Sa MF, Bodanese-Zanettini MH (2014) Expression of an osmotin-like protein from Solanum nigrum confers drought tolerance in transgenic soybean. BMC Plant Biol 14:343. https://doi.org/10.1186/s12870-014-0343-y
Woloshuk CP, Muelenhoff JS, Sela-Buurlage M, van den Elzen PJ, Cornelissen BJ (1991) Pathogen induced proteins with inhibitory activity towards Phytophthora infestans. Plant Cell 3:619–628. https://doi.org/10.1105/tpc.3.6.619
Yang F, Li W, Derbyshire M, Larsen MR, Rudd JJ, Palmisano G (2015) Unraveling incompatibility between wheat and the fungal pathogen Zymoseptoria tritici through apoplastic proteomics. BMC Genomics 16:362–374. https://doi.org/10.1186/s12864-015-1549-6
Yang L, Wang D, Xu Y, Zhao H, Wang L, Cao X, Chen Y, Chen Q (2017) A new resistance gene against potato late blight originating from Solanum pinnatisectum located on potato Chromosome 7. Front Plant Sci 8:1729. https://doi.org/10.3389/fpls.2017.01729
Acknowledgements
Amanpreet Kaur is thankful to University Grants Commission (UGC), New Delhi for the award of Maulana Azad National Fellowship for minority students. TIFAC-CORE is thanked for the facilities to carry out research work.
Author information
Authors and Affiliations
Contributions
AK conducted the experiments, compiled data, analyzed the results and wrote the initial draft of the manuscript, AK and AK conceived and designed the experiments, PKP provided the vector carrying Osmotin gene, MSR helped in statistical analysis and molecular studies, AK finalized the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Henryk Flachowsky.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kaur, A., Sudhakara Reddy, M., Pati, P.K. et al. Over-expression of Osmotin (OsmWS) gene of Withania somnifera in potato cultivar ‘Kufri Chipsona 1’ imparts resistance to Alternaria solani. Plant Cell Tiss Organ Cult 142, 131–142 (2020). https://doi.org/10.1007/s11240-020-01847-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01847-w