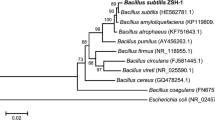
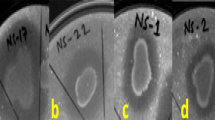
Abstract
Ten Argentinean Waitea circinata-like isolates (Corticiales, Basidiomycota) obtained from soil samples were identified to species level based on morphological features and molecular tools, and tested in order to explore their biological control abilities. Eight of them were identified as Waitea arvalis and two as W. circinata. Waitea arvalis isolates were tested as antagonists, and W. circinata isolates were used as pathogens. Most isolates performed well in in vitro antagonistic assays, inhibiting by 54–59% the growth of seven plant pathogens: Bipolaris sorokiniana, Fusarium graminearum, Fusarium oxysporum, Pythium irregulare, two different isolates of W. circinata and Sclerotium rolfsii. The most inhibited pathogen was P. irregulare (72.6%), followed by S. rolfsii (66.7%) and W. circinata (~ 65%). There was no evidence of mycoparasitism. Their capacity for chitinase, cellulase, protease and ligninase (laccase, Mn-peroxidase and lignin-peroxidase) secretion was evaluated. Most of the isolates tested positive for chitinase activity. All the evaluated W. arvalis isolates produced cellulase, protease and laccase activities, and some of them also rendered positive results for Mn-peroxidase and decolorized the dye Azure B. Isolates INTA-L003 and INTA-L005 displayed top results for cellulase activity (both in carboxymethylcellulose plate assay and in filter paper tube assay). These activities might be responsible for their better antagonistic behavior on Pythium sp. Thus, these isolates are promising as biocontrol agents and as sources of new enzymes for industrial applications. Moreover, to the best of our knowledge, this is the first citation of W. arvalis in Argentina.









Similar content being viewed by others
Abbreviations
- PDA:
-
Potato dextrose agar
- MEA:
-
Malt extract agar
- SNA:
-
Special nutrient agar
- PDB:
-
Potato dextrose broth
- ITS:
-
Internal transcribed spacer
- nc LSU rDNA:
-
Nuclear large subunit ribosomal DNA
- FPase:
-
Filter paper degrading enzymes
- CMC:
-
Carboxymethylcellulose
- CMCase:
-
Carboxymethylcellulose degrading enzymes
- Lac:
-
Laccase
- MnP:
-
Mn-peroxidase
- LiP:
-
Lignin-peroxidase
- EI:
-
Enzymatic index
References
Agrawal S, Bhatt A (2023) Microbial endophytes: emerging trends and biotechnological applications. Curr Microbiol 80(8):249. https://doi.org/10.1007/s00284-023-03349-2
Allen MF, Boosalis MG, Kerr ED, Muldoon AE, Larsen HJ (1985) Population dynamics of sugar beets, Rhizoctonia solani, and Laetisaria arvalis: responses of a host, plant pathogen, and hyperparasite to perturbation in the field. Appl Environ Microbiol 50:1123–1127. https://doi.org/10.1128/aem.50.5.1123-1127.1985
Al-Saeedi SS, Al-Ani BM (2014) Study of antagonistic capability of Trichoderma harzianum isolates against some pathogenic soil borne fungi. ABJNA 5:15–23. https://doi.org/10.5251/abjna.2014.5.1.15.23
Andreotti M, Rojo R, López N, Beribe MJ, Agamennoni R, Baffoni P et al (2012) Efecto de las rotaciones en cultivo de cebolla sobre la presencia de Fusarium oxysporum Schlecht. y hongos antagonistas en el suelo. In: Proceedings of VIX Jornadas Fitosanitarias Argentinas (Potrero de Los Funes, Argentina) 365
Archibald FS (1992) A new assay for lignin-type peroxidases employing the dye Azure B. Appl Environ Microbiol 58:3110–3116. https://doi.org/10.1128/aem.58.9.3110-3116.1992
Barrera VA, Rojo R, Chiessa G, López N, Baffoni P, Agamennoni R et al (2012) Abundancia y diversidad de comunidades microbianas de suelos bajo diferentes rotaciones. JBAG 23(Suppl.):225–226
