Abstract
In the light of ongoing climate change, European beech trees face a loss of vitality due to an expected increase in the frequency of droughts, exacerbated by prolonged periods of high temperatures and solar radiation. Such conditions increase the predisposition to European beech splendour beetle attacks, and trigger sunburn, fungal infections and complex diseases. As a result, European beech forests are seriously threatened by climate change. Following prolonged and repeated periods of exceptional drought since 2018, European beech decline have been observed in several regions of Germany and Europe. Vitality losses, in some cases severe, of due to the extraordinarily warm, dry summers of 2018 and 2019 have been observed and investigated in several regions of Germany. The first symptoms of vitality loss of European beech associated with severe signs of crown dieback were recorded in midsummer 2018. Mortality of branches and individual trees was often associated with sunburn, bark necrosis and bleeding spots of sap flux. The first signs of mortality were observed locally in predisposed stands in autumn 2018. The stem stability and break resistance of affected trees were significantly reduced. To elucidate the complex interactions involved, the occurrence of vitality loss of European beech and associated causative agents were examined in north-western Germany. Disease symptoms of individual trees were studied, associated fungi (endophytes, plant pathogens and saprobes) were isolated and identified. Frequent potentially severe pathogens included Neonectria coccinea, Diplodia corticola and D. mutila. The presented cases of disease were the first, where D. corticola was recorded on beech. In addition, inoculation tests were used to assess the potential roles of Botryosphaeria species in the observed symptomatology.
Similar content being viewed by others
Introduction
European beech (Fagus sylvatica L.) is the most important broadleaved tree species in terms of areal coverage in natural woodland vegetation in Germany (BMEL 2018). There, it is the most common tree species in managed forests. Beech forests (Fagion sylvaticae) are characteristic elements of German potential natural zonal vegetation, i.e. the vegetation that would prevail under current environmental conditions in the absence of human intervention (Tüxen and Preising 1956; Ellenberg 1996; Bohn et al. 2003).
European beech has a wide ecological (climatic and geological) amplitude, as well as high shade tolerance and growth capacity (Ellenberg 1996). It is a moderately anisohydric species with usually smooth bark. Beech trees are considered to be severely threatened by increasing frequencies and intensities of drought (Rennenberg et al. 2004; Ammer et al. 2005; Pflug et al. 2018; Leuschner 2020).
Following periods of exceptional drought since 2018, European beech decline have been observed in Germany (Langer et al. 2020; Langer and Bußkamp 2021; Purahong et al. 2021) and Europe (Neycken et al. 2022; Martinez Del Castillo et al. 2022). Overall, affected trees were exposed to attacks by fungal pathogens and insects and exhibited severe crown dieback (Neycken et al. 2022). The observed symptoms can be assigned to one of the most common complex diseases of beech, the so-called Vitality loss of beech (VLB) (Brück-Dyckhoff et al. 2019; Langer 2019a, b). Beside beech bark disease (BBD) (Ehrlich 1934) and disease patterns initially caused by Phytophthora spp. (Jung 2009; Langer 2019a), VLB is a major threat to mature European beech stands in Europe. Because both, BBD and VLB, are associated with bleeding spots of sap flux and are often attacked by the same secondary fungal pathogens and insects, they are not easily distinguished without differential diagnosis. Therefore, a brief description of BBD and VLB (Fig. 1) is given below.
Disease symptoms of vitality loss of European beech (VLB): a affected beech tree with peeling bark; b affected beech trunk with bleeding spots of sap flux; c–d bleeding spots with removed bark, red arrow: isolation locus of Neonectria coccinea, yellow arrows: isolation loci of Diplodia mutila; e basal stem disc with central wood stain by air inclusion and wood rot due to Biscogniauxia nummularia (blue arrows); f basal stem disc with wood and bark necroses due to Diplodia mutila (isolation loci: yellow arrows), green arrow: isolation locus Diplodia sapinea
In Europe, BBD has frequently occurred in multi-annual disease outbreaks (Niesar 2007). The disease is initially started by the Woolly beech scale (Cryptococcus fagisuga Lind., Eriococcidae, Hemiptera) and was first reported in Germany (McIntosh 1849; Hartig 1878). The leading fungal causative agent in Europe is Neonectria coccinea (Pers.) Rossman & Samuels, which is an abundant endophyte of healthy beech wood and wood of other broadleaved trees (Sieber 2007). Pathogenic growth of this ascomycete is preceded by excessive infestation with the phloem-feeding C. fagisuga. Neonectria coccinea caused annual necrotic bark lesions and sap flow bleeding. The necroses and cankers caused by N. coccinea are often strip-like (Grüner and Metzler 2006), followed by bark beetle infestation and subsequent infection by secondary pathogens. Affected beech trees usually die when the stem is girdled, often while the crown is still green (Langer 2019a).
Unlike BBD and infection with Phytophthora, VLB is induced by abiotic factors, particularly abnormal weather conditions, above all drought or prolonged heatwaves. Affected trees or stands are often predisposed by site factors, for instance, a southern exposition with high direct solar irradiation of the stem(s) (Langer 2019a). Such conditions reduce the vitality of affected trees and cause sunburn, increasing their susceptibility to attack by bark and wood-decaying fungi, the European beech bark beetle (Agrilus viridis L.) and other bark and wood-feeding beetles (Bressem 2008; Brück-Dyckhoff et al. 2019; Langer 2019a). The infestation with these insects and fungi lead to leaf loss, dieback, unusual accumulation of dead knots, bleeding spots on the stem (Fig. 1b), stem necroses (Fig. 1c–d) and, subsequently, tree mortality (Fig. 1a). There is evidence that Botryosphaeriaceae species, in addition to N. coccinea, may play an important role in current disease outbreaks. (Langer et al. 2020; Wohlgemuth et al. 2020).
The ascomycetous Botryosphaeriaceae family encompasses morphologically diverse, endophytic, pathogenic or saprophytic fungi, which mostly have woody hosts (Phillips et al. 2013). Several authors (Arx and Müller 1954, 1975; Eriksson 1981; Sivanesan 1984; Barr 1987; Phillips et al. 2008, 2013; Liu et al. 2012) have provided detailed descriptions of the family. Due to climatic warming and more extreme weather conditions, a recent increase in pathogenic activity and geographic range expansion of the Botryosphaeriaceae in Europe has been observed (Piškur et al. 2011; Fabre et al. 2011; Zlatković et al. 2017; Brodde et al. 2019). This is consistent with the observation that latent pathogens often switch from an endophytic to a pathogenic lifestyle when their hosts are stressed, e.g. by drought (Slippers and Wingfield 2007; Mehl et al. 2013). It also raises the question of whether new pathogenic species have appeared in Germany as a result of climate warming and whether VLB or globalisation and/or native members of the family with no previously recorded pathogenicity have contributed to VLB.
Low precipitation, together with extraordinarily high temperatures and solar radiation in the summer (DWD 2019; NOAA 2019), led to the most severe drought in Europe for the last 500 years. The Northwest German Forest Research Institute (NW-FVA) observed the first signs of VLB in the summer of 2018 (Langer 2019a; Langer et al. 2020). First investigations on associated microorganisms in the current outbreak of the VLB were performed by Purahong et al. (2021) and Langer and Bußkamp (2021). As little is known about the composition of associated fungi and their specific role in damage intensification, the objectives of the study reported here were to (1) identify and isolate fungi associated with current VLB, (2) conduct pathogenicity tests on beech for the isolated Botryosphaeriaceae.
