Abstract
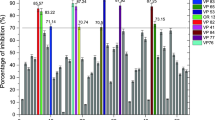
Antioxidant compounds were produced from submerged fermentation using Botryosphaeria dothidea. Sonication times (1–15 min) and agitations (0–150 rpm) applied to different fermentation phases (2–7 days) were evaluated for their effect on the broth antioxidant activity. In approximately 70% of assays performed with ultrasound, the antioxidant activity of the broth containing exocellular metabolites was higher than the control (without sonication), reaching a maximum value of 96%. The solid fraction (biomass after supernatant removal) corresponding to the best assay of antioxidant activity was submitted to extractions with water, ethanol or ethyl acetate to recover intracellular metabolites. The yields of extracts were 3.7 ± 2.6 wt %, 20.6 ± 3.9 wt % and 32.0 ± 4.2 wt % for ethyl acetate, ethanol and water, respectively. The use of ethanol could provide a larger number of compounds such as pentadecanoic acid, ergosta-5,8,22-trien-3-ol, and 1,4-diaza-2,5-dioxobicyclo [4.3.0] nonane. Both the supernatant and solid fractions revealed some bioactive compounds with promising antioxidant activity with the potential to replace some synthetic antioxidants.



Similar content being viewed by others
References
Abaide ER, Zabot GL, Tres MV, Martins RF, Fagundez JL, Nunes LF, Druzian S, Soares JF, Dal Prá V, Silva JRF, Kuhn RC, Mazutti MA (2017) Yield, composition, and antioxidant activity of avocado pulp oil extracted by pressurized fluids. Food Bioprod Process 102:289–298. https://doi.org/10.1016/j.fbp.2017.01.008
Alara OR, Abdurahman NH, Ukaegbu CIJ (2018) Soxhlet extraction of phenolic compounds from Vernonia cinerea leaves and its antioxidant activity. J Appl Res Med Aromat Plants 11:12–17. https://doi.org/10.1016/j.jarmap.2018.07.003
Alves CQ, David JM, David JP, Bahia MV, Aguiar RM (2010) Methods for determination of in vitro antioxidant activity for extracts and organic compounds. Quim Nova 33:2202–2221. https://doi.org/10.1590/S0100-40422010001000033
Avhad DN, Niphadkar SS, Rathod VK (2014) Ultrasound assisted three phase partitioning of a fibrinolytic enzyme. Ultrasonon Sonochem 21:628–633. https://doi.org/10.1016/j.ultsonch.2013.10.002
Bayar N, Bouallegue T, Achour M, Kriaa M, Bougatef A, Kammoun R (2017) Ultrasonic extraction of pectin from Opuntia ficus indica cladodes after mucilage removal: optimization of experimental conditions and evaluation of chemical and functional properties. Food Chem 235:275–282. https://doi.org/10.1016/j.foodchem.2017.05.029
Bhattacharya M, Srivastav PP, Mishra HN (2014) Optimization of process variables for supercritical fluid extraction of ergothioneine and polyphenols from Pleurotus ostreatus and correlation to free-radical scavenging activity. J Supercrit Fluids 95:51–59. https://doi.org/10.1016/j.supflu.2014.07.031
Confortin TC, Todero I, Soares JF, Brun T, Luft L, Ugalde GA, Prá VD, Mazutti MA, Zabot GL, Tres MV (2017) Extraction and composition of extracts obtained from Lupinus albescens using supercritical carbon dioxide and compressed liquefied petroleum gas. J Supercrit Fluids 128:395–403. https://doi.org/10.1016/j.supflu.2017.06.006
Corazza GO, Bilibio D, Zanella O, Luis-Nunes A, Paulo-Bender J, Carniel N, dos Santos PP, Priamo WL (2018) Pressurized liquid extraction of polyphenols from Goldenberry: Influence on antioxidant activity and chemical composition. Food and Bioproducts Processing 112:63–68. https://doi.org/10.1016/j.fbp.2018.09.001