Barrera VA, Iannone L, Romero AI, Chaverri P (2021) Expanding the Trichoderma harzianum species complex: Three new species from Argentine natural and cultivated ecosystems. Mycologia 113(6):1136–1155. https://doi.org/10.1080/00275514.2021.1947641
Bobba V, Conway KE (2003) Competitive saprophytic ability of Laetisaria arvalis compared with Sclerotium rolfsii. POAS 83:17–22
Bowers WS, Hoch HC, Evans PH, Katayama M (1986) Thallophytic allelopathy: Isolation and identification of laetisaric acid. Science 232:105–106. https://doi.org/10.1126/science.232.4746.105
Brewer MT, Larkin RP (2005) Efficacy of several potential biocontrol organisms against Rhizoctonia solani on potato. Crop Prot 24:939–950. https://doi.org/10.1016/j.cropro.2005.01.012
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A et al (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.3929/ethz-b-000240890
Burdsall HH, Hoch HC, Boosalis MG, Setliff EC (1980) Laetisaria arvalis (Aphyllophorales, Corticiaceae): A possible biological control agent for Rhizoctonia solani and Pythium species. Mycologia 72:728–736. https://doi.org/10.1080/00275514.1980.12021241
Chakrabarti S, Tiwari N, Khule P, Marar S (2018) Isolation of protease producing bacteria from spoiled food and extraction of protease enzyme. IJAR 4:141–146
Chen D, Hou Q, Fan B, Zhang H, Jia L, Sun K (2022) Biocontrol potential of endophytic Trichoderma citrinoviride HT-1 against root rot of Rheum palmatum through both antagonistic effects and induced systemic resistance. World J Microbiol Biotechnol 38:1–9. https://doi.org/10.1007/s11274-022-03272-x
Collinge DB, Jensen DF, Rabiey M, Sarrocco S, Shaw MW, Shaw RH (2022) Biological control of plant diseases–What has been achieved and what is the direction? Plant Pathol 71(5):1024–1047. https://doi.org/10.1111/ppa.13555
Conway KE (1995) Comparison of biological and chemical treatments for control of southern blight of apple seedlings in the field. Biol & Cult Tests 10:49
Conway KE, Maness NE, Motes JE (1997) Integration of biological and chemical controls for Rhizoctonia aerial blight and root rot of rosemary. Plant Dis 81:795–798. https://doi.org/10.1094/PDIS.1997.81.7.795
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: Version II. Plant Mol Biol Rep 1:19–21. https://doi.org/10.1007/BF02712670
Diederich P, Lawrey JD, Sikaroodi M, Gillevet PM (2011) A new lichenicolous teleomorph is related to plant pathogens in Laetisaria and Limonomyces (Basidiomycota, Corticiales). Mycologia 103:525–533. https://doi.org/10.3852/10-255
Diederich P, Lawrey JD, Ertz D (2018) The 2018 classification and checklist of lichenicolous fungi, with 2000 non-lichenized, obligately lichenicolous taxa. Bryologist 121:340–425. https://doi.org/10.1639/0007-2745-121.3.340
Druzhinina IS, Seidl-Seiboth V, Herrera-Estrella A, Horwitz BA, Kenerley CM, Monte E et al (2011) Trichoderma: The genomics of opportunistic success. Nat Rev Microbiol 9:749–759. https://doi.org/10.1038/nrmicro2637
Dutt K, Meghwanshi GK, Gupta P, Saxena RK (2008) Role of casein on induction and enhancement of production of a bacterial milk clotting protease from an indigenously isolated Bacillus subtilis. Lett Appl Microbiol 46:513–518. https://doi.org/10.1111/j.1472-765X.2008.02324.x
Fernández di Pardo A, Chiocchio VM, Barrera V, Colombo RP, Martinez AE, Gasoni L et al (2015) Mycorrhizal fungi isolated from native terrestrial orchids of pristine regions in Córdoba (Argentina). Rev Biol Trop 63:275–284