Material and methods
Studied sites and disease cases
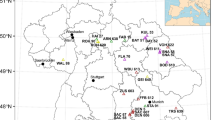
The sites and disease cases studied in 2018 and 2019 were located in forest stands in north-western Germany (Fig. 2). Altitudes of the sites range from 5 to 800 m above sea level. The studied stands grow mainly on brown earth and parabrown soils. To create maps, QGIS (www.qgis.org) was used, while site conditions were registered using the Soil Map of Germany (BÜK200), published by the Federal Institute for Geosciences and Natural Resources (BGR 2020) and a digital elevation model provided by the Federal Agency for Cartography and Geodesy (BKG 2019) to characterise potential predisposing and triggering factors. In 55 cases of disease (reported here and studied in detail, Online Resource 1), the causal agents were identified in 86 trees.
Map of Germany with reports of stands and cases of disease with vitality loss of European beech (VLB) in the area in the behalf of the NW-FVA (Schleswig–Holstein, Lower Saxony, Saxony-Anhalt and Hesse) and an enlarged portion of the map showing the twelve location of the forest stands with evidence of Diplodia spp. Species incidences are indicated by differently coloured circular segments. Sources: Data on reports of forest stands with VLB retrieved from the forest protection reporting portal “Waldschutzmeldeportal” of the NW-FVA and of the 55 VLB cases investigated in detail by the authors obtained in the period 01.01.2018–31.12.2019.
Isolation and identification of fungi
The scientific name of the fungal species is usually given according to the Index Fungorum (www.indexfungorum.org). Fungi associated with cases of disease were identified in the field when fruiting bodies were observed. Fruiting bodies that could not be identified ad hoc were investigated using a Zeiss Axiostar Plus microscope following standard procedures for fungi described by Lee and Langer (2012). In addition to standard literature recommended by Oertel (2003) for determination of fungi and forest diseases, we also consulted relevant works by Alves et al. (2004), Arx (1981), Booth (1971), Butin (2011), Domsch et al. (1980), Gerlach and Nirenberg (1982), Hartmann et al. (2007), Kuch et al. (2014), Shoemaker (1964), Slippers et al. (2004), and Zlatković et al. (2018, 2019).
To isolate the disease-causing fungal pathogens involved, symptomatic tissues (bark necroses, leaking spots and wood discolouration) were sampled and incubated on the standard culture medium malt yeast peptone agar (MYP), modified according to Langer (1994), containing 0.7% malt extract (Merck 1.05391.0500, Darmstadt, Germany), 0.05% yeast extract (Fluka 70,161-100G, Seelze, Germany), 0.1% peptone (Merck 1.07272.0500) and 1.5% agar (Fluka 05,040-1 KG). Tree tissues were exposed for sampling by removing bark at the necrotic surfaces, followed by sectioning. After spray surface disinfection with 70% ethanol and removal of the outermost layer, 1 × 0.5 cm wood or bark chips were obtained and placed in Petri dishes with culture medium, then incubated at room temperature in daylight. The material in Petri dishes was periodically checked for emerging mycelia, which were subsequently sub-cultured on fresh MYP medium to obtain pure cultures. Isolated, representative fungal strains were stored in MYP slant tubes at 4 °C in the fungal culture collection of the Northwest German Forest Research Institute (NW‐FVA).
Isolated strains were assigned to morphotypes and identified by micro-morphological characters and/or sequencing of the ITS region 1–4 (White et al. 1990). At least one representative strain of each morphotype was used for molecular identification, involving DNA extraction from the mycelium, following the method of Izumitsu et al. (2012), then PCR and sequencing of the target region, following the method of Bußkamp et al.(2020). Sequences obtained were aligned and manually edited using MEGA 5 software (Tamura et al. 2011). For fungal taxon determination, blastn searches based on ITS sequences were performed on the GenBank database (http://www.ncbi.nlm.nih.gov/genbank, Altschul et al. 1997), excluding uncultured/environmental sample sequences from the search. Results were interpreted critically, with emphasis on well-curated culture collections, such as the Westerdijk Fungal Biodiversity Collection (CBS). In general, species-level BLAST results below a threshold of 98% identity were not trusted to be accurate enough for final determination. Generally, morphological characteristics were used to confirm the results of molecular identification. Relative Species Abundance (RSA = number of disease cases associated with the particular fungus/number of disease cases) was given as the percentage of that particular fungus in all disease cases.
Pathogenicity tests
To confirm the pathogenicity of Botryosphaeriaceae strains isolated from damaged European beech tissues, inoculation tests were conducted according to Henle–Koch postulates (Evans 1976), in two experiments in a greenhouse located in Göttingen, following the method of Zlatković et al. (2019). The first test was conducted in summer 2019 (mean air temperature, 25 °C) and the second in autumn 2019 (mean air temperature, 11 °C). Pieces of 110, apparently healthy branches, were cut (70 in June and 40 in October) from approximately 50 closely spaced, healthy, mature European beech trees in a mixed forest stand in Dransfeld, close to Göttingen. Each piece was 110 cm long, ca. 1.5 cm in diameter at the basal end, and the trimmed shoot tips were sealed with wax. Ten branches per test were inoculated with a fungal strain (one-week-old culture), 70 cm from the basal end. A set of 10 untreated controls for each series was not inoculated at all, and mock controls were prepared by inoculating 10 branches with a sterile pure culture medium plug of MYP. The plugs were placed in wounds made with a sterile cork borer (5 mm diameter) and the removed bark was replaced on top of the inoculum then wrapped with Parafilm. During incubation, the test branches were maintained in buckets filled with tap water. Every second or third day, basal ends of the incubated branches were re-trimmed diagonally to increase water uptake and the water in the buckets was replaced. To keep them fresh, branches were sprayed with tap water daily until the end of the experiments. After three weeks, the bark was peeled away and the branches were cut longitudinally to detect and measure the extent of necroses. Fungi were re-isolated from the discoloured tissue and the resulting isolates were identified. Tested strains of Botryosphaeriaceae (Botryosphaeria dothidea = Fusicoccum aesculi Corda, Diplodia corticola A.J.L. Phillips, A. Alves & J. Luque ≡ Botryosphaeria corticola A.J.L. Phillips, A. Alves & J. Luque, Diplodia mutila (Fr.) Mont. = B. stevensii Shoemaker, and Diplodia sapinea (Fr.) Fuckel ≡ Sphaeropsis sapinea (Fr.) Dyko & B. Sutton) are listed in Table 1.
The significance of differences in necrosis length associated with differences in fungal taxa and temperatures was assessed using ANOVA and the Tukey HSD post hoc test. R version 3.6.2 (R Core Team 2019) was used for all data analyses.
