Abstract
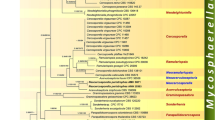
Cinereomyces clade is a newly proposed monophyletic group of polypores containing currently four genera and five species, including two promising biopulping fungi, Ceriporiopsis rivulosa and C. subvermispora. The Cinereomyces clade is well-delimited in nrDNA-based phylogenetic analysis, but its position in Polyporales remains unclear. Its closest relative may be found in the core polyporoid clade. Only a few morphological characters are common for all the species in the clade, e.g. CB– and CRB+ hyphae, white fruiting bodies, presence of oil, and middle-sized spores. Culturally, the species are unified by producing simple-septate generative hyhpae in the margin, which produce simple-clamped hyphae backwards. The genus Gelatoporia is the correct place for Ceriporiopsis subvermispora. Two new genera are described in the group: Obba to incorporate C. rivulosa and a new austral species, Obba valdiviana, known from southern Argentina and recorded here also from Tasmania, and Sebipora to accommodate a new species from tropical Asia, S. aquosa. ITS sequences imply that Eurasian Gelatoporia subvermispora may belong to a different species from the North American one. G. subvermispora is recorded as new to Indonesia.






Similar content being viewed by others
References
Akhtar M, Attridge MC, Blanchette RA, Myers GC, Wall MB, Sykes MS, Koning JW Jr, Burgess RR, Wegner TH, Kirk TK (1992) The white-rot fungus Ceriporiopsis subvermispora saves electrical energy and improves strength properties during biomechanical pulping of wood. In: Kuwahara M, Shimada M (eds) Biotechnology in pulp and paper industry. UNi, Tokyo, pp 3–8
Akhtar M, Scott GM, Swaney RE, Shipley DF (2000) Biomechanical pulping: a mill-scale evaluation. Resour Conserv Recycl 28:241–252
Amirta R, Tanabe T, Watanabe T, Honda Y, Kuwahara M, Watanabe T (2006) Methane fermentation of Japanese cedar wood pretreated with a white rot fungus, Ceriporiopsis subvermispora. J Biotech 123:71–77
Atik C, Imamoglu S, Bermek H (2006) Impact of xylanase pre-treatment on peroxide bleaching stage of biokraft pulp. Int Biodeter Biodegr 58:22–26
Bernicchia A (2005) Polyporaceae s.l. Edizione Candusso, Alassio
Binder M, Hibbett DS, Larsson KH, Larsson E, Langer E, Langer G (2005) The phylogenetic distribution of resupinate forms across the major clades of homobasidiomycetes. Syst Biodivers 3:113–157
Boidin J (1971) Nuclear behaviour in the mycelium and the evolution of the Basidiomycetes. In: Petersen RH (ed) Evolution in the higher Basidiomycetes. University of Tennessee Press, Knoxville, pp 129–148
Corner EJH (1992) Additional resupinate non-xanthochroic polypores from Brazil and Malesia. Nova Hedwigia 55:119–152
Dai YC (1998) Changbai wood-rotting fungi 9. Three new species and other species in Rigidoporus, Skeletocutis and Wolfiporia (Basidiomycota, Aphyllophorales). Ann Bot Fenn 35:143–154
Dai YC, Wang Z, Binder M, Hibbett DS (2006) Phylogeny and a new species of Sparassis (Polyporales, Basidiomycota): evidence from mitochondrial atp6, nuclear rDNA and rpb2 genes. Mycologia 98:584–592
David A (1971) Caractères myceliens de Rigidoporus rivulosus (Berk. et Curt.) comb. nov., espèce nouvelle pour l'Europe. Bull Soc Mycol Fr 87:415–419
David A (1972) Comportement nucléaire des représentants du genre Gloeoporus Mont.: G. conchoides Mont., G. dichrous (Fr.) Bres. et G. pannocinctus (Rom.) J. Erikss. récolté pour la première fois en France. Bull Soc Mycol Fr 88:209–213