Floudas D, Binder M, Riley R, Barry K, Blanchette RA, Henrissat B et al (2012) The paleozoic origin of enzymatic lignin decomposition reconstructed from 31 fungal genomes. Science 336:1715–1719. https://doi.org/10.1126/science.1222218
Ghobad-Nejhad M, Langer E, Nakasone K, Diederich P, Nilsson RH, Rajchenberg M et al (2021) Digging up the roots: Taxonomic and phylogenetic disentanglements in Corticiaceae s.s. (Corticiales, Basidiomycota) and evolution of nutritional modes. Front Microbiol 12:1–21. https://doi.org/10.3389/fmicb.2021.704802
Ghoshroy K, Cisneros D, Lartey RT, Ghoshroy S (2012) Structural analysis of the inhibition of Pyrenophora teres by Laetisaria arvalis. Microsc Microanal 18:168–169. https://doi.org/10.1017/S1431927612002693
Goloboff PA (1999) Analyzing large data sets in reasonable times: solutions for composite optima. Cladistics 15:415–428. https://doi.org/10.1111/J.1096-0031.1999.TB00278.X
Goloboff PA, Farris JS, Nixon KC (2008) TNT, a free program for phylogenetic analysis. Cladistics 24:774–786. https://doi.org/10.1111/J.1096-0031.2008.00217.X
Grassi E, Scodeller P, Filiel N, Carballo R, Levin L (2011) Potential of Trametes trogii culture fluids and its purified laccase for the decolorization of different types of recalcitrant dyes without the addition of redox mediators. Int Biodeterior Biodegradation 65:635–643. https://doi.org/10.1016/j.ibiod.2011.03.007
Gruhn G, Ghobad-Nejhad M (2020) A new species and a new combination in Waitea (Corticiales, Basidiomycota) and the phylogenetic affinity of Disporotrichum. Mycol Iran 7:171–179. https://doi.org/10.22043/mi.2021.124280
Gupta VP, Tewari SK, Bajpai AK (1999) Ultrastructure of mycoparasitism of Trichoderma, Gliocladium and Laetisaria species on Botryodiplodia theobromae. J Phytopathol 147:19–24. https://doi.org/10.1046/j.1439-0434.1999.147001019.x
Guzmán-Guzmán P, Porras-Troncoso MD, Olmedo-Monfil V, Herrera-Estrella A (2019) Trichoderma species: versatile plant symbionts. Phytopathology 109(1):6–16. https://doi.org/10.1094/PHYTO-07-18-0218-RVW
Hermosa R, Viterbo A, Chet I, Monte E (2012) Plant-beneficial effects of Trichoderma and of its genes. Microbiology 158(1):17–25. https://doi.org/10.1099/mic.0.052274-0
Herr LJ (1988) Biocontrol of Rhizoctonia crown and root rot of sugar beet by binucleate Rhizoctonia spp. and Laetisaria arvalis. Ann Appl Biol 113:107–118. https://doi.org/10.1111/j.1744-7348.1988.tb03287.x
Hinton MJ, Parry DW (1993) Screening selected fungi for antagonism towards Pseudocercosporella herpotrichoides (Fron) Deighton, the cause of eyespot disease of cereals. Biocontrol Sci Technol 3:13–19. https://doi.org/10.1080/09583159309355254
Hoch HC, Abawi GS (1979) Biological control of Pyhtium root rot of table beet with Corticium sp. Phytopathology 69:417–419. https://doi.org/10.1094/Phyto-69-417
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755. https://doi.org/10.1093/bioinformatics/17.8.754
Inglis PW, Mello SCM, Martins I, Silva JBT, Macedo K, Sifuentes DN et al (2020) Trichoderma from Brazilian garlic and onion crop soils and description of two new species: Trichoderma azevedoi and Trichoderma peberdyi. PLoS ONE 15:1–23. https://doi.org/10.1371/journal.pone.0228485
Inokuma K, Takano M, Hoshino K (2013) Direct ethanol production from N-acetylglucosamine and chitin substrates by Mucor species. Biochem Eng J 72:24–32. https://doi.org/10.1016/j.bej.2012.12.009
Jayawardena RS, Hyde KD, Jeewon R, Ghobad-Nejhad M, Wanasinghe DN, Liu N et al (2019) One stop shop II: taxonomic update with molecular phylogeny for important phytopathogenic genera: 26–50. Fungal Divers 94:41–129. https://doi.org/10.1007/s13225-019-00418-5