Results
Disease cases and patterns
From 1 January 2018 to 31 December 2019, 2582 reports of forest damage due to VLB and heat stress were registered (Fig. 2). Most of these reports (2379) were registered in 2019, and only 203 in 2018. Across all the 55 investigated cases of VLB (Online Resource 1), 90% of affected trees exhibited leaking spots and bark necrosis (Fig. 1b–d). Cryptococcus fagisuga appeared to have a minor influence in the damage history, as it was detected in only 4% of cases. Bark beetles (Taphrorhychus bicolor Hrbst, Trypodendron domesticum L., Try. lineatum Olivier or Xylosandrus germanus Blandford) were found to be associated with 49% of the leaks investigated. In addition, A. viridis, timberworm beetles (Lymexyloidea) and longhorn beetles (Cerambycidae) were observed in 13% and 4% of the stands, respectively. We detected black (dead) bast and dead cambium in 42% of the disease cases, and wood staining by air inclusion or coloured heartwood in 44% (Fig. 1e). No signs of trophobiotic association of lachnids and forest ants were observed in any cases.
Associated fungi
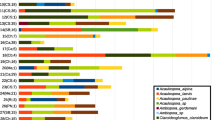
Species and higher-order taxa associated with the studied disease cases are listed in Table 2. For convenience, the percentages of cases in which an organism or phenomenon was observed are referred to hereafter as “Relative Species Abundance” (RSA).
In 51% of the diseased forest stands, bark necroses were associated with N. coccinea (Fig. 3a–b). According to our observations in the affected stands, ascocarps of N. coccinea were observed in all stages of VLB examined, especially in the early stages of the disease. In cases where leaking spots were not associated with a bark beetle, Botryosphaeriaceae species (Fig. 4, 30% RSA), N. coccinea (Fig. 3a, b) or both (Fig. 1d, 16% RSA) were isolated. In 42% of the disease cases, Eutypella quaternata (Pers.) Rappaz (Fig. 3c–d) was also associated with the bark necroses. Diplodia mutila (20% RSA, Fig. 4c–d) was isolated from diseased tissues, leaking spots and necrotic bark of mature European beech trees collected in six Hessian forest departments. Diplodia corticola (2% RSA, Fig. 4a–b) caused leaking spots and bark necroses in a mature stand in the forest department of Frankfurt, Hesse. Diplodia sapinea (2% RSA, Fig. 4f) was isolated together with D. mutila from the same necroses collected in Hesse. In addition, Diplodia spp. associated with necroses were detected in Hessen (4% RSA) but not identified due to fungal mortality.
Diplodia corticola. (a–b): a anamorph, b strain NW-FVA 4897, growing seven days on MYP-agar; Diplodia mutila (c–d): c anamorph with whitish, tendril of conidia, d strain NW-FVA4915, growing seven days on MYP-agar; e Botryosphaeria dothidea, strain NW-FVA5219, growing seven days on MYP-agar; e Diplodia sapinea, strain NW-FVA5932, growing 7 days on MYP-agar
Botryosphaeria dothidea (7% RSA, Fig. 4e) was found to be associated with bark necroses and die-off of European beech trees in pole stands in Saxony-Anhalt. Trees in these stands also exhibited bark necroses due to N. coccinea, dieback of shoots, mainly caused by Diaporthe sp., wood rot, bark blisters and ruptures caused by Biscogniauxia nummularia (Bull.) Kuntze (Fig. 5a–c), as well as root rot due to Armillaria spp. We could prove A. gallica Marxm. & Romagn. if we identified it to species level. Fructifications of Asterosporium asterospermum (Pers.) S. Hughes and Neohendersonia kickxii (Westend.) B. Sutton & Pollack projected from the dead tissues of branches and stems. Combinations of the aforementioned pathogenic fungi and decomposers in the affected trees led to very rapid disease progression and die-off within a few weeks. B. dothidea also occurred in Hesse in mature European beech stands, and ascocarps of the species were found on the bark of diseased trees.
Wood-decay fungi were observed in 58% of all disease cases. The following species were detected (as fruitbodies or isolated from diseased European beech tissue): Biscogniauxia mediterranea (De Not.) Kuntze (Fig. 5d, 5% RSA), B. nummularia (Fig. 5a–c, 33% RSA), Eutypa spinosa (Pers.) Tul. & C. Tul. (2% RSA), Diatrype stigma (Hoffm.) Fr. (4% RSA), Fomitopsis pinicola (Sw.) P. Karst. (2% RSA), Hypoxylon fragiforme (Pers.) J. Kickx f. (15% RSA), Kretschmaria deusta (Hoffm.) P.M.D. Martin (5% RSA), Schizophyllum commune Fr. (9% RSA, associated with sunburn), Pleurotus ostreatus (Jacq.) P. Kumm. (2% RSA) and Xylaria corniformis (Fr.) Fr (2% RSA). Additionally, affected trees were attacked by opportunistic soil-borne wood-rot fungi, for example, Armillaria spp. (27% RSA).
Bark blisters (Fig. 5b) and ruptures caused by the anamorph of B. nummularia were observed in 25% of all studied disease cases, mainly in 2019. Biscogniauxia mediterranea (5% RSA) was found in association with shoot dieback of beech in a pole stand surrounded by older oak.
Pathogenicity tests
Within three weeks of incubation at a mean air temperature of 25 °C (June, summer), D. corticola and D. mutila caused necroses, with positively correlated lengths and widths, on inoculated European beech branches (Table 1, Fig. 6). Lengths of necroses (n = 19), caused by the two different D. mutila strains, ranged from 25 to 215 mm (mean: 73 mm). Both strains (NW-FVA 4915 and 4997) did not differ significantly in respect to lesion length (see Fig. 6).
Necrosis caused by several Botryosphaeriaceae on European beech branches within 3 weeks, Boxplot of necrosis length (n = 10 branches per strain). Number indicates strain number, S = summer, A = autumn; no necrosis were evoked by Diplodia sapinea in summer and in the control group in autumn; Groups with different identification letters (a, b, c, d, e, f, g) differ significantly on the significance level of 0.05.
Variation in necrosis length was evident both in necrosis associated with different strains and in necrosis associated with the same strain. Lengths of necroses (n = 10) due to D. corticola varied from 15 to 173 mm (mean: 58 mm). No visible necroses were observed in any of the 20 branches inoculated (in June or October) with D. sapinea, or in any controls, except for a 39 mm long necrosis (from which H. fragiforme was isolated) in one of the mock controls. At a mean temperature of 11 °C (October, autumn), D. mutila strain NW-FVA 4915 caused necroses, but they were significantly smaller than in summer (mean: 18 mm; range 0–40 mm, n = 10 necroses, Fig. 6). Botryosphaeria dothidea also caused necrosis in October (mean length, 3 mm, range 0–8 mm, n = 10 necroses). The inoculated Botryosphaeria s. l. strains could be re-isolated from the treated beech tissue, regardless of whether necrosis had occurred. Neonectria coccinea was also isolated from the necrotic tissues of beech branches used in the infection test with B. dothidea, and in healthy tissues of mock controls.
Discussion
In the period covered by this study, VLB occurred mainly in three of the four considered federal states of Germany: Lower Saxony, Hesse and Saxony-Anhalt (Fig. 1). There was only a single VLB report from Schleswig–Holstein, which is the most oceanic state in the examined area and had the lowest precipitation deficit in 2018, compared to the long-term mean of the international reference period 1961–1990, and no deficit in 2019 (NW-FVA 2019; NW-FVA 2020). All the other disease cases were located in areas with a more continental climate, or relatively warm and dry regions with climate-sensitive vegetation, such as the Rhine valley, or areas with distinct precipitation deficits.