David A (1982) Étude monographique du genre Skeletocutis (Polyporaceae). Naturaliste Can 109:235–272
David A, Déquatre B (1984) Deux “ultraespecies”: Antrodia malicola (Berk. & Curt.) Donk et A. ramentacea (Berk. & Br.) Donk (Basidiomycetes, Aphyllophorales). Cryptogam Mycol 5:293–300
David A, Déquatre B (1985) Antrodia albidoides (Polyporaceae) nouvelle ultraspecies meridionale. Mycol Helv 6:357–369
Fackler K, Gradinger C, Schmutzer M, Tavzes C, Burgert I, Schwanninger M, Hinterstoisser B, Watanabe T, Messner K (2007) Biomodification of wood with selective white-rot fungi. Food Technol Biotechnol 45:269–276
Galkin A, Vares T, Kalsi M, Hatakka A (1998) Production of organic acids by different white-rot fungi as detected using capillary zone electrophoresis.Biotechnol Tech 12:267–271
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gilbertson RL, Ryvarden L (1986) North American polypores 1. Abortiporus to Lindtneria. Fungiflora, Oslo
Harreither W, Sygmund C, Dünhofen E, Vicuña R, Haltrich D, Ludwig R (2007) Cellobiose dehydrogenase from the ligninolytic basidiomycete Ceriporiopsis subvermispora. Appl Env Microbiol 75:2750–2757
Hakala TK, Lundell T, Galkin S, Maijala P, Kalkkinen N, Hatakka A (2005) Manganese peroxidases, laccases and oxalic acid from the selective white-rot fungus Physisporinus rivulosus grown on spruce wood chips. Enz Microb Technol 36:461–468
Hallenberg N (1984) Compatibility between species of Corticiaceae s.l. (Basidiomycetes) from Europe and North America. Mycotaxon 21:335–388
Hattori T (2001a) Type studies of the polypores described by E. J. H. Corner from Asia and West Pacific Areas II. Species described in Gloeophyllum, Heteroporus, Microporellus, Oxyporus, Paratrichaptum, and Rigidoporus. Mycoscience 42:19–28
Hattori T (2001b) Type studies of the polypores described by E. J. H. Corner from Asia and West Pacific Areas 3. Species described in Trichaptum, Albatrellus, Boletopsis, Diacanthodes, Elmerina, Fomitopsis and Gloeoporus. Mycoscience 42:423–431
Hattori T (2002) Type studies of the polypores described by E.J.H. Corner from Asia and West Pacific Areas 4. Species described in Tyromyces (1). Mycoscience 43:307–315
Hattori T (2003a) Type studies of the polypores described by E.J.H. Corner from Asia and West Pacific Areas 5. Species described in Tyromyces (2). Mycoscience 44:265–276
Hattori T (2003b) Type studies of the polypores described by E.J.H. Corner from Asia and West Pacific Areas 6. Species described in Tyromyces (3), Cristelloporia, Grifola, Hapalopilus, Heterobasidion, Ischnoderma, Loweporus, and Stecchericium. Mycoscience 44:453–463
Hibbett DS, Binder M, Bischoff JF, Blackwell M, Cannon PF, Eriksson OE, Huhndorf S, James T, Kirk PM, Lücking R, Lumbsch T, Lutzoni F, Matheny PB, Mclaughlin DJ, Powell MJ, Redhead S, Schoch CL, Spatafora JW, Stalpers JA, Vilgalys R, Aime MC, Aptroot A, Bauer R, Begerow D, Benny GL, Castlebury LA, Crous PW, Dai YC, Gams W, Geiser DM, Griffith GW, Gueidan C, Hawksworth DL, Hestmark G, Hosaka K, Humber RA, Hyde K, Ironside JE, Kõljalg U, Kurtzman CP, Larsson KH, Lichtwardt R, Longcore J, Miądlikowska J, Miller A, Moncalvo JM, Mozley-Standridge S, Oberwinkler F, Parmasto E, Reeb V, Rogers JD, Roux C, Ryvarden L, Sampaio JP, Schüßler A, Sugiyama J, Thorn RG, Tibell L, Untereiner WA, Walker C, Wang Z, Weir A, Weiß M, White MM, Winka K, Yao YJ, Zhang N (2007) A higher-level phylogenetic classification of the Fungi. Mycol Res 111:509–247
