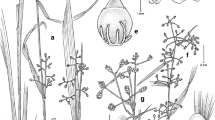
Abstract
Three new species in the genera Marasmiellus, Pusillomyces, and Gymnopus are described based on specimens found growing in the leaf litter of a mountain cloud forest relict from eastern Mexico. Distinctive macro- and micromorphological characters in combination with a phylogenetic analysis based on ITS sequences support their taxonomic identity and position in each of the above mentioned genera of the Omphalotaceae. Species here described form rhizomorphs. Morphological descriptions, including illustrations, photographs, and taxonomic discussions are provided.












Similar content being viewed by others
Data availability
The sequences generated in this study are available in the NCBI GenBank (https://www.ncbi.nlm.nih.gov/genbank/) under the accession numbers explained in the text. The ITS alignment of the phylogeny obtained in this study was deposited in the TreeBASE as Study ID 25993 (https://www.treebase.org/).
References
Antonín V (2003) New species of marasmioid genera (Basidiomycetes, Tricholomataceae) from tropical Africa - II. Gloiocephala, Marasmius, Setulipes and two new combinations. Mycotaxon 88:53–78
Antonín V, Noordeloos ME (2010) A monograph of marasmioid and collybioid fungi in Europe. IHW Verlag, Eching
Antonín V, Ryoo R, Ka KH (2014) Marasmioid and gymnopoid fungi of the Republic of Korea. 7. Gymnopus sect. Androsacei. Mycol Prog 13:703–718. https://doi.org/10.1007/s11557-013-0953-z
Aubrecht G, Huber W, Weissenfofer A (2013) Coincidence or benefit? The use of Marasmius (horse-hair fungus) filaments in bird nests. Avian Biol Res 6:26–30. https://doi.org/10.3184/175815512X13531739538638
Benson DA, Cavanaugh M, Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW (2017) GenBank. Nucleic Acids Res 45(Database issue):D37–D42. https://doi.org/10.1093/nar/gkw1070
Boddy L (1999) Saprotrophic cord-forming fungi: meeting the challenge of heterogeneous environments. Mycologia 91:13–32. https://doi.org/10.2307/3761190
César E, Bandala VM, Montoya L, Ramos A (2018) A new Gymnopus species with rhizomorphs and its record as nesting material by birds (Tyrannideae) in the subtropical cloud forest from eastern Mexico. MycoKeys 42:21–34. https://doi.org/10.3897/mycokeys.42.28894
Chaparro S, Ruiz JM (2014) Anidación del Hormiguerito de Cherrie (Myrmotherula cherriei) en Colombia, con una revisión de los nidos y huevos en Myrmotherula. Ornitol Colomb 14:136–144
Dassanayake N, Wanigasundara W, Balasuriya A, Amaratunge M (2009) A field assessment of the factors affecting horse hair blight (Marasmius equicrinis) in tea in the Ratnapura District. J Agric Sci 4:59–66. https://doi.org/10.4038/jas.v4i2.1645
Desjardin DE, Horak E (1997) Marasmius and Gloiocephala in the South Pacific Region: Papua New Guinea, New Caledonia, and New Zealand Taxa. In: Petrini O, Petrini LE, Horak E (eds) Taxonomic monographs of Agaricales II. J. Cramer, Morlenbach
Gardes M, Bruns D (1993) ITS primers with enhanced specificity for basidiomycetes application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Gilliam MS (1975) New North American species of Marasmius. Mycologia 67:817–844. https://doi.org/10.1080/00275514.1975.12019811
Gómez VH, Arriaga SL, Capello S, Rosique E, Cifuentes J (2014) Nidos de aves elaborados con material fúngico: un dato no registrado en México. Ornitología Neotropical 25:107–111