Among all fungal isolates from the studied diseased beech, N. coccinea was by far the most frequently observed (51% RSA). Several species of Botryosphaeriaceae (B. dothidea, D. corticola, D. mutila and D. sapinea) were associated with bark necroses in the studied VLB. These species are often endophytes (Alvarez-Loayza et al. 2008, 2011; Bußkamp et al. 2020) and, for example, B. dothidea and D. corticola are latent plant pathogens which have been frequently isolated from physiologically impaired oak trees (Panzavolta et al. 2017). Our pathogenicity tests proved that all tested Botryosphaeriaceae strains had capacity to cause necroses in beech. The results could be confirmed Langer and Bußkamp (2021) based on pathogenicity tests in planta with beech saplings.
We detected B. dothidea in both adult and pole beech stands. This is a latent pathogen of global importance to woody plant health, with more than 24 known host genera, both coniferous and deciduous (Crist and Schoeneweiss 1975; Smith and Stanosz 2001; Butin 2011; Piškur et al. 2011; Phillips et al. 2013; Abdollahzadeh et al. 2014; Marsberg et al. 2017). It has already been detected in Germany (Kehr 2004) and seems to be native to Europe (Piškur et al. 2011). Associated disease symptoms (branch and stem cankers, tip and branch dieback, fruit rot and blue stain) may lead to plant death (Marsberg et al. 2017). Botryosphaeria dothidea has been identified as the causal agent of the dieback of Ostrya carpinifolia Scop. following extreme weather conditions. (Piškur et al. 2011). The latter is also an opportunistic pathogen of Sequoiadendron giganteum (Lindl.) J.Buchholz, which can cause Botryosphaeria cankers after drought or extreme frost has affected the host trees (Cech and Tomiczek 2013). The ability of B. dothidea to colonise wood and bark and to form canker increases with decrease in water potentials (Crist and Schoeneweiss 1975). The prolonged latent infection or endophytic phase of B. dothidea in hosts enables it to cause disease symptoms when its hosts are stressed (Marsberg et al. 2017). Our preliminary inoculation tests, at a mean air temperature of 11 °C, confirmed its potential necrotic activity on European beech, and fulfilled Koch’s postulates.
The specimen of D. corticola collected near Frankfurt was the first record of this species in Germany and on beech worldwide. In the current standard list of fungal species of Germany, there is no entry for it. (Matzke-Hajek et al. 2016). At the time of the survey, there were no reports of the species causing damage to forest trees in Germany or other northern and central European countries. Diplodia corticola was first described from Portuguese samples of Quercus suber L. and can be distinguished from D. mutila by conidia size and DNA phylogeny (Alves et al. 2004). It has been previously detected in the Mediterranean area and North America (Alves et al. 2004; Dreaden et al. 2011; Smahi 2017). Diplodia corticola causes the so-called “Bot canker of oak” (Lynch et al. 2010; Dreaden et al. 2011), dieback of oak (Smahi 2017), grapevine cankers (Úrbez-Torres et al. 2010) and has been linked to oak decline (Linaldeddu et al. 2014; Panzavolta et al. 2017). Our inoculation tests have shown that D. corticola is pathogenic to F. sylvatica. Observations in forest samples also demonstrate its presence in German beech forests, but further research is needed to determine whether it is a native or invasive species in Germany.
Diplodia mutila is a plant pathogen that is present in many parts of the world, but its spread seems to be limited by low temperatures (CABI 2019). Its incidence in the Mediterranean region has increased in recent years (CABI 2019). It is widely distributed in Germany (HMUELV Hessen 2011; LFE 2016), but only sporadically found in northern Europe (CABI 2019). Several cases of disease associated with D. mutila have been reported in southern Europe, due to the predisposition of oaks to attack as a consequence of unfavourable climatic conditions, particularly drought (CABI 2019; Luisi et al. 1996; Ragazzi et al. 1995, 2000). In addition to endophytic and saprophytic growth, this fungus causes cankers, branch dieback, wilting, yellowing and death of leaves and stem necrosis in several tree species, and can lead to mortality of the entire host (Vajna 1986; Alves et al. 2004; Slippers and Wingfield 2007). Lack of water in the host tissue facilitates the transition to the parasitic stage of D. mutila. European host plants include European beech (LFE 2016), several other deciduous shrubs and trees (Graf and Manser 2000; HMUELV Hessen 2011; Kehr & Wulf 1993; Kuch et al. 2014; Luisi et al. 1996; Ragazzi et al. 1999, 2000) and grape (Lehoczky 1974). Preliminary inoculation tests in this study proved that D. mutila has a strong capacity to cause necrosis in beech, especially at the mean air temperature of 25 °C. Similarly, Kuch et al. (2014) found that the optimum temperature for a tested D. mutila strain was 25 °C.
Diplodia sapinea is a common endophyte of pine species in many parts of the world (Bihon et al. 2012; Langer et al. 2011) and may cause tip blight in pine or other conifers. It is widely distributed in conifers in Germany (Bußkamp et al. 2020) and the frequency of disease cases is increasing with global warming and drought combined with heat. In this study, the species was isolated from European beech for the first time in Germany. The affected beech stand in Hesse was adjacent to pine forests suffering from Diplodia tip blight. Whether or not D. sapinea is native to Europe is still unclear (Desprez-Loustau et al. 2009) and is the subject of discussion. (Bußkamp et al. 2020). The presence of D. sapinea on European beech reported here is the second recorded association of D. sapinea with woody tissues of F. sylvatica. The first was in the western Balkans (Zlatković et al. 2017), and it has also recently been found on Cork oak in Algeria (Smahi 2017). The strain we isolated did not cause necroses on beech branches in the preliminary inoculation tests, but was re-isolated from the infected plants. This remarkable observation clearly indicates that D. sapinea can survive in beech tissue for at least three weeks. In contrast, a D. sapinea strain originating from the western Balkans caused lesions on F. sylvatica in pathogenicity tests reported by Zlatković et al. (2017). However, the cited authors did not classify D. sapinea as a pathogen of European beech. Algerian S. sapinea strains tested by Smahi (2017) caused necrotic lesions on Q. suber, but these were significantly smaller than those caused by D. corticola.
In the VLB studied, the pathogenic bark fungi discussed above were followed by secondary pathogens and saprobes, mainly xylariaceous decomposers, including E. quaternata, B. nummularia, B. mediterranea, D. stigma and H. fragiforme. White-rot fungi, which act as saprophytes or opportunistic pathogens on dying or injured wood tissue, for instance, after sunburn, have also been detected. These included the Split-gill mushroom (S. commune) and Tree oyster mushroom (P. ostreatus). Schizophyllum commune also typically colonises trees stressed by heat and drought (Costello et al. 2011). The lack of occurrence of other basidiomycetous wood rotting fungi, such as Fomes fomentarius (L.) Fr., may be due to the early stage of the VLB (Langer 2019a; Langer et al. 2020).
Eutypa quaternata, which is typically found on dead branches of F. sylvatica (Vasilyeva and Scheuer 1996; Vasilyeva 2011), often occurred together with N. coccinea. In most cases, the teleomorph of E. quaternata appeared later than its anamorphic form (Fig. 3c).
It is not surprising that N. coccinea was often associated with devitalisation of beech, as it is one of the most common pathogens, together with H. fragiforme and B. nummularia, to develop latently in woody beech tissues (Chapela and Boddy 1988; Chapela 1989; Hendry et al. 2002). In an in planta pathogenicity test, Langer and Bußkamp (2021) showed that N. coccinea can cause necrosis in beech stems.
