Abstract
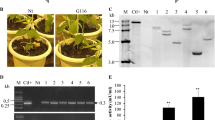
Tomatoes are an essential part of the human diet worldwide, and their production has been restricted by fungal disease. One of the most common fungal diseases of Lycopersicon esculentum L. is Sclerotinia stem rot, caused by Sclerotinia sclerotiorum. In this work, overexpression of the chimeric chit42 gene from Trichoderma atroviride with Serratia marcescens Chitinase B C-terminal fused chitin-binding domain (ChBD) in tomato was reported. Thirty independent transgenic lines were regenerated and adapted to greenhouse conditions. Five transgenic lines was selected for subsequent molecular and biological analysis. RT-PCR analysis of transgenic plants showed different expression patterns in independent transgenic events. Young leaves of T1 plants challenged with S. sclerotiorum revealed that expression of chimeric chitinase enhanced plant resistance against sclerotinia stem rot. A radial diffusion assay showed that transgenic lines with constitutive expression of the ChBD gene inhibited S. sclerotiorum growth significantly. Lesion sizes of transgenic tomatoes caused by S. sclerotiorum were significantly reduced compared to non-transgenic tomato plants. This is the first study reporting the evaluation of transgenic lines of tomato harboring Trichoderma chitinase (chit42) with chitin binding domain (ChBD) gene for resistance to one of the most important fungal diseases, S. sclerotiorum.
Key message
The chimeric chit42 gene with the Serratia marcescens chitinase binding domain has been expressed in tomato. The resistance of transgenic tomatoes to Sclerotinia stem rot has increased.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Abbas T, Zahir ZA, Naveed M, Kremer RJ (2018) Limitations of existing weed control practices necessitate development of alternative techniques based on biological approaches. Adv Agron 147:239–280. https://doi.org/10.1016/bs.agron.2017.10.005
Aghazadeh R, Zamani M, Motallebi M, Moradyar M (2016) Co-transformation of canola by chimeric chitinase and tlp genes towards improving resistance to Sclerotinia sclerotiorum. World J Microbiol Biotechnol 32(9):144. https://doi.org/10.1007/s11274-016-2104-6
Ayers AR, Ebel J, Valent B, Albersheim P (1976) Host–pathogen interactions: X. Fractionation and biological activity of an elicitor isolated from the mycelial walls of Phytophthora megasperma var. sojae. Plant Physiol 57:760–765. https://doi.org/10.1104/pp.57.5.760
Badrhadad A, Nazarian F, Ismaili A (2018) Fusion of a chitin-binding domain to an antibacterial peptide to enhance resistance to Fusarium solani in tobacco (Nicotiana tabacum). 3 Biotech 8:391. https://doi.org/10.1007/s13205-018-1416-7
Berini F, Casartelli M, Montali A, Reguzzoni M, Tettamanti G, Marinelli F (2019) Metagenome-sourced microbial chitinases as potential insecticide proteins. Front Microbiol 18(10):1358. https://doi.org/10.3389/fmicb.2019.01358
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Broekaert WF, Marien W, Terras F, Bolle M, Proost P, Damme J, Dillen L, Claeys M, Rees S (1992) Antimicrobial peptides from Amaranthus caudatus seeds with sequence homology to the cysteine/glycinerich domain of chitin-binding proteins. Biochemistry 31:4308–4314. https://doi.org/10.1021/bi00132a023
Chanda B, Shamimuzzaman M, Gilliard A, Ling KS (2021) Effectiveness of disinfectants against the spread of tobamoviruses: Tomato brown rugose fruit virus and Cucumber green mottle mosaic virus. Virol J 18(1):1–2. https://doi.org/10.1186/s12985-020-01479-8
Chhikara S, Chaudhury D, Dhankher OP, Jaiwal PK (2012) Combined expression of a barley class II chitinase and type I ribosome inactivating protein in transgenic Brassica juncea provides protection against Alternaria brassicae. Plant Cell Tissue Organ Cult 108:83–89. https://doi.org/10.1007/s11240-011-0015-7
Cunha WG, Tinoco MLP, Pancoti HL, Ribeiro RE, Aragão FJL (2010) High resistance to Sclerotinia sclerotiorum in transgenic soybean plants transformed to express an oxalate decarboxylase gene. Plant Pathol 59:654–660. https://doi.org/10.1111/j.1365-3059.2010.02279.x