Jones RK, Belmar SB (1989) Characterization and pathogenicity of Rhizoctonia spp. isolated from rice, soybean, and other crops grown in rotation with rice in Texas. Plant Dis 73:1004–1010. https://doi.org/10.1094/PD-73-1004
Joshi D, Singh P, Singh AK, Srivastava TK, Tripathi N (2015) Relative effects of different organic amendments on Trichoderma communities in soils under a sugarcane plant-ratoon agro-ecosystem. IJST 30:32–38
Khiralla A, Spina R, Saliba S, Laurain-Mattar D (2019) Diversity of natural products of the genera Curvularia and Bipolaris. Fungal Biol Rev 33:101–122. https://doi.org/10.1016/J.FBR.2018.09.002
Kirk PM, Cannon PF, David JC, Stalpers JA (2001) Dictionary of the fungi, 9th edn. CAB International, Wallingford
Köhl J, Kolnaar R, Ravensberg WJ (2019) Mode of action of microbial biological control agents against plant diseases: relevance beyond efficacy. Front Plant Sci 10:845. https://doi.org/10.3389/fpls.2019.00845
Kuzu SB, Güvenmez HK, Denizci AA (2012) Production of a thermostable and alkaline chitinase by Bacillus thuringiensis subsp. kurstaki strain HBK-51. Biotechnol Res Int 2012:135498. https://doi.org/10.1155/2012/135498
Larkin RP, Brewer MT (2020) Effects of crop rotation and biocontrol amendments on Rhizoctonia disease of potato and soil microbial communities. Agriculture 10:128. https://doi.org/10.3390/agriculture10040128
Larkin RP, Fravel DR (1998) Efficacy of various fungal and bacterial biocontrol organisms for control of Fusarium wilt of tomato. Plant Dis 82:1022–1028. https://doi.org/10.1094/PDIS.1998.82.9.1022
Larsen HJ, Boosalis MG, Kerr ED (1985) Temporary depression of Rhizoctonia solani field populations by soil amendment with Laetisaria arvalis. Plant Dis 69:347–350
Lartey RT, Curl EA, Peterson CM, Williams JC (1991) Control of Rhizoctonia solani and cotton seedling disease by Laetisaria arvalis and a mycophagous insect Proisotoma minuta (Collembola). J Phytopathol 133:89–98. https://doi.org/10.1111/j.1439-0434.1991.tb00141.x
Lewis JA, Papavizas GC (1992) Potential of Laetisaria arvalis for the biocontrol of Rhizoctonia solani. Soil Biol Biochem 24:1075–1079. https://doi.org/10.1016/0038-0717(92)90055-3
Lopes MA, Gomes DS, Bello Koblitz MG, Pirovani CP, Cascardo J, Góes-Neto A et al (2008) Use of response surface methodology to examine chitinase regulation in the basidiomycete Moniliophthora perniciosa. Mycol Res 112:399–406. https://doi.org/10.1016/j.mycres.2007.10.017
Marion F, Rojo R, Torres M, Martínez MC, Gasoni L, Barrera VA (2015) Estudios preliminares sobre un género nuevo para Argentina, Laetisaria. In: Proceedings of III Congreso Argentino de Microbiología Agrícola y Ambiental (CABA, Argentina), pp 153. https://panel.aam.org.ar/img_up/07112022.1.pdf
Martin SB, Hoch HC, Abawi GS (1983) Population dynamics of Laetisaria arvalis and low-temperature Pythium spp. in untreated and pasteurized beet field soils. Phytopathology 73:1445–1449. https://doi.org/10.1094/Phyto-73-1445
Martin SB, Abawi GS, Hoch HC (1984) Influence of the antagonist Laetisaria arvalis on infection of table beets by Phoma betae. Phytopathology 74:1092–1096. https://doi.org/10.1094/Phyto-74-1092
Martin SB, Abawi GS, Hoch HC (1986) The relation of population densities of the antagonist, Laetisaria arvalis, to seedling diseases of table beet incited by Pythium ultimum. Can J Microbiol 32:156–159. https://doi.org/10.1139/m86-031