Hopple JS Jr, Vilgalys R (1999) Phylogenetic relationships in the mushroom genus Coprinus and dark-spored allies based on sequence data from the nuclear gene coding for the large ribosomal subunit RNA:divergent domains, outgroups, and monophyly. Mol Phylogenet Evol 13:1–19
Jülich W (1974) Notes on the cyanophily of spores, with discussion of the genus Leucogyrophana (Corticiaceae). Persoonia 8:51–58
Katoh K, Kuma K, Toh H, Miyata T (2005) MAFFT version 5: improvement in accuracy of multiple sequence alignment. Nucleic Acids Res 33:511–518
Katoh K, Toh H (2008) Improved accuracy of multiple ncRNA alignment by incorporating structural information into a MAFFT-based framework. BMC Bioinform 9:212
Kotiranta H (1985) Physisporinus rivulosus, an interesting polypore species. Karstenia 25:66–69
Kotlaba F, Pouzar Z (1964) Preliminary results on the staining of spores and other structures of Homobasidiomycetes in Cotton Blue and its importance for taxonomy. Feddes Repertorium 69:131–142
Larsson KH (2007) Re-thinking the classification of corticioid fungi. Mycol Res 111:1040–1063
Larsson KH, Larsson E, Kõljalg U (2004) High phylogenetic diversity among corticioid homobasidiomycetes. Mycol Res 108:983–1002
Lindner DL, Banik MT (2008) Molecular phylogeny of Laetiporus and other brown rot polypore genera in North America. Mycologia 100:417–430
Lowe JL (1966) Polyporaceae of North America. The genus Poria. State Univ Coll For Syracuse Univ Techn Publ 90:1–183
Maijala P, Kleen M, Westin C, Poppius-Levlin K, Herranen K, Lehto JH, Reponen P, Mäentausta O, Mettälä A, Hatakka A (2008) Biomechanical pulping of softwood with enzymes and white-rot fungus Physisporinus rivulosus. Enz Microb Technol 43:169–177
Matheny PB, Wang Z, Binder M, Curtis JM, Lim YW, Nilsson RH, Hughes KW, Hofstetter V, Ammirati JF, Schoch CL, Langer E, Langer G, McLaughlin DJ, Wilson AW, Froslev T, Ge ZW, Kerrigan RW, Slot JC, Yang ZL, Baroni TJ, Fischer M, Hosaka K, Matsuura K, Seidl MT, Vauras J, Hibbett DS (2007) Contributions of rpb2 and tef1 to the phylogeny of mushrooms and allies (Basidiomycota, Fungi). Mol Phyl Evol 43:430–451
Melo I, Tellería MT (1992) Novidades para a micoflora portuguesa (Aphyllophorales, Basidiomycotina). Portug Acta Biol Sér B 16:11–50
Miettinen O, Larsson KH (2010) Sidera, a new genus in Hymenochaetales with poroid and hydnoid species. Mycol Prog (in press, doi:10.1007/s11557-010-0682-5)
Miettinen O, Niemelä T, Spirin W (2006) Northern Antrodiella species:the identity of A. semisupina, and type studies of related taxa. Mycotaxon 96:211–239
Müller J, Müller K, Quandt D (2008) PhyDE – Phylogenetic Data Editor, version 0.995. http://phyde.de
Nakasone KK (1981) Cultural studies on Poria cinerascens, P. rivulosa and P. subvermispora (Aphyllophorales, Basidiomycotina). Mycotaxon 13:105–111
Niemelä T (1985) On Fennoscandian polypores 9. Gelatoporia n.gen. and Tyromyces canadensis, plus notes on Skeletocutis and Antrodia. Karstenia 25:21–40
Niemelä T (2005) Polypores, lignicolous fungi. Norrlinia 13:1–320 (in Finnish, with English summary)
Nilsson RH, Veldre V, Hartmann M, Unterseher M, Amend A, Bergsten J, Kristiansson E, Ryberg M, Jumpponen A, Abarenkov K (2010) An open source software package for automated extraction of ITS1 and ITS2 from fungal ITS sequences for use in high-throughput community assays and molecular ecology. Fungal Ecol 3:284–287
Nobles MK (1958) Cultural characters as a guide to the taxonomy and phylogeny of the Polyporaceae. Can J Bot 36:883–926
Nobles KM (1965) Identification of cultures of wood-inhabiting Hymenomycetes. Can J Bot 43:1097–1139