da Silva Santos SD, da Silva AA, Polonio JC, Polli AD, Orlandelli RC, dos Santos Oliveira JA, Brandão Filho JU, Azevedo JL, Pamphile JA (2022) Influence of plant growth-promoting endophytes Colletotrichum siamense and Diaporthe masirevici on tomato plants (Lycopersicon esculentum Mill.). Mycology 18:1–4. https://doi.org/10.1080/21501203.2022.2050825
Dana M, Pintor-Toro T, Cubero B (2006) Transgenic tobacco plants overexpressing chitinases of fungal origin show enhanced resistance to biotic and abiotic stress agents. Plant Physiol 142:722–730. https://doi.org/10.1104/pp.106.086140
Dowd PF, Naumann TA, Price NP, Johnson ET (2018) Identification of a maize (Zea mays) chitinase allele sequence suitable for a role in ear rot fungal resistance. Agri Gene 7:15–22. https://doi.org/10.1016/j.aggene.2017.10.001
El-Sobki AE, Ali AA (2020) Biochemical effects of some chitin synthesis inhibitors against red palm weevil, Rhynchophorus ferrugineus insect. Egypt Acad J Biol Sci F Toxicol Pest Control 12(1):127–39. https://doi.org/10.21608/eajbsf.2020.83542
Eslahi N, Kowsari M, Zamania MR, Motallebia M (2021) The profile change of defense pathways in Phaseouls vulgaris L. by biochemical and molecular interactions of Trichoderma harzianum transformants overexpressing a chimeric chitinase. Biol Control 152:104304. https://doi.org/10.1016/j.biocontrol.2020.104304
Ghorbanpour M, Omidvari M, Abbaszadeh-Dahaji P, Omidvar R, Kariman K (2018) Mechanisms underlying the protective effects of beneficial fungi against plant diseases. Biol Control 117:147–157. https://doi.org/10.1016/j.biocontrol.2017.11.006
Girhepuje PV, Shinde GB (2011) Transgenic tomato plants expressing a wheat endochitinase gene demonstrate enhanced resistance to Fusarium oxysporum f. sp. lycopersici. Plant Cell Tissue Organ Cult 105:243–251. https://doi.org/10.1007/s11240-010-9859-5
Hou J, Li X, Kaczmarek MB, Chen P, Li K, Jin P, Liang Y, Daroch M (2019) Accelerated CO2 hydration with thermostable sulfurihydrogenibium azorense carbonic anhydrase-chitin binding domain fusion protein immobilised on chitin support. Int J Mol Sci 20(6):1494. https://doi.org/10.3390/ijms20061494
Huang JK, Ma PL, Ji SY, Zhao XL, Tan JX, Sun XJ, Huang FD (2013) Age-dependent alterations in the presynaptic active zone in a Drosophila model of Alzheimer’s disease. Neurobiol Dis 51:161–167. https://doi.org/10.1016/j.nbd.2012.11.006
Jambhulkar PP, Sharma P, Manokaran R, Lakshman DK, Rokadia P, Jambhulkar N (2018) Assessing synergism of combined applications of Trichoderma harzianum and Pseudomonas fluorescens to control blast and bacterial leaf blight of rice. Eur J Plant Pathol 152(3):747–757. https://doi.org/10.1007/s10658-018-1519-3
Karmakar S, Molla KA, Chanda PK, Sarkar SN, Datta SK, Datta K (2016) Green tissue-specific co-expression of chitinase and oxalate oxidase 4 genes in rice for enhanced resistance against sheath blight. Planta 243(1):115–130. https://doi.org/10.1007/s00425-015-2398-x
Khan RS, Darwish NA, Khattak B, Ntui VO, Kong K, Shimomae K, Nakamura I, Mii M (2014) Retransformation of marker-free potato for enhanced resistance against fungal pathogens by pyramiding chitinase and wasabi defensin genes. Mol Biotechnol 56(9):814–823. https://doi.org/10.1007/s12033-014-9760-2
Kowsari M, Zamani MR, Motallebi M (2014a) Enhancement of Trichoderma harzianum activity against Sclerotinia sclerotiorum by overexpression of Chit42. Iran J Biotechnol 12(2):e13869
Kowsari M, Motallebi M, Zamani MR (2014b) Protein engineering of chit42 towards improvement of chitinase and antifungal activities. Curr Microbiol 68:495–502. https://doi.org/10.1007/s00284-013-0494-3
Li YH, Zhang XY, Zhang F, Peng LC, Zhang DB, Kondo A, Zhao XQ (2018) Optimization of cellulolytic enzyme components through engineering Trichoderma reesei and on-site fermentation using the soluble inducer for cellulosic ethanol production from corn stover. Biotechnol Biofuels 11:49. https://doi.org/10.1186/s13068-018-1048-5
Litvak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Loc NH, Huy ND, Quang HT, Lan TT, Ha TT (2020) Characterisation and antifungal activity of extracellular chitinase from a biocontrol fungus, Trichoderma asperellum PQ34. Mycology 11(1):38–48. https://doi.org/10.1080/21501203.2019.1703839