The main determinant for the initial development of B. nummularia, H. fragiforme and N. coccinea in beech xylem is the water content of the wood (Chapela and Boddy 1988), which was probably low in the trees affected by VLB, due to the high mean precipitation deficits in 2018 (NW-FVA 2019) in Lower Saxony (− 32.3%), Hesse (− 26.9%), Saxony-Anhalt (− 34.2%) and Schleswig–Holstein (− 26,4%). The pathogenic behaviour of N. coccinea may be facilitated by increases in oxygen levels, and decreases in nutrient availability (Rodríguez et al. 2011) and/or vigour of the host tree (Gonthier et al. 2005). The vigour of the trees affected by VLB was clearly impaired, and their oxygen content was probably increased, as evidenced by the bark cracking and wood discolouration caused by air inclusion. Thus, the pathogenic growth of N. coccinea is plausible. The increased growth and virulence of the wood-decay fungi B. nummularia, which is derived from inoculum latent in healthy living xylem tissue, was probably favoured by warm temperatures (25–30 °C) in summer 2019 (cf. Hendry et al. 2002). Biscogniauxia nummularia induced severe damage, such as strip-cankers and wood decay, in drought-stressed trees in 2018 and 2019, as described by Greenhalgh and Chesters (1968), Granata and Sidoti (2004), and Luchi et al. (2015).
Few attempts have been made to identify silvicultural measures that could prevent, reduce or mitigate infections with the pathogens described. However, the main drivers of VLB appear to be climatic, and according to Mausolf et al. (2018), the climate sensitivity of beech stands is strongly related to their management. They also found that trees in unmanaged stands were less sensitive to drought than those in managed stands, according to tree-ring analysis. Assemblages of wood-inhabiting fungi have also been shown to be related to the intensity of silvicultural management (Müller et al. 2007). Thus, management regimes that minimise the exposure of beech stems to intense solar radiation may at least help efforts to control VLB.
A further complication is that the frequency of drought stress in general may increase due to climate change, thereby increasing the threat posed by latent, opportunistic pathogens of European beech, such as B. dothidea, D. corticola, D. mutila, and N. coccinea. Climate change may also have highly uncertain effects on the role of D. sapinea in the course of VLB, which is far from fully understood. In this study, infections with Botryosphaeriaceae were only evident in stressed hosts and when disease symptoms were visible. As there is still insufficient knowledge of the pathogenicity of Botryosphaeriaceae on European beech, more and longer pathogenicity tests, preferably on living trees, are needed, together with a more comprehensive assessment of the likely effects of climate change.
References
Abdollahzadeh J, Hosseini F, Javadi A (2014) New records from Botryosphaeriaceae (Ascomycota) for mycobiota of Iran. Mycol Iran 1(1):43–51. https://doi.org/10.22043/mi.2014.4180
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein databases search programs. Nucl Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Alvarez-Loayza P, White JF Jr, Bergen M, Cadenas C (2008) Diplodia mutila causing seedling mortality of the palm Iriartea deltoidea. Plant Pathol 57(2):382–382. https://doi.org/10.1111/j.1365-3059.2007.01747.x
Alvarez-Loayza P, White J, Torres M, Balslev H, Kristiansen T, Svenning J-C, Gil N (2011) Light converts endosymbiotic fungus to pathogen, influencing seedling survival and niche-space filling of a common tropical tree Iriartea deltoidea. PLoS ONE 6:16386. https://doi.org/10.1371/journal.pone.0016386
Alves A, Correia A, Luque J, Phillips A (2004) Botryosphaeria corticola sp. nov. on Quercus species, with notes and description of Botryosphaeria stevensii and its anamorph Diplodia mutila. Mycologia 96(3):598–613
Ammer C, Albrecht L, Borchert H, Brosinger F, Dittmar C, Elling W, Ewald J, Felbermeier B, Gilsa H, Huss J, Kenk G, Kölling C, Kohnle U, Meyer P, Mosandl R, Moosmayer H-U, Palmer S, Reif A, Rehfuess KE, Stimm B et al (2005) Future suitability of beech (Fagus sylvatica L.) in Central Europe: critical remarks concerning a paper of Rennenberg et al. (2004). Allg Forst-Jagdztg 176:60–67
Barr ME (1987) Prodromus to Class Loculoascomycetes, Spi. Lubrecht & Cramer Ltd, Amherst
BGR (2020) Bundesanstalt für Geowissenschaften und Rohstoffe—Projekte—Bodenübersichtskarte 1:200.000 (BÜK200) Accessed 13 May 2020. https://www.bgr.bund.de/DE/Themen/Boden/Projekte/Informationsgrundlagen-laufend/BUEK200/BUEK200.html
Bihon W, Slippers B, Burgess T, Wingfield MJ, Wingfield BD (2012) Diverse sources of infection and cryptic recombination revealed in South African Diplodia pinea populations. Fungal Biol 116(1):112–120. https://doi.org/10.1016/j.funbio.2011.10.006
BKG (Bundesamt für Kartographie und Geodäsie) Digitales Geländemodell Gitterweite 25 m (DGM25). Accessed 7 Dec 2019. https://sg.geodatenzentrum.de/web_public/gdz/dokumentation/deu/dgm25.pdf
BMEL (2018) Holzmarktbericht2018.Accessed from. https://www.bmel-statistik.de/fileadmin/daten/FHB-0120005-2018.pdf
Bohn U, Neuhäusl R, Gollub G, Hettwer C, Neuhäuslová Z, Raus Th, Schlüter H, Weber H (2003) Karte der natürlichen vegetation europas/map of the natural vegetation of Europe. Maßstab/Scale 1:2500000., 2000/2003 edn. Landwirtschaftsverlag, Münster
Booth C (1971) The genus Fusarium C.A.B. Commonwealth Mycological Institute, Kew
Bressem U (2008) Komplexe Erkrankungen an Buche. Ergebnisse angewandter Forschung zur Buche. Universitätsdrucke Göttingen, Nordwestdeutsche Forstliche Versuchsanstalt, pp 101–121
Brodde L, Adamson K, Julio Camarero J, Castaño C, Drenkhan R, Lehtijärvi A, Luchi N, Migliorini D, Sánchez-Miranda Á, Stenlid J, Özdağ Ş, Oliva J (2019) Diplodia tip blight on its way to the north: drivers of disease emergence in Northern Europe. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01818
Brück-Dyckhoff C, Petercord R, Schopf R (2019) Vitality loss of European beech (Fagus sylvatica L.) and infestation by the European beech splendour beetle (Agrilus viridis L., Buprestidae, Coleoptera). For Ecol Manag 432:150–156. https://doi.org/10.1016/j.foreco.2018.09.001