Penttilä R, Kotiranta H (1996) Short-term effects of prescribed burning on wood-rotting fungi. Silva Fenn 30:399–419
Rajchenberg M (1995) Basidiomicetos xilofilos (Aphyllophorales) de los bosques andinopatagonicos, adiciones y correcciones IV. Bol Soc Argent Bot 30:153–161
Robledo GL (2009) Hongos degradadores de la madera de las Yungas argentinas. Diversidad de Políporos (Aphyllophorales, Basidiomycota). Doctoral thesis, Universidad Nacional de Córdoba
Robledo GL, Amalfi M, Castillo G, Rajchenberg M, Decock C (2009) Perenniporiella chaquenia sp. nov. and further notes on Perenniporiella and its relationships with Perenniporia (Poriales, Basidiomycota). Mycologia 101:657–673
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Ryvarden L (1984) Type studies in the Polyporaceae 16. Species described by J.M. Berkeley, either alone or with other mycologists from 1856 to 1886. Mycotaxon 20:329–363
Ryvarden L, Gilbertson RL (1994) European polypores. Part 2. Meripilus – Tyromyces. Fungiflora, Oslo
Sotome K, Hattori T, Ota Y, To-anun C, Salleh B, Kakishima M (2008) Phylogenetic relationships of Polyporus and morphologically allied genera. Mycologia 100:603–615
Spirin WA (2005) Notes on some rare polypores, found in Russia 2. Junghuhnia vitellina sp. nova, plus genera Cinereomyces and Skeletocutis. Karstenia 45:103–113
Stalpers JA (1978) Identification of wood-inhabiting fungi in pure culture. Stud Mycol 16:1–248
Tomšovský M, Menkis A, Vasaitis R (2010) Phylogenetic relationships in European Ceriporiopsis species inferred from nuclear and mitochondrial ribosomal DNA sequences. Fungal Biol 114:350–358
Tortić M (1976) Some experiences with the use of cresyl blue in the determination of polypores. Kew Bull 31:611–616
Wang, Z, Dai YC, Hibbett DS (2004) Phylogenetic relationships of Sparassis inferred from nuclear and mitochondrial ribosomal DNA and RNA polymerase sequences. Mycologia 96:1015–1029
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sinisky JJ, White TJ (eds) PCR protocols: a guide to method and application. Academic, San Diego, pp 315–322
Wu SH, Nilsson HR, Chen CT, Yu SY, Hallenberg N (2010) The white-rotting genus Phanerochaete is polyphyletic and distributed throughout the phlebioid clade of the Polyporales (Basidiomycota). Fungal Divers 42:107–118
Acknowledgements
We express our gratitude to Arbinis Arbain, Syamsurdi, Rusdji Tamin, Hernawati, and Nurainas (Padang), and Pertti Uotila (H) for providing generous assistance to work in Indonesia. Genevieve Gates (Hobart) provided us with valuable specimens. Tuomo Niemelä (Helsinki) revised the manuscript, and Teuvo Ahti (Helsinki) revised the Latin. Tuuli Timonen and Pirkko Harju (Helsinki) identified some host tree species from wood attached to fungal specimens. We thank Pekka Maijala (Helsinki) for his ideas and support for this work, and Annele Hatakka (Helsinki) for providing lab facilities. Ellen Larsson, Elisabet Sjökvist (Göteborg) and Petri Oinonen (Stockholm) did most of the DNA work. The Ella and Georg Ehrnrooth Foundation provided financial support. MR is a researcher for the National Research Council of Argentina (CONICET).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miettinen, O., Rajchenberg, M. Obba and Sebipora, new polypore genera related to Cinereomyces and Gelatoporia (Polyporales, Basidiomycota). Mycol Progress 11, 131–147 (2012). https://doi.org/10.1007/s11557-010-0736-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-010-0736-8