Hartig R (1873) Vorlaufige Mitteilung über den Parasitismus von Agaricus melleus und dessen Rhizomorphen. Botanische Zeitung 31:295–297
He MQ, Zhao RL, Hyde KD et al (2019) Notes, outline and divergence times of Basidiomycota. Fungal Divers 99:105–367. https://doi.org/10.1007/s13225-019-00435-4
Hedger J (1990) Fungi in the tropical forest canopy. Mycologist 4:200–202
Kalyaanamoorthy S, Minh BQ, Wong TKF, Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14:587–589. https://doi.org/10.1038/nmeth.4285
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1166. https://doi.org/10.1093/bib/bbx108
Koch RA, Lodge DJ, Sourell S, Nakasone K, McCoy AG, Aime MC (2018) Tying up loose threads: revised taxonomy and phylogeny of an avian-dispersed Neotropical rhizomorph-forming fungus. Mycol Prog 17:989–998. https://doi.org/10.1007/s11557-018-1411-8
Kornerup A, Wanscher JH (1967) Methuen handbook of colour. Methuen Co, London
Largent D, Johnson D, Watling R (1977) How to identify mushrooms to genus III: microscopic features. Mad River Press, Michigan
Mata JL, Hughes KW, Petersen RH (2007) An investigation of Omphalotaceae (Fungi: Euagarics) with emphasis on the genus Gymnopus. Sydowia 58:191–289
Menezes JC, Barbosa TC, Prezoto F (2014) Previously unreported nesting associations of the yellow-olive fly catcher (Tolmomyas sulphurescens) (Aves: Tyrannidae) with social wasps and bees. Ornitologia Neotropical 25:363–368
Mešić A, Tkalčec Z, Deng CY, Li TH, Pleše B, Ćetković H (2011) Gymnopus fuscotramus (Agaricales), a new species from southern China. Mycotaxon 117:321–330. https://doi.org/10.5248/117.321
Moncalvo JM, Vilgalys R, Redhead SA, Johnson JE, James TY, Aime MC, Hofstetter V, Verduin S, Larsson E, Baroni TJ, Thorn RG, Jacobsson S, Clémençon H, Miller OK (2002) One hundred and seventeen clades of Euagarics. Mol Phylogenet Evol 23:357–400
Montoya L, Caro A, Ramos A, Bandala VM (2019) Two new species of Lactifluus (Fungi, Russulales) from tropical Quercus forest in eastern Mexico. MycoKeys 59:27–45. https://doi.org/10.3897/mycokeys.59.38359
Moore D (1994) Tissue Formation. In: Gow N, Gadd GM (eds) The growing fungus. Chapmann and Hall, London, pp 423–457
Moore D (1998) Fungal morphogenesis. Cambridge University Press, Cambridge
Morrison DJ (2004) Rhizomorph growth habit, saprophytic ability and virulence of 15 Armillaria species. For Pathol 34:15–26. https://doi.org/10.1046/j.1439-0329.2003.00345.x
Müller J, Müller K, Neinhuis C, Quandt D (2010) PhyDE – Phylogenetic Data Editor, version 0.9971. http://www.phyde.de
Munsell (1994) Munsell soil color charts. Macbeth, New Windsor
Nguyen LT, Schmidt HA, Haeseler A, Minh BQ (2015) IQ-TREE: A fast and effective stochastic algorithm for estimating maximum likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Oliveira JJ, Vargas-Isla R, Cabral TS, Rodrigues DP, Ishikawa NK (2019) Progress on the phylogeny of the Omphalotaceae: Gymnopus s. str., Marasmiellus s. str., Paragymnopus gen. nov. and Pusillomyces gen. nov. Mycol Prog 18:713–739. https://doi.org/10.1007/s11557-019-01483-5
Peck CH (1873) Descriptions of new species of fungi. Bull Buffalo Soc Nat Sci 1:41–72
Petersen RH, Hughes KW (2016) Micromphale sect. Perforantia (Agaricales, Basidiomycetes); expansion and phylogenetic placement. MycoKeys 18:1–122. https://doi.org/10.3897/mycokeys.18.10007
Petersen RH, Hughes KW (2019) Two additional species of Gymnopus (Euagarics, Basidiomycotina). Mycokeys 45:1–24. https://doi.org/10.3897/mycokeys.45.29350