Bußkamp J, Langer GJ, Langer EJ (2020) Sphaeropsis sapinea and fungal endophyte diversity in twigs of Scots pine (Pinus sylvestris) in Germany. Mycol Prog 19:985–999. https://doi.org/10.1007/s11557-020-01617-0
Butin H (2011) Krankheiten der wald- und parkbäume—diagnose, biologie, bekämpfung. Eugen Ulmer, Stuttgart
CABI (2019) Botryosphaeria stevensii (Botryosphaeria disease, grapevine). CABI Compendium, Wallingford
Cech TL, Tomiczek C (2013) Botryosphaeria-Krebs (Botryosphaeria dothidea) des Mammutbaumes nimmt in Ostösterreich zu. Forstsch Aktuell 57(58):37–40
Chapela IH (1989) Fungi in healthy stems and branches of American beech and aspen: a comparative study. New Phytol 113(1):65–75. https://doi.org/10.1111/j.1469-8137.1989.tb02396.x
Chapela IH, Boddy L (1988) Fungal colonization of attached beech branches II. Spatial and temporal organization of communities arising from latent invaders in bark and functional sapwood, under different moisture regimes. New Phytol 110(1):47–57. https://doi.org/10.1111/j.1469-8137.1988.tb00236.x
Costello LR, Hagen BW, Jones KS (2011) Oaks in the Urban Landscape: Selection, Care, and Preservation. UCANR Publications
Crist C, Schoeneweiss D (1975) The influence of controlled stresses on susceptibility of European white birch stems to attack by Botryosphaeria dothidea. Phytopathology 65(4):369–373
Desprez-Loustau M-L, Courtecuisse R, Robin C, Husson C, Moreau P-A, Blancard D, Selosse M-A, Lung-Escarmant B, Piou D, Sache I (2009) Species diversity and drivers of spread of alien fungi (sensu lato) in Europe with a particular focus on France. Biol Invasions 12(1):157–172
Domsch KH, Gams W, Anderson TH (1980) Compendium of soil fungi Volume 1. Academic Press, London
Dreaden TJ, Shin K, Smith JA (2011) First report of Diplodia corticola causing branch cankers on live Oak (Quercus virginiana) in Florida. Plant Dis 95(8):1027–1027. https://doi.org/10.1094/PDIS-02-11-0123
DWD (2019) Wetter und Klima—Deutscher Wetterdienst—Archiv 2019 - Deutschlandwetter im April 2019. In: Pressemitteilungen. Accessed 8 Aug 2019. https://www.dwd.de/DE/presse/pressemitteilungen/DE/2019/20190429_deutschlandwetter_april.html?nn=656450
Ehrlich J (1934) The beech bark disease: a Nectria disease of Fagus, following Cryptococcus fagi (Baer.). Can J Res 10(6):593–692
Ellenberg H (1996) Vegetation mitteleuropas mit den alpen, 5th edn. Ulmer Verlag, Stuttgart
Eriksson O (1981) The families of bitunicate ascomycetes. Nord J Bot 1(6):800–800. https://doi.org/10.1111/j.1756-1051.1981.tb01167.x
Evans AS (1976) Causation and disease: the henle-koch postulates revisited. Yale J Biol Med 49(2):175–195
Fabre B, Piou D, Desprez-Loustau M-L, Marçais B (2011) Can the emergence of pine Diplodia shoot blight in France be explained by changes in pathogen pressure linked to climate change? Glob Change Biol 17(10):3218–3227. https://doi.org/10.1111/j.1365-2486.2011.02428.x
Gerlach N, Nirenberg H (1982) Nr. 209 (1982): The genus Fusarium—a pictorial atlas
Gonthier P, Gennaro M, Nicolotti G (2005) Effects of water stress on the endophytic mycota of Quercus robur. Fungal Divers 21:69–80
Graf E, Manser P (2000) Beitrag zum eingeschleppten Schwarzen Nutzholzborkenkäfer Xylosandrus germanus. Biologie und Schadenpotential an im Wald gelagertem Rundholz im Vergleich zu Xyloterus lineatus und Hylecoetus dermestoides. Schweiz Z Forstw 151:271–281
Granata G, Sidoti A (2004) Biscogniauxia nummularia: pathogenic agent of a beech decline. For Pathol 34(6):363–367. https://doi.org/10.1111/j.1439-0329.2004.00377.x
Greenhalgh GN, Chesters CGC (1968) Conidiophore morphology in some British members of the Xylariaceae. Trans Br Mycol Soc 51(1):57–36. https://doi.org/10.1016/S0007-1536(68)80122-6
Grüner J, Metzler B (2006) Nectria-Arten an Buchenrinde mit Phloemnekrosen. In: Strategien zur Sicherung von Buchenwäldern, pp 129–138
Hartig R (1878) Die krebsartigen Krankheiten der Rotbuche. Z Forst- Jagdwes 9:377–383
Hartmann G, Nienhaus F, Butin H (2007) Farbatlas Waldschäden -, 3rd edn. Ulmer
Hendry SJ, Boddy L, Lonsdale D (2002) Abiotic variables effect differential expression of latent infections in beech (Fagus sylvatica). New Phytol 155(3):449–460. https://doi.org/10.1046/j.1469-8137.2002.00473.x
HMUELV Hessen (2011) Rindenbrand bei Apfel. Hessisches Ministerium für Umwelt, Klimaschutz, Landwirtschaft und Verbraucherschutz
Izumitsu K, Hatoh K, Sumita T, Kitade Y, Morita A, Tanaka C, Gafur A, Ohta A, Kawai M, Yamanaka T, Neda H, Ota Y (2012) Rapid and simple preparation of mushroom DNA directly from colonies and fruiting bodies for PCR. Mycoscience 53(5):396–401
Jung T (2009) Beech decline in Central Europe driven by the interaction between Phytophthora infections and climatic extremes. For Pathol 39(2):73–94. https://doi.org/10.1111/j.1439-0329.2008.00566.x
Kehr R (2004) Triebschäden an Mammutbaum (Sequoiadendron giganteum) durch Botryosphaeria dothidea auch in Deutschland nachgewiesen. Nachrichtenblatt Dtsch Pflanzenschutzdienstes 56(2):37–37
Kehr RD, Wulf A (1993) Fungi associated with above-ground portions of declining oaks (Quercus robur) in Germany. Eur J for Pathol 23(1):18–27. https://doi.org/10.1111/j.1439-0329.1993.tb00803.x
Kuch J, Cech TL, Konrad H, Bedlan G (2014) Erstnachweis von Diplodia mutila an Ligustrum vulgare – Beiträge zur Taxonomie von Botryosphaeria stevensii Shoemaker. J Für Kult 66(4):136–143
Langer GJ (1994) Die Gattung Botryobasidium DONK (Corticiaceae, Basidiomycetes). J Cramer, Stuttgart
Langer GJ (2019a) Komplexe Erkrankungen bei älteren Rotbuchen. AFZ-Wald 24:32–35
Langer GJ (2019b) Warum schwinden den Buchen die Kräfte? Land Forst 35:3
Langer GJ, Bußkamp J (2021) Fungi associated with woody tissues of European beech and their impact on tree health. Front Microbiol 12:702467
Langer GJ, Bressem U, Habermann M (2011) Diplodia-Triebsterben der Kiefer in Blattwespenfraßgebieten der Letzlinger Heide. AFZ 11:28–31
Langer GJ, Bußkamp J, Langer EJ (2020) Absterbeerscheinungen bei Rotbuche durch Wärme und Trockenheit. AFZ-Wald 4(2020):24–27
Lee I-S, Langer E (2012) New records of Hyphodontia species from Taiwan. Nova Hedwig 94(1–2):239–244. https://doi.org/10.1127/0029-5035/2012/0094-0239
Lehoczky J (1974) Black dead-arm disease of grapevine caused by Botryosphaeria stevensii infection. Acta Phytopathol Acadamiae Sci Hung 9:319–327