Martin ME (2019) Caracterización de aislamientos nativos de hongos del género Cladorrhinum y su potencial como agentes antagonistas y promotores del crecimiento vegetal. Revista de Divulgación Técnica Agropecuaria, Agroindustrial y Ambiental. Facultad de Ciencias Agrarias. UNLZ. 6(2):31. https://revistafcaunlz.gramaweb.com.ar/2019/06/
Melignani E, Martin ME, Mema VY, Taibo CB, Sabio y García JV, Barrera VA (2023) Mycoparasitic interaction between Trichoderma afroharzianum strain Th2RI99 and Rhizoctonia solani. Rev Arg Microbiol 55:199–200. https://doi.org/10.1016/j.ram.2022.09.005
Melignani E, Barrera V, Guidobono JS, Levin L (2018a) Evaluación de la acción quitinolítica en aislamientos nativos del género Laetisaria Burds. (Corticiaceae, Basidiomycota). In: Proceedings of IV Congreso Argentino de Microbiología Agrícola y Ambiental (Mar del Plata, Argentina), pp 262
Melignani E, Levin L, Guidobono JS, Barrera V (2018b) Evaluación de antagonismo en aislamientos nativos del género Laetisaria Burds. (Corticiaceae, Basidiomycota). In: Proceedings of IV Congreso Argentino de Microbiología Agrícola y Ambiental (Mar del Plata, Argentina), pp 201
Meng L, Mestdagh H, Ameye M, Audenaert K, Höfte M, Van Labeke M-C (2020) Phenotypic variation of Botrytis cinerea isolates is influenced by spectral light quality. Front Plant Sci 11:1233. https://doi.org/10.3389/fpls.2020.01233
Mercado Cárdenas GE, Galván MZ, Barrera VA, Rodriguero MS, Carmona MA, March GJ et al (2015) Molecular identification and pathogenicity of Rhizoctonia spp. from tobacco growing areas in northwestern Argentina. Trop Plant Pathol 40:160–168. https://doi.org/10.1007/s40858-015-0035-7
Mukherjee PK, Mendoza-Mendoza A, Zeilinger S, Horwitz BA (2022) Mycoparasitism as a mechanism of Trichoderma-mediated suppression of plant diseases. Fungal Biol Rev 39:15–33. https://doi.org/10.1016/J.FBR.2021.11.004
Murdoch CW, Leach SS (1993) Evaluation of Laetisaria arvalis as a biological control agent of Rhizoctonia solani on white potato. Am Potato J 70:625–634. https://doi.org/10.1007/BF02849152
Muthamilan M, Jeyarajan R (1992) Effect of antagonistic fungi on Sclerotium rolfsii causing root rot of groundnut. J Biol Control 6:88–92
Muthukumar A, Venkatesh A (2013) Effect of light and aeration on the growth of Sclerotium rolfsii in vitro. Afr J Biotechnol 12:6843–6846. https://doi.org/10.5897/AJB2013.13202
Nagy P, Fischl G (2002a) The effect of UV and visible light irradiation on the development of microsclerotium of the fungus Macrophomina phaseolina. Cereal Res Commun 30:383–389. https://doi.org/10.1007/BF03543433
Nagy P, Fischl G (2002b) Effect of UV and visible light irradiation on mycelial growth and sclerotium formation of Sclerotinia sclerotiorum. Acta Phytopathol Entomol Hung 37:83–89. https://doi.org/10.1556/APhyt.37.2002.1-3.9
Nakasone KK, Peterson SW, Jong SC (2004) Preservation and distribution of fungal cultures. In: Mueller GM, Bills GF, Foster MS (eds) Biodiversity of fungi: Inventory and monitoring methods. Elsevier Academic Press, Amsterdam, pp 37–47. https://doi.org/10.1016/B978-012509551-8/50006-4
Navarro D, Rosso M-N, Haon M, Olivé C, Bonnin E, Lesage-Meessen L et al (2014) Fast solubilization of recalcitrant cellulosic biomass by the basidiomycete fungus Laetisaria arvalis involves successive secretion of oxidative and hydrolytic enzymes. Biotechnol Biofuels 7:143–156. https://doi.org/10.1186/s13068-014-0143-5
Nelson N (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J Biol Chem 153:375–380. https://doi.org/10.1016/S0021-9258(18)71980-7