Dalagnol LMG, Dal Magro L, Silveira VCC, Rodrigues E, Manfroi V, Rodrigues RC (2017) Combination of ultrasound, enzymes and mechanical stirring: a new method to improve Vitis vinifera Cabernet Sauvignon must yield, quality and bioactive compounds. Food Bioprod Process 105:197–204. https://doi.org/10.1016/j.fbp.2017.07.009
De Meneses AC, Machado TO, Sayer C, Araújo PHH, De Oliveira D (2018) Development of antioxidant poly(thioether-ester) nanoparticles. Braz J Chem Eng 35:691–697. https://doi.org/10.1590/0104-6632.20180352s20160605
De Souza ARC, Baldoni DB, Lima J, Porto V, Marcuz C, Ferraz RC, Kuhn RC, Jacques RJS, Guedes JVC, Mazutti MA (2015) Bioherbicide production by Diaporthe sp. isolated from the Brazilian Pampa biome. Biocatal Agric Biotechnol 4:575–578. https://doi.org/10.1016/j.bcab.2015.09.005
de Valente IL, Confortin TC, Luft L, Ugalde GA, Zabot GL, Mazutti MA, de Terra LM (2018) Extraction of bioactive compounds from Botryosphaeria dothidea using supercritical carbon dioxide and compressed liquefied petroleum gas. J Supercrit Fluids 136:52–59. https://doi.org/10.1016/j.supflu.2018.02.013
Dong H, Zhang Q, Li L, Liu J, Shen L, Li H, Qin W (2015) Antioxidant activity and chemical compositions of essential oil and ethanol extract of Chuanminshen violaceum. Ind Crops Prod 76:290–297. https://doi.org/10.1016/j.indcrop.2015.04.051
Dutta SC, Neog B (2016) Accumulation of secondary metabolites in response to antioxidant activity of turmeric rhizomes co-inoculated with native arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria. Sci Hortic 204:179–184. https://doi.org/10.1016/j.scienta.2016.03.028
Favareto R, Teixeira MB, Soares FAL, Belisário CM, Cabral JF, Da Silva EA, Moia TA, Cardozo-Filho L (2019) Extraction of bioactive compounds of leaves of Duguetia furfuracea (annonaceae) using green and organic solvents. Braz J Chem Eng J 36:549–556. https://doi.org/10.1590/0104-6632.20190361s20170451
Garmus TT, Kopf SFM, Paula JT, Aguiar AC, Duarte GHB, Eberlin MN, Cabral FA (2019) Ethanolic and hydroalcoholic extracts of pitanga leaves (Eugenia uniflora L.) and their fractionation by supercritical technology. Braz J Chem Eng J 36:1041–1051. https://doi.org/10.1590/0104-6632.20190362s20180159
Jardim C, Macedo D, Figueira I, Dobson G, McDougall GJ, Stewart D, Ferreira RB, Menezes R, Santos CN (2017) (Poly)phenol metabolites from Arbutus unedo leaves protect yeast from oxidative injury by activation of antioxidant and protein clearance pathways. J Funct Foods 32:333–346. https://doi.org/10.1016/j.jff.2017.03.010
Kulkarni VM, Rathod VK (2015) A novel method to augment extraction of mangiferin by application of microwave on three phase partitioning. Biotechnol Rep 6:8–12. https://doi.org/10.1016/j.btre.2014.12.009
Kumar S, Yadav M, Yadav A, Yadav JP (2017) Impact of spatial and climatic conditions on phytochemical diversity and in vitro antioxidant activity of Indian Aloe vera (L.) Burm.f. S Afr J Botany 111:50–59. https://doi.org/10.1016/j.sajb.2017.03.012
Kwiatkowska B, Bennett J, Akunna J, Walker GM, Bremner DH (2011) Stimulation of bioprocesses by ultrasound. Biotechnol Adv 29:768–780. https://doi.org/10.1016/j.biotechadv.2011.06.005