Malik MS, Haider S, Rehman A, Rehman SU, Jamil M, Naz I, Anees M (2022) Biological control of fungal pathogens of tomato (Lycopersicon esculentum) by chitinolytic bacterial strains. J Basic Microbiol 62(1):48–62. https://doi.org/10.1002/jobm.202100512
Marra R, Lombardi N, Piccolo A, Bazghaleh N, Prashar P, Vandenberg A, Woo S (2021) Mineral biofortification and growth stimulation of lentil plants inoculated with Trichoderma strains and metabolites. Microorganisms 10(1):87. https://doi.org/10.3390/microorganisms10010087
Matroodi S, Motallebi M, Zamani MR, Moradyar M (2013) Designing a new chitinase with more chitin binding and antifungal activity. World J Microbiol Biotechnol 29:1517–1523. https://doi.org/10.1007/s11274-013-1318-0
Mazumdar P (2021) Sclerotinia stem rot in tomato: a review on biology, pathogenicity, disease management and future research priorities. J Plant Dis Prot 128(6):1403–1431. https://doi.org/10.1007/s41348-021-00509-z
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nalluri N, Karri VR (2020) Recent advances in genetic manipulation of crops: a promising approach to address the global food and industrial applications. Plant Sci Today 7(1):70–92. https://doi.org/10.14719/pst.2020.7.1.659
Nuwamanya AM, Runo S, Mwangi M (2022) In-vitro sensitivity of Alternaria solani isolates to azoxystrobin and difenoconazole fungicides in Kenya and detection of Cyt b mutations associated with azoxystrobin resistance. Crop Prot 30:106010. https://doi.org/10.1016/j.cropro.2022.106010
Ojaghian S, Wang L, Xie GL, Zhang JZ (2018) Increased resistance against storage rot in transgenic carrots expressing chitinase chit42 from Trichoderma harzianum. Sci Hortic 234:81–86. https://doi.org/10.1016/j.scienta.2018.02.025
Olowe OM, Nicola L, Asemoloye MD, Akanmu AO, Babalola OO (2022) Trichoderma: Potential bio-resource for the management of tomato root rot diseases in Africa. Microbiol Res 2:126978. https://doi.org/10.1016/j.micres.2022.126978
Richa K, Tiwari IM, Devanna BN, Botella JR, Sharma V, Sharma TR (2017) Novel CHITINASE Gene LOC_Os11g47510 from indica rice tetep provides enhanced resistance against sheath blight pathogen Rhizoctonia solani in rice. Front Plant Sci 8:596. https://doi.org/10.3389/fpls.2017.00596
Rogers SO, Bendich AJ (1985) Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Mol Biol 5:69–76. https://doi.org/10.1007/BF00020088
Salas CE, Badillo-Corona JA, Ramírez-Sotelo G, Oliver-Salvador C (2015) Biologically active and antimicrobial peptides from plants. Biomed Res Int 2015:1–11. https://doi.org/10.1155/2015/102129
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Santoso P, Minamihata K, Ishimine Y, Taniguchi H, Komada T, Sato R, Goto M, Takashima T, Taira T, Kamiya N (2022) Enhancement of the antifungal activity of chitinase by palmitoylation and the synergy of palmitoylated chitinase with amphotericin B. ACS Infect Dis. https://doi.org/10.1021/acsinfecdis.2c00052
Sharma S, Kumar S, Khajuria A, Ohri P, Kaur R, Kaur R (2020) Biocontrol potential of chitinases produced by newly isolated Chitinophaga sp. S167. World J Microbiol Biotechnol 36(6):1–5. https://doi.org/10.1007/s11274-020-02864-9
Solgi T, Moradyar M, Zamani MR, Motallebi M (2015) Transformation of canola by chit33 gene towards improving resistance to Sclerotinia sclerotiorum. Plant Prot Sci 51(1):6–12. https://doi.org/10.17221/83/2013-PPS
Tien NQ, Hoa PT, Tue NH, Thanh DV, Thi HA, Luong NN, Huy NX, Loc NH (2021) Transient expression of chi42 genes from Trichoderma asperellum in Nicotiana benthamiana by agroinfiltration. Int J Agric Biol 26:177–184. https://doi.org/10.17957/IJAB/15.1822
Tue NH, Tuong TG, Trang PT, Chung ND, Hoa PT, Tien NQ, Loc NH (2022) Cloning the root-specific Asy promoter and genes encoding chitinase 42 kDa of Trichoderma asperellum into the plant expression vector. J Appl Biol Biotechnol. https://doi.org/10.7324/JABB.2022.100302
Vaghela B, Vashi R, Rajput K, Joshi R (2022) Plant chitinases and their role in plant defense—a comprehensive review. Enzym Microb Technol 30:110055. https://doi.org/10.1016/j.enzmictec.2022.110055