Ng LC, Ngadin A, Azhari M, Zahari NA (2015) Potential of Trichoderma spp. as biological control agents against bakanae pathogen (Fusarium fujikuroi) in rice. Asian J Plant Pathol 9:46–58. https://doi.org/10.3923/ajppaj.2015.46.58
Nixon KC (2002) WinClada version1.00.08. Published by the author, Ithaca, NY
Odvody GN, Boosalis MG, Kerr ED (1980) Biological control of Rhizoctonia solani with a soil-inhabiting basidiomycete. Phytopathology 70:655–658. https://doi.org/10.1094/Phyto-70-655
Parveen T, Sharma K (2015) Pythium diseases, control and management strategies: a review. IJPAES 5:244–257
Petrisor C, Paica A, Constantinescu F (2015) Screening of Trichoderma sp. strains for producing hydrolytic enzymes. Rom J Plant Prot 8:7–10
Pinheiro J, Bates D, R Core Team (2023) nlme: Linear and nonlinear mixed effects models. R package version 3.1–162. https://CRAN.R-project.org/package=nlme
Pitchurajan K, Aishwarya M, Sujitha S, Saranya Devi K, Mohan V (2020) Screening and selection of potential plant beneficial rhizosphere fungi from medicinally important plant species in Tamil Nadu, India. J Acad Indus Res 8:153–159
R Core Team (2022) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org
di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2020) InfoStat Versión 2020
Riley R, Salamov AA, Brown DW, Nagy LG, Floudas D, Held BW et al (2014) Extensive sampling of basidiomycete genomes demonstrates inadequacy of the white-rot/brown-rot paradigm for wood decay fungi. PNAS 111:9923–9928. https://doi.org/10.1073/PNAS.1400592111
Romano N, Gioffré A, Sede SM, Campos E, Cataldi A, Talia P (2013) Characterization of cellulolytic activities of environmental bacterial consortia from an Argentinian native forest. Curr Microbiol 67:138–147. https://doi.org/10.1007/s00284-013-0345-2
Romero Peláez RD, Wischral D, Demarchi Mendes T, Fraga Pacheco T, Fontes Urben A, Vieira Helm C et al (2021) Co-culturing of micro- and macro-fungi for producing highly active enzyme cocktail for producing biofuels. Bioresour Technol Rep 16:100833. https://doi.org/10.1016/j.biteb.2021.100833
Sanivada SK, Challa M (2014) Mycolytic effect of extracellular enzymes of entomopathogenic fungi to Colletotrichum falcatum, red rot pathogen of sugarcane. J Biopest 7:33–37. https://doi.org/10.13140/2.1.4705.8562
Steffen KT, Hofrichter M, Hatakka A (2000) Mineralisation of 14C-labelled synthetic lignin and ligninolytic enzyme activities of litter-decomposing basidiomycetous fungi. Appl Microbiol Biotechnol 54:819–825. https://doi.org/10.1007/s002530000473
Sumner DR, Bell DK (1994) Survival of Rhizoctonia spp. and root diseases in rotation of corn, snap bean, and peanut in microplots. Phytopathology 84:113–118. https://doi.org/10.1094/Phyto-84-113
Sumner DR, Lewis JA, Gitaitis RD (1992) Chemical and biological control of Rhizoctonia solani AG-4 in snap bean double-cropped with corn. Crop Prot 11:121–126. https://doi.org/10.1016/0261-2194(92)90093-K
Tamura K, Stecher G, Kumar S (2021) MEGA11: Molecular evolutionary genetics analysis Version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Thambugala KM, Daranagama DA, Phillips AJL, Kannangara SD, Promputtha I (2020) Fungi vs. fungi in biocontrol: an overview of fungal antagonists applied against fungal plant pathogens. Front Cell Infect Microbiol 10: 604923
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
van den Boogert PHJF, Reinartz H, Sjollema KA, Veenhuis M (1989) Microscopic observations on the interaction of the mycoparasite Verticillium biguttatum with Rhizoctonia solani and other soil-borne fungi. Antonie Leeuwenhoek 56:161–174. https://doi.org/10.1007/BF00399979