Li GQ, Liu FF, Li JQ, Liu QL, Chen SF (2016) Characterization of Botryosphaeria dothidea and Lasiodiplodia pseudotheobromae from English Walnut in China. J Phytopathol 164:348–353. https://doi.org/10.1111/jph.12422
Liu R, Li W, Sun LY, Liu CZ (2012) Improving root growth and cichoric acid derivatives production in hairy root culture of Echinacea purpurea by ultrasound treatment. Biochem Eng J 60:62–66. https://doi.org/10.1016/j.bej.2011.10.001
Liu L, Xu P, Zeng G, Huang D, Zhao M, Lai C, Chen M, Li N, Huang C, Wang C, Cheng M, He X, Lai M, He Y (2014) Inherent antioxidant activity and high yield production of antioxidants in Phanerochaete chrysosporium. Biochem Eng J 90:245–254. https://doi.org/10.1016/j.bej.2014.06.014
Maiti S, Sarma SJ, Brar SK, Le Bihan Y, Drogui P, Buelna G, Verma M (2016) Agro-industrial wastes as feedstock for sustainable bio-production of butanol by Clostridium beijerinckii. Food Bioprod Process 98:217–226. https://doi.org/10.1016/j.fbp.2016.01.002
Mazzutti S, Ferreira SRS, Riehl CAS, Smania A, Smania FA, Martínez J (2012) Supercritical fluid extraction of Agaricus brasiliensis: antioxidant and antimicrobial activities. J Supercrit Fluids 70:48–56. https://doi.org/10.1016/j.supflu.2012.06.010
Mehl JWM, Slippers B, Roux J, Wingfield MJ (2017) Overlap of latent pathogens in the Botryosphaeriaceae on a native and agricultural host. Fungal Biology 121:405–419. https://doi.org/10.1016/j.funbio.2016.07.015
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Moo-Huchin VM, Moo-Huchin MI, Estrada-León RJ, Cuevas-Glory L, Estrada-Mota IA, Ortiz-Vázquez E, Betancur-Ancona D, Sauri-Duch E (2015) Antioxidant compounds, antioxidant activity and phenolic content in peel from three tropical fruits from Yucatan, Mexico. Food Chem 166:17–22. https://doi.org/10.1016/j.foodchem.2014.05.127
Naveena B, Armshaw P, Tony Pembroke J (2015) Ultrasonic intensification as a tool for enhanced microbial biofuel yields. Biotechnol Biofuels 8:1–13. https://doi.org/10.1186/s13068-015-0321-0
Parveen R, Shamsi TN, Singh G, Athar T, Fatima S (2018) Phytochemical analysis and in-vitro biochemical characterization of aqueous and methanolic extract of Triphala, a conventional herbal remedy. Biotechnol Rep 17:126–136. https://doi.org/10.1016/j.btre.2018.02.003
Patial PK, Sharma A, Kaur I, Cannoo DS (2019) Correlation study among the extraction techniques, phytochemicals, and antioxidant activity of Nepeta spicata aerial part. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2019.101275
Poodi Y, Bimakr M, Ganjloo A, Zarringhalami S (2018) Intensification of bioactive compounds extraction from Feijoa (Feijoa sellowiana Berg.) leaves using ultrasonic waves. Food Bioprod Process 108:37–50. https://doi.org/10.1016/j.fbp.2017.12.004
Rodrigues S, Fernandes FAN, de Brito ES, Sousa AD, Narain N (2015) Ultrasound extraction of phenolics and anthocyanins from jabuticaba peel. Ind Crops Prod 69:400–407. https://doi.org/10.1016/j.indcrop.2015.02.059
Samaga PV, Rai VR (2016) Diversity and bioactive potential of endophytic fungi from Nothapodytes foetida, Hypericum mysorense and Hypericum japonicum collected from Western Ghats of India. Ann Microbiol 66:229–244. https://doi.org/10.1007/s13213-015-1099-9
Sánchez-Carvajal AL, Alatorre-Santamaría S, Valerio-Alfaro G, Hérnández-Vázquez L, Navarro-Ocaña A (2018) Waste residues from Opuntia ficus indica for peroxidase-mediated preparation of phenolic dimeric compounds. Biotechnol Reps 20:e00291. https://doi.org/10.1016/j.btre.2018.e00291