Petersen RH, Hughes KW (2020) Two new genera of gymnopoid/marasmioid euagarics. Mycotaxon 135:1–95. https://doi.org/10.5248/135.1
Prange S, Nelson DH (2006) Use of fungal rhizomorphs as nesting material by Glaucomys volans (Southern Flying Squirrels). Southeast Nat 5:355–360
Rambaut A (2018) FigTree v1.4.4 software. Institute of Evolutionary Biology, University of Edinburgh. http://tree.bio.ed.ac.uk/software/figtree/
Ronquist F, Teslenko M, Van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Scatena FN, Bruijnzeel LA, Bubb P, Das S (2010) Setting the stage. In: Bruijnzeel LA, Scatena FN, Hamilton LS (eds) Tropical Montane Cloud Forests: Science for Conservation and Management. Cambridge University Press, Cambridge
Seaver FJ (1944) The horse-hair fungi. Mycologia 36:340–342
Singer R (1965) Monographic studies on South American Basidiomycetes, especially those of the east slope of the Andes and Brazil. 2. The genus Marasmius in South America. Sydowia 18:106–358
Singer R (1973) The genera Marasmiellus, Crepidotus, and Simocybe in the Neotropics. Beihefte zur Nova Hedwigia 44:1–517
Singer R (1976) Marasmieae (Basidiomycetes- Tricholomataceae) Flora neotropica 17. The New York Botanical Garden Bronx, New York
Snaddon JL, Turner EC, Fayle TM, Khen CV, Eggleton P, Foster WA (2012) Biodiversity hanging by a thread: the importance of fungal litter-trapping systems in tropical rainforests. Biol Lett 8:397–400. https://doi.org/10.1098/rsbl.2011.1115
Su HJ, Thseng FM, Chen JS, Ko WH (2011) Production of volatile substances by rhizomorphs of Marasmius crinisequi and its significance in nature. Fungal Divers 49:199–202. https://doi.org/10.1007/s13225-010-0084-7
Takahashi H (2002) Four new species of Crinipellis and Marasmius in eastern Honshu, Japan. Mycoscience 43:343–350. https://doi.org/10.1007/S102670200050
Thiers B (2020) [continuously updated] Index Herbariorum: a global directory of public herbaria and associate staff. New York Botanical Garden’s Virtual Herbarium. http://sweetgum.nybg.org/science/ih [accessed 14 January 2020]
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Wilson AW, Desjardin DE (2005) Phylogenetic relationships in the gymnopoid and marasmioid fungi (Basidiomycetes, euagarics clade). Mycologia 97:667–679. https://doi.org/10.3852/mycologia.97.3.667
Yafetto L (2018) The structure of mycelial cords and rhizomorphs of fungi: A mini-review. Mycosphere 9:984–998. https://doi.org/10.5943/mycosphere/9/5/3
Acknowledgments
We recognize the support given by the strategic project INECOL-20035-30890 to study the macrofungi in the Santuario del Bosque de Niebla. E. César is grateful for the postdoctoral Instituto de Ecología A.C. (INECOL) grant. Thanks are given to Biol. D. Ramos (INECOL) by the assistance in the field and in the laboratory. We acknowledge the support given by the Consejo Nacional de Ciencia y Tecnología (CONACYT) (225382) to the Laboratorio de Presecuenciación, Red Biodiversidad y Sistemática, INECOL.
Funding
This research was supported by the Instituto de Ecología A.C. project 20035-30890.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Zhu-Liang Yang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
César, E., Montoya, L., Bandala, V.M. et al. Three new marasmioid-gymnopoid rhizomorph-forming species from Mexican mountain cloud forest relicts. Mycol Progress 19, 1017–1029 (2020). https://doi.org/10.1007/s11557-020-01608-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-020-01608-1