Leuschner C (2020) Drought response of European beech (Fagus sylvatica L.)—a review. Perspect Plant Ecol Evol Syst 47:125576. https://doi.org/10.1016/j.ppees.2020.125576
LFE (2016) Diagnose Report—Diagnostische Arbeiten unter besonderer Berücksichtigung pilzlicher Organismen. Produkt: Waldschutzberatung (222030). Landeskompetenzzentrum Forst Eberswalde (LFE)
Linaldeddu BT, Scanu B, Maddau L, Franceschini A (2014) Diplodia corticola and Phytophthora cinnamomi: the main pathogens involved in holm oak decline on Caprera Island (Italy). For Pathol 44(3):191–200. https://doi.org/10.1111/efp.12081
Liu J-K, Phookamsak R, Doilom M, Wikee S, Li Y-M, Ariyawansa H, Boonmee S, Chomnunti P, Dai D-Q, Bhat DJ, Romero A, Zhuang W-Y, Monkai J, Jones E, Chukeatirote E, Ko Ko T, Zhao Y, Wang Y, Hyde K (2012) Towards a natural classification of Botryosphaeriales. Fungal Divers 57:149–210. https://doi.org/10.1007/s13225-012-0207-4
Luchi N, Capretti P, Feducci M, Vannini A, Ceccarelli B, Vettraino AM (2015) Latent infection of Biscogniauxia nummularia in Fagus sylvatica: a possible bioindicator of beech health conditions. Iforest Biogeosciences for 9:e1–e6. https://doi.org/10.3832/ifor1436-008
Luisi N, Lerario P, Bianco MC (1996) Botryosphaeria stevensii: pathogenicity on oaks and phytotoxic effects. Ital for E Mont 51(4):250–263
Lynch SC, Eskalen A, Zambino P, Scott T (2010) First Report of bot canker caused by Diplodia corticola on coast live Oak (Quercus agrifolia) in California. Plant Dis 94(12):1510–1510. https://doi.org/10.1094/PDIS-04-10-0266
Manion PD (1981) Tree disease concepts. Prentice-Hall, Hoboken
Marsberg A, Kemler M, Jami F, Nagel J, Postma A, Naidoo S, Wingfield MJ, Crous P, Spatafora JW, Hesse CN, Robbertse B, Slippers B (2017) Botryosphaeria dothidea : a latent pathogen of global importance to woody plant health. Mol Plant Pathol 18(4):477–488. https://doi.org/10.1111/mpp.1249
Martinez Del Castillo E, Zang CS, Buras A, Hacket-Pain A, Esper J, Serrano-Notivoli R, Hartl C, Weigel R, Klesse S, Resco de Dios V, Scharnweber T, Dorado-Liñán I, van der Maaten-Theunissen M, van der Maaten E, Jump A, Mikac S, Banzragch BE, Beck W, Cavin L, Claessens H, Čada V, Čufar K, Dulamsuren C, Gričar J, Gil-Pelegrín E, Janda P, Kazimirovic M, Kreyling J, Latte N, Leuschner C, Longares LA, Menzel A, Merela M, Motta R, Muffler L, Nola P, Petritan AM, Petritan IC, Prislan P, Rubio-Cuadrado Á, Rydval M, Stajić B, Svoboda M, Toromani E, Trotsiuk V, Wilmking M, Zlatanov T, de Luis M (2022) Climate-change-driven growth decline of European beech forests. Commun Biol 5(1):163. https://doi.org/10.1038/s42003-022-03107-3
Matzke-Hajek G, Hofbauer N, Bedlan G (2016) Rote Liste gefährdeter Tiere, Pflanzen und Pilze Deutschlands - Bd 8: Pilze (Teil 1)-Großpilze. NaBiV Heft 708 Bundesamt Für Naturschutz Bonn-Bad Godesberg 70(8):440
Mausolf K, Wilm P, Härdtle W, Jansen K, Schuldt B, Sturm K, von Oheimb G, Hertel D, Leuschner C, Fichtner A (2018) Higher drought sensitivity of radial growth of European beech in managed than in unmanaged forests. Sci Total Environ 642:1201–1208. https://doi.org/10.1016/j.scitotenv.2018.06.065
McIntosh C (1849) Disease in beech trees. North Br Agric J Hortic 1:409
Mehl JWM, Slippers B, Roux J, Wingfield MJ (2013) Cankers and other diseases caused by the Botryosphaeriaceae. In: Gonthier P, Nicolotti G (eds) Infectious forest diseases. CABI, Wallingford, pp 298–317
Müller J, Engel H, Blaschke M (2007) Assemblages of wood-inhabiting fungi related to silvicultural management intensity in beech forests in southern Germany. Eur J for Res 126:513–527. https://doi.org/10.1007/s10342-007-0173-7
Neycken A, Scheggia M, Bigler C, Lévesque M (2022) Long-term growth decline precedes sudden crown dieback of European beech. Agric for Meteorol 324:109103. https://doi.org/10.1016/j.agrformet.2022.109103
Niesar M (2007) Symptom- und Ursachenanalyse der aktuellen Buchenerkrankung in höheren Lagen von Nordrhein-Westfalen Georg-August-Universität zu Göttingen. Cuvillier Verlag, Göttingen
NOAA (2019) National Centers for Environmental Information, State of the Climate: Global Climate Report for Annual 2018. Accessed from 23 Oct 2019. https://www.ncdc.noaa.gov/sotc/global/201813
NW-FVA (2020) Waldschutzinfo Nr. 1/2020-Witterung 2019. Nordwestdeutsche Forstliche Versuchsanstalt, Göttingen
NW-FVA, (2019) Waldschutzinfo Nr 1/ 2019 - Witterung 2018. Nordwestdeutsche Forstliche Versuchsanstalt, Göttingen
Oertel B (2003) Bibliographische Recherchen in der Mykologie. Ein Leitfaden für Leser im deutschsprachigen Raum und darüber hinaus. Z MYKOL 69(1):3–42
Panzavolta T, Panichi A, Bracalini M, Croci F, Ginetti B, Ragazzi A, Tiberi R, Moricca S (2017) DispeRSAl and propagule pressure of Botryosphaeriaceae species in a declining oak stand is affected by insect vectors. Forests 8(7):228
Pflug EE, Buchmann N, Siegwolf RTW, Schaub M, Rigling A, Arend M (2018) Resilient leaf physiological response of european beech (Fagus sylvatica L.) to summer drought and drought release. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00187
Phillips AJL, Alves A, Pennycook SR, Johnston PR, Ramaley A, Akulov A, Crous PW (2008) Resolving the phylogenetic and taxonomic status of dark-spored teleomorph genera in the Botryosphaeriaceae. Persoonia Mol Phylogeny Evol Fungi 21:29–55. https://doi.org/10.3767/003158508X340742
Phillips AJL, Alves A, Abdollahzadeh J, Slippers B, Wingfield MJ, Groenewald JZ, Crous PW (2013) The Botryosphaeriaceae: genera and species known from culture. Stud Mycol 76:51–167
Piškur B, Pavlic D, Slippers B, Ogris N, Maresi G, Wingfield MJ, Jurc D (2011) Diversity and pathogenicity of Botryosphaeriaceae on declining Ostrya carpinifolia in Slovenia and Italy following extreme weather conditions. Eur J for Res 130(2):235–249. https://doi.org/10.1007/s10342-010-0424-x
Purahong W, Tanunchai B, Wahdan SFM, Buscot F, Schulze E-D (2021) Molecular screening of microorganisms associated with discolored wood in dead european beech trees suffered from extreme drought event using next generation sequencing. Plants 10(10):2092. https://doi.org/10.3390/plants10102092
R Core Team (ed) (2019) R Core Team (2019). Eur Environ Agency