Sharma S, Vig AP (2014) Preliminary phytochemical screening and in vitro antioxidant activities of parkinsonia aculeata linn. Biomed Res Int 2014:756184. https://doi.org/10.1155/2014/756184
Sun CX, Ma YJ, Wang JW (2017) Enhanced production of hypocrellin A by ultrasound stimulation in submerged cultures of Shiraia bambusicola. Ultrason Sonochem 38:214–224. https://doi.org/10.1016/j.ultsonch.2017.03.020
Tabaraki R, Nateghi A (2011) Optimization of ultrasonic-assisted extraction of natural antioxidants from rice bran using response surface methodology. Ultrason Sonochem 18:1279–1286. https://doi.org/10.1016/j.ultsonch.2011.05.004
Tatay E, Font G, Ruiz MJ (2016) Cytotoxic effects of zearalenone and its metabolites and antioxidant cell defense in CHO-K1 cells. Food Chem Toxicol 96:43–49. https://doi.org/10.1016/j.fct.2016.07.027
Walia M, Kumar S, Agnihotri VK (2016) UPLC-PDA quantification of chemical constituents of two different varieties (golden and royal) of apple leaves and their antioxidant activity. J Sci Food Agric 96:1440–1450. https://doi.org/10.1002/jsfa.7239
Wang F, Ma A, Guo C, Zhuang G, Liu C (2013) Ultrasound-intensified laccase production from Trametes versicolor. Ultrason Sonochem 20:118–124. https://doi.org/10.1016/j.ultsonch.2012.05.003
Wang T, Guo N, Wang SX, Kou P, Zhao CJ, Fu YJ (2018) Ultrasound-negative pressure cavitation extraction of phenolic compounds from blueberry leaves and evaluation of its DPPH radical scavenging activity. Food Bioprod Process 108:69–80. https://doi.org/10.1016/j.fbp.2018.01.003
Xiao J, Zhang Q, Gao Y, Tang J, Zhang A, Gao J (2014) Secondary metabolites from the endophytic Botryosphaeria dothidea. J Agric Food Chem 62:3584–3590. https://doi.org/10.1021/jf500054f
Xue Z, Li J, Yu W, Lu X, Kou X (2016) Effects of nonthermal preservation technologies on antioxidant activity of fruits and vegetables: a review. Food Sci Technol Int 22:440–458. https://doi.org/10.1177/1082013215606835
Yao LY, Zhu YX, Jiao RH, Lu YH, Tan RX (2014) Enhanced production of fumigaclavine C by ultrasound stimulation in a two-stage culture of Aspergillus fumigatus CY018. Biores Technol 159:112–117. https://doi.org/10.1016/j.biortech.2014.02.072
Zabot GL, Moraes MN, Meireles MAA (2018) Process integration for producing tocotrienols-rich oil and bixin-rich extract from annatto seeds: a techno-economic approach. Food Bioprod Process 109:122–138. https://doi.org/10.1016/j.fbp.2018.03.007
Acknowledgements
The authors thank the National Council of Technological and Scientific Development (CNPq) and Research Support Foundation of the State of Rio Grande do Sul (FAPERGS) for the financial support of this work, as well as the Coordination for the Improvement of Higher Education Personnel (CAPES, finance code 001) for scholarships. M.A. Mazutti and G.L. Zabot thank the CNPq (304882/2018-6) for the research productivity grants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors confirm that there are no conflicts of interest regarding this work.
Rights and permissions
About this article
Cite this article
Valente, I.d., Confortin, T.C., Luft, L. et al. Effects of ultrasound on submerged fermentation for producing antioxidant metabolites from Botryosphaeria dothidea. Braz. J. Chem. Eng. 37, 475–484 (2020). https://doi.org/10.1007/s43153-020-00044-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-020-00044-8