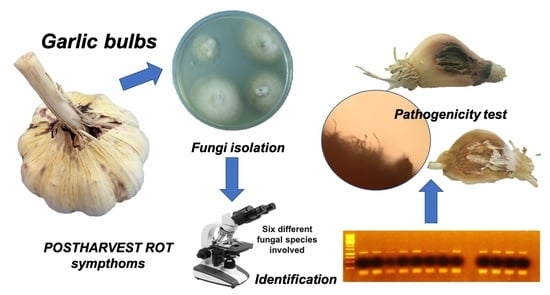
Incidence and Etiology of Postharvest Fungal Diseases Associated with Bulb Rot in Garlic (Alllium sativum) in Spain
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Samples
2.2. Sample Processing and Description of Symptoms
2.3. Fungal Isolation
2.4. Identification of Fungal Isolates
2.5. Pathogenicity Tests
2.6. Statistical Analysis
3. Results
3.1. Observed Symptoms on Garlic Cloves
3.2. Incidence and Severity of Post-Harvest Fungal Diseases
3.3. Identification of the Fungal Species Associated with Each Symptom
3.4. Postharvest Pathogenicity Test on Garlic Cloves
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ansary, J.; Forbes-Hernández, T.Y.; Gil, E.; Cianciosi, D.; Zhang, J.; Elexpuru-Zabaleta, M.; Simal-Gandara, J.; Giampieri, F.; Battino, M. Potential health benefit of garlic based on human intervention studies: A brief overview. Antioxidants 2020, 9, 619. [Google Scholar] [CrossRef]
- FAOSTAT. FAO Statistical Division 2019. Available online: http://www.fao.org/faostat/en#data (accessed on 17 April 2021).
- Sivakumar, D.; Bautista-Baños, S. A review on the use of essential oils for postharvest decay control and maintenance of fruit quality during storage. Crop Prot. 2014, 64, 27–37. [Google Scholar] [CrossRef]
- Sholberg, P.L.; Conway, W.S. Postharvest Pathology. In The Commercial Storage of Fruits, Vegetables, and Florist and Nursery Stocks, USDA-ARS Agriculture Handbook; Gross, K.C., Wang, C.Y., Saltveit, M., Eds.; United States Department of Agriculture: Washington, DC, USA, 2016; Volume 66, pp. 111–127. [Google Scholar]
- Chretien, P.L.; Laurent, S.; Bornard, S.; Troulet, C.; El Maâtaoui, M.; Leyronas, C. Unraveling the infection process of garlic by Fusarium proliferatum, the causal agent of root rot. Phytopathol. Mediterr. 2020, 59, 285–293. [Google Scholar]
- Palmero, D.; Gálvez, L.; García, M.; Gil-Serna, J.; Benito, S. The effects of storage duration, temperature and cultivar on the severity of garlic clove rot caused by Fusarium proliferatum. Postharvest Biol. Technol. 2013, 78, 34–39. [Google Scholar]
- Burba, J.L. Producción de ajo. In Ediciones Instituto Nacional de Tecnología Agropecuaria; INTA EEA La Consulta: Mendoza, Argentina, 2003. [Google Scholar]
- Piccolo, R. Enfermedades Micológicas y Bacterianas del ajo (Allium sativum). Posgrado en Horticultura. 2007. Available online: http://inta.gob.ar/sites/default/files/script-tmp-enfermedades_del_ajo.pdf (accessed on 12 January 2021).