Ragazzi A, Vagniluca S, Moricca S, Vigniluca S (1995) European expansion of oak decline: involved microrganisms and methodological approaches. Phytopathol Mediterr 34(3):207–226
Ragazzi A, Dellavalle I, Moricca S, Capretti P, Raddi P (eds) (2000). Decline of oak species in Italy. Problems and perspectives. Accademia Italiana di Scienzi Forestali, Firenze
Ragazzi A, Moricca S, Capretti P, Dellavalle I (1999) Endophytic presence of Discula quercina on Declining Quercus cerris. J Phytopathol 147:437–440. https://doi.org/10.1111/j.1439-0434.1999.tb03847.x
Rennenberg H, Seiler W, Matyssek R, Gessler A, Kreuzwieser J (2004) European beech (Fagus sylvatica L.)—a forest tree without future in the south of Central Europe? Allg Forst Jagdztg 175:210–224
Rodríguez J, Elissetche J, Valenzuela S (2011) Tree endophytes and wood biodegradation. Springer, Dordrecht, pp 81–93
Shoemaker RA (1964) Conidial states of some Botryosphaeria species on Vitis and Quercus. Can J Bot 42(9):1297–1301. https://doi.org/10.1139/b64-122
Sieber TN (2007) Endophytic fungi in forest trees: are they mutualists? Fungal Biol Rev 21(2):75–89. https://doi.org/10.1016/j.fbr.2007.05.004
Sivanesan A (1984) The bitunicate ascomycetes and their anamorphs. J Cramer, Vaduz
Slippers B, Wingfield MJ (2007) Botryosphaeriaceae as endophytes and latent pathogens of woody plants: diversity, ecology and impact. Fungal Biol Rev 21(2):90–106. https://doi.org/10.1016/j.fbr.2007.06.002
Slippers B, Crous PW, Denman S, Coutinho TA, Wingfield BD, Wingfield MJ (2004) Combined multiple gene genealogies and phenotypic characters differentiate several species previously identified as Botryosphaeria dothidea. Mycologia 96:83–101
Smahi H (2017) Molecular characterization and pathogenicity of Diplodia corticola and other Botryosphaeriaceae species associated with canker and dieback of Quercus suber in Algeria. Mycosphere 8(2):1261–1272. https://doi.org/10.5943/mycosphere/8/2/10
Smith D, Stanosz G (2001) Molecular and morphological differentiation of Botryosphaeria dothidea (Anamorph Fusicoccum aesculi) from some other fungi with Fusicoccum Anamorphs. Mycologia 93:505. https://doi.org/10.2307/3761736
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. https://doi.org/10.1093/molbev/msr121
Tüxen R, Preising E (1956) Die heutige potentielle natürliche Vegetation als Gegenstand der Vegetationskartierung: mit 10 Tabellen. Zentralstelle für Vegetationskartierung
Úrbez-Torres JR, Peduto F, Rooney-Latham S, Gubler WD (2010) First report of Diplodia corticola causing grapevine (Vitis vinifera) cankers and trunk cankers and dieback of Canyon live oak (Quercus chrysolepis) in California. Plant Dis 94(6):785
Vajna L (1986) Branch canker and dieback of sessile oak (Quercus petraea) in Hungary caused by Diplodia mutila. 1: identification of the pathogen. Eur J for Pathol Ger FR 16:223–229
Vasilyeva L (2011) Quaternaria carpinicola, a comb. nov. (Diatrypaceae). Mycosphere 2(4):515–517
Vasilyeva L, Scheuer C (1996) Neuere Aufsammlungen stromatischer Pyrenomyceten aus Österreich, insbesondere der Steiermark: 61–82
von Arx JA (1981) The genera of fungi sporulating in pure culture, 3rd edn. Cramer, Vaduz
von Arx JA, Müller E (1954) Die Gattungen der amerosporen Pyrenomyceten. Beitr Kryptogamenflora Schweiz 11(1):1–434
von Arx JA, Müller E (1975) A re-evaluation of the bitunicate Ascomycetes with keys to families and genera. Centraalbureau voor Schimmelcultures, Baar
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA Genes for phylogenetics. PCR Protoc Guide Methods Appl 18:315–322
Wohlgemuth T, Kistler M, Aymon C, Hagedorn F, Gessler A, Gossner MM, Querloz V, Vögtli I, Wasem U, Vitasse Y, Rigling A (2020) Früher Laubfall der Buche während der Sommertrockenheit 2018: Resistenz oder Schächesymptom. Schweiz Z Forstwes 171(5):257–269
Zlatković M, Keča N, Wingfield MJ, Jami F, Slippers B (2017) New and unexpected host associations for Diplodia sapinea in the Western Balkans. For Pathol 47(3):12328. https://doi.org/10.1111/efp.12328
Zlatković M, Wingfield MJ, Jami F, Slippers B (2018) Host specificity of co-infecting Botryosphaeriaceae on ornamental and forest trees in the Western Balkans. For Pathol 48(2):e12410. https://doi.org/10.1111/efp.12410
Zlatković M, Wingfield MJ, Jami F, Slippers B (2019) Genetic uniformity characterises the invasive spread of Neofusicoccum parvum and Diplodia sapinea in the Western Balkans. For Pathol 49(1):e12491. https://doi.org/10.1111/efp.12491
Acknowledgements
The authors are grateful for the technical support from Annette Ihlemann, Martina Hille, Kerstin Herwig, Etta Starick, Ursula Rabel and Peter Gawehn (NW-FVA) as well as from Ulrike Frieling, Sylvia Heinemann (University of Kassel). We also gratefully acknowledge the linguistic improvement of the manuscript by native speaker Robert Larkin (NW-FVA), Sees-editing Ltd, and https://www.deepl.com / DeepL Write (free version).
Funding
Open Access funding enabled and organized by Projekt DEAL. The study was performed in preparation for and during the implementation of the Buche-Akut project. The latter project receives funding via the Waldklimafonds (WKF) funded by the German Federal Ministry of Food and Agriculture (BMEL) and Federal Ministry for the Environment, Nature Conservation, Nuclear Safety and Consumer Protection (BMUV) administrated by the Agency for Renewable Resources (FNR) under grant agreements No. 2220WK10B1.
Author information
Authors and Affiliations
Contributions
The first draft of the manuscript was written by Gitta Langer and Johanna Bußkamp added substantially to the manuscript. Both authors contributed equally to the study and approved the submitted version. The expertise of Robert Larkin, as a native English speaker, Sees-editing Ltd, and https://www.deepl.com / DeepL Write (free version) was used to improve the English language and grammar of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest. There are only non-financial research interests, related directly or indirectly to this work submitted for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Langer, G.J., Bußkamp, J. Vitality loss of beech: a serious threat to Fagus sylvatica in Germany in the context of global warming. J Plant Dis Prot 130, 1101–1115 (2023). https://doi.org/10.1007/s41348-023-00743-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-023-00743-7