Wang Z, Ma LM, Cao J, Li YL, Ding LN, Zhu KM, Yang YH, Tan ZL (2019) Recent advances in mechanisms of plant defense to Sclerotinia sclerotiorum. Front Plant Sci 10:1314. https://doi.org/10.3389/fpls.2019.01314
Wang F, Yang S, Wang Y, Zhang B, Zhang F, Xue H, Jiang Q, Ma Y (2021) Overexpression of chitinase gene enhances resistance to Colletotrichum gloeosporioides and Alternaria alternata in apple (Malus × domestica). Sci Hortic 277:109779. https://doi.org/10.1016/j.scienta.2020.109779
Xia H, Li YY, Liu ZC, Li YQ, Chen J (2018) Transgenic expression of chit42 gene from Metarhizium anisopliae in Trichoderma harzianum enhances antagonistic activity against Botrytis cinerea. Mol Biol 52(5):668–675. https://doi.org/10.1134/S002689331805014X
Yang X, Yang J, Wang Y, He H, Niu L, Guo D, Xing G, Zhao Q, Zhong X, Sui L, Li Q, Dong Y (2019) Enhanced resistance to sclerotinia stem rot in transgenic soybean that overexpresses a wheat oxalate oxidase. Transgenic Res 28(1):103–114. https://doi.org/10.1007/s11248-018-0106-x
Yang X, Yang J, Li H, LuNiu L, Xing G, Zhang Y, Xu W, Zhao Q, Li Q, Dong Y (2020) Overexpression of the chitinase gene CmCH1 from Coniothyrium minitans renders enhanced resistance to Sclerotinia sclerotiorum in soybean. Transgenic Res 29:187–198. https://doi.org/10.1007/s11248-020-00190-2
Zarinpanjeh N, Motallebi M, Zamani MR, Ziaie M (2016) Enhanced resistance to Sclerotinia sclerotiorum in Brassica napus by co-expression of defensin and chimeric chitinase genes. J Appl Genet 57:417–425. https://doi.org/10.1007/s13353-016-0340-y
Zeilinger S, Galhaup C, Payer K, Woo SL, Mach RL, Fekete C, Lorito M, Kubicek CP (1999) Chitinase gene expression during mycoparasitic interaction of Trichoderma harzianum with its host. Fungal Genet Biol 26:131–140. https://doi.org/10.1006/fgbi.1998.1111
Zhang F, Ge H, Zhang F, Guo N, Wang Y, Chen L, Li C (2016) Biocontrol potential of Trichoderma harzianum isolate T-aloe against Sclerotinia sclerotiorum in soybean. Plant Physiol Biochem 100:64–74. https://doi.org/10.1016/j.plaphy.2015.12.017
Zhang WY, Zhang SY, Chen XH, Qiao H, Jiang SF, Xiong YW, Jin SB, Gong YS, Fu HT (2018) Molecular cloning and expression analysis of MnCht1C gene from oriental river pawn (Macrobrachium nipponense). Genomics Appl Biol 37(2):723–732
Zhang Y, Chen W, Sang X, Wang T, Gong H, Zhao Y, Zhao P, Wang H (2021) Genome-wide identification of the thaumatin-like protein family genes in Gossypium barbadense and analysis of their responses to Verticillium dahliae infection. Plants 10(12):2647. https://doi.org/10.3390/plants10122647
Zhu M, Lu S, Zhuang M, Zhang Y, Lv H, Ji J, Hou X, Fang Z, Wang Y, Yang L (2021) Genome-wide identification and expression analysis of the Brassica oleracea L. chitin-binding genes and response to pathogens infections. Planta 253:80. https://doi.org/10.1007/s00425-021-03596-2
Acknowledgements
The authors thank to Dr. Mostafa Motallebi from National Institute of Genetic Engineering and Biotechnology (NIGEB) for provide the chimeric chitinase construct and the Mycology laboratory, Plant protection department of Tabriz University to provide the fungi and support us for the bioassay experiments.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Lab work have been done by VG by adviser of ED. ED conceived and designed the experiment. MZ help us in all bioassay analysis test and give the good advises in writing the manuscript. VG wrote the manuscript with support of ED. MV give the good and benefit advice in lab techniques and revised the manuscript finally. All author critical feedback and helped shape the research, analysis and manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Additional information
Communicated by Joyce Van Eck.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gougerdchi, V., Dorani, E., Valizadeh, M. et al. Overexpression of the chimeric chitinase (ChBD) gene in Lycopersicon esculentum L. enhanced resistance to Sclerotinia sclerotiorum. Plant Cell Tiss Organ Cult 151, 165–175 (2022). https://doi.org/10.1007/s11240-022-02340-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02340-2