- Schwartz, H.F.; Mohan, S.K. Compendium of Onion and Garlic Diseases and Pests; APS Press: St. Paul, MI, USA, 2008. [Google Scholar]
- Snowdon, A.L. Chapter 7: Bulbs. In A Color Atlas of Post-Harvest Diseases & Disorders of Fruits & Vegetables; CRC Press: London, UK, 1992; Volume 2, pp. 236–261. [Google Scholar]
- Khatoon, A.; Mohapatra, A.; Satapathy, K.B. Studies on fungi associated with stora garlic (Allium sativum). Studies 2017, 6, 19–24. [Google Scholar]
- Dugan, F.M.; Hellier, B.C.; Lupien, S.L. Pathogenic fungi in garlic seed cloves from the United States and China, and efficacy of fungicides against pathogens in garlic germplasm in Washington State. J. Phytopathol. 2007, 155, 437–445. [Google Scholar] [CrossRef]
- Valdez, J.G.; Makuch, M.A.; Ordovini, A.F.; Frisvad, J.C.; Overy, D.P.; Masuelli, R.; Piccolo, R.J. Identification, pathogenicity and distribution of Penicillium spp. isolated from garlic in two regions in Argentina. Plant Pathol. 2009, 58, 352–361. [Google Scholar] [CrossRef]
- Moharam, M.H.A.; Farrag, E.S.H.; Mohamed, M.D.A. Pathogenic fungi in garlic seed cloves and first report of Fusarium proliferatum causing cloves rot of stored bulbs in upper Egypt. Arch. Phytopathol. Plant Protect. 2013, 46, 2096–2103. [Google Scholar] [CrossRef]
- Mondani, L.; Mondani, L.; Chiusa, G.; Pietri, A.; Battilani, P. Monitoring the incidence of dry rot caused by Fusarium proliferatum in garlic at harvest and during storage. Postharvest Biol. Technol. 2021, 173, 111407. [Google Scholar] [CrossRef]
- Tonti, S.; Mandrioli, M.; Nipoli, P.; Pisi, A.; Toschi, T.G.; Prodi, A. Detection of fumonisins in fresh and dehydrated commercial garlic. J. Agric. Food Chem. 2017, 65, 7000–7005. [Google Scholar] [CrossRef]
- Palmero, D.; De Cara, M.; Nosir, W.; Gálvez, L.; Cruz, A.; Woodward, S.; González-Jaén, M.T.; Tello, J.C. Fusarium proliferatum isolated from garlic in Spain: Identification, toxigenic potential and pathogenicity on related Allium species. Phytopathol. Mediterr. 2012, 51, 207–218. [Google Scholar]
- Thanushree, M.P.; Sailendri, D.; Yoha, K.S.; Moses, J.A.; Anandharamakrishnan, C. Mycotoxin contamination in food: An exposition on spices. Trends Food Sci. Technol. 2019, 93, 69–80. [Google Scholar] [CrossRef]
- Barnett, H.L.; Hunter, B. Ilustrated Genera of Imperfect Fungi; APS Press: St. Paul, MI, USA, 1998. [Google Scholar]
- Ellis, M.B. More Dematiaceous Hyphomycetes; CAB International: Denver, CO, USA, 1976. [Google Scholar]
- Simmons, E.G. Perfect states of Stemphylium. Mycologia 1969, 61, 1–26. [Google Scholar] [CrossRef]
- Simmons, E.G. Helminthosporium allii as type of a new genus. Mycologia 1971, 63, 380–386. [Google Scholar] [CrossRef]
- Simmons, E.G. Perfect states of Stemphylium II. Sydowia 1985, 38, 284–293. [Google Scholar]
- Simmons, E.G. Alternaria. In An Identification Manual; CBS Fungal Biodiversity Centre: Ultrercht, The Netherlands, 2007. [Google Scholar]
- Frisvad, J.C.; Samson, R.A. Polyphasic taxonomy of Penicillium subgenus Penicillium. A guide to identification of food and air-borne terverticillate penicillia and their mycotoxins. Stud. Mycol. 2004, 49, 1–174. [Google Scholar]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing: Ames, IA, USA, 2006. [Google Scholar]
- Querol, A.; Barrio, E.; Ramón, D. A comparative study of different methods of yeast strain characterization. Syst. Appl. Microbiol. 1992, 15, 439–446. [Google Scholar] [CrossRef]