Verma S, Dohroo NP (2005) Comparative efficacy of biocontrol agents against Fusarium wilt of pea. In: Shama RC, Sharma JN (eds) Integrated plant disease management. Scientific Publishers, India, pp 93–99
White TJ, Bruns T, Lee SJWT, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: A guide to methods and applications, Academic Press, New York, pp 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Wu W, Ogawa F, Ochiai M, Yamada K, Fukui H (2020) Common strategies to control Pythium disease. Rev Agric Sci 8:58–69. https://doi.org/10.7831/ras.8.0_58
Yu Z, Fischer R (2018) Light sensing and responses in fungi. Nat Rev 17:25–36. https://doi.org/10.1038/s41579-018-0109-x
Zhao J, Koker TH, Janse BJH (1996) Comparative studies of lignin peroxidases and manganese-dependent peroxidases produced by selected white rot fungi in solid media. FEMS Microbiol Lett 145:393–399. https://doi.org/10.1111/j.1574-6968.1996.tb08606.x
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer-Verlag, New York. https://doi.org/10.1007/978-0-387-87458-6
Acknowledgements
The authors wish to thank Federico Marion for the contributions made to the Waitea collection.
Funding
The present work was supported with funds from Instituto Nacional de Tecnología Agropecuaria (INTA) through the Disciplinary Project 201-PD-E4-I069-001, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET PIP11220170100283) and Universidad de Buenos Aires (UBACYT 2020020170100163).
Author information
Authors and Affiliations
Contributions
Eliana Melignani, Rodrigo Alejandro Rojo, Laura Noemí Levin and Viviana Andrea Barrera contributed to the study conception and design. Material preparation, data collection and analysis were performed by Eliana Melignani, Rodrigo Alejandro Rojo and Viviana Andrea Barrera. The statistical analysis was performed by Juan Santiago Guidobono. Funding acquisition and resources were provided by Viviana Andrea Barrera and Laura Noemí Levin. The first draft of the manuscript was written by Eliana Melignani, Laura Noemí Levin and Viviana Andrea Barrera and all authors commented on previous versions of the manuscript. Supervision at all stages of this work was performed by Viviana Andrea Barrera and Laura Noemí Levin.
All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors agree with the content of the manuscript, all authors give explicit consent to submit the manuscript and all authors obtained consent from the responsible authorities at the institutions where the work has been carried out.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Section Editor: Martin Ruhl
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Laura Noemí Levin and Viviana Andrea Barrera share last authorship.
Supplementary Information
Below is the link to the electronic supplementary material.
11557_2024_1944_MOESM2_ESM.pdf
Supplementary file2 (PDF 77 KB) Statistical results for the growth radius (mm) of W. arvalis isolates in different media and temperatures (after 72 h).
11557_2024_1944_MOESM6_ESM.pdf
Supplementary file6 (PDF 105 KB) Growth Inhibition percentages (GI, %) of the pathogens from dual culture assays performed with W. arvalis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Melignani, E., Guidobono, J.S., Rojo, R.A. et al. Studies on the biotechnological potential of Argentinean isolates of Waitea arvalis (Corticiales, Basidiomycota) for the biological control of phytopathogenic fungi. Mycol Progress 23, 16 (2024). https://doi.org/10.1007/s11557-024-01944-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-024-01944-6