- Gálvez, L.; Urbaniak, M.; Waśkiewicz, A.; Stępień, L.; Palmero, D. Fusarium proliferatum—Causal agent of garlic bulb rot in Spain: Genetic variability and mycotoxin production. Food Microbiol. 2017, 67, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Dugan, F.; Hellier, B.; Lupien, S. First report of Fusarium proliferatum causing rot of garlic bulbs in North America. Plant Pathol. 2003, 52, 426. [Google Scholar] [CrossRef]
- Tonti, S.; Prà, M.D.; Nipoti, P.; Prodi, A.; Alberti, I. First report of Fusarium proliferatum causing rot of stored garlic bulbs (Allium sativum L.) in Italy. J. Phytopathol. 2012, 160, 761–763. [Google Scholar] [CrossRef]
- Simey, E.I. Garlic rot caused by Fusarium proliferatum (Matsushima) Nirenferg var. minus Nirenferg in Hungary. Növényvédelem 1990, 26, 397–399. [Google Scholar]
- Palmero, D.; De Cara, M.; Iglesias, C.; Moreno, M.; Gonzalez, N.; Tello, J. First report of Fusarium proliferatum causing rot of garlic bulbs in Spain. Plant Dis. 2010, 94, 277. [Google Scholar] [CrossRef] [PubMed]
- Salvalaggio, A.; Ridao, A.D.C. First report of Fusarium proliferatum causing rot on garlic and onion in Argentina. Plant Dis. 2013, 97, 556. [Google Scholar] [CrossRef]
- Sankar, N.R.; Babu, G.P. First report of Fusarium proliferatum causing rot of garlic bulbs (Allium sativum) in India. Arch. Phytopathol. Plant Protect. 2012, 1, 8. [Google Scholar] [CrossRef]
- Leyronas, C.; Chrétien, P.L.; Troulet, C.; Duffaud, M.; Villeneuve, F.; Morris, C.; Hunyadi, H. First report of Fusarium proliferatum causing garlic clove rot in France. Plant Dis. 2018, 102, 2658. [Google Scholar] [CrossRef]
- Anisimova, O.K.; Timofey, M.S.; Danilova, O.A.; Filyushin, M. First report of Fusarium proliferatum causing garlic clove rot in Russian Federation. Plant Dis. 2021. [Google Scholar] [CrossRef]
- Haapalainen, M.; Latvala, S.; Kuivainen, E.; Qiu, Y.; Segerstedt, M.; Hannukkala, A. Fusarium oxysporum, F. proliferatum and F. redolens associated with basal rot of onion in Finland. Plant Pathol. 2016, 120, 112–120. [Google Scholar]
- Dugan, F.M. Diseases and disease management in seed garlic: Problems and prospects. Am. J. Plant Sci. Biotechnol. 2007, 1, 47–51. [Google Scholar]
- Valdez, J.G.; Makuch, M.A.; Ordovini, A.F.; Masuelli, R.W.; Overy, D.P.; Piccolo, R.J. First report of Penicillium allii as a field pathogen of garlic (Allium sativum). Plant Pathol. 2006, 55, 583. [Google Scholar] [CrossRef]
- Overy, D.P.; Karlshøj, K.; Due, M. Low temperature growth and enzyme production in Penicillium ser. Corymbifera species, casual agents of blue mold storage rot in bulbs. J. Plant Pathol. 2005, 87, 57–63. [Google Scholar]
- Overy, D.P.; Frisvad, J.C.; Steinmeier, U.; Thrane, U. Clarification of the agents causing blue mold storage rot upon various flower and vegetable bulbs: Implications for mycotoxin contamination. Postharvest Biol. Technol. 2005, 35, 217–221. [Google Scholar] [CrossRef]
- Dugan, F.; Lupien, S.; Vahling-Armstrong, C.; Chastagner, G.; Schroeder, B. Host ranges of North American isolates of Penicillium causing blue mold of bulb crops. Crop Protect. 2014, 64, 129–136. [Google Scholar] [CrossRef]
- Smalley, E.B.; Hansen, H.N. Penicillium decay of garlic. Phytopathology 1962, 52, 666–678. [Google Scholar]
- Basallote, M.J.; Prados, A.M.; Pérez de Algaba, A.; Melero-Vara, J.M. First report in Spain of two leaf spots of garlic caused by Stemphylium vesicarium. Plant Dis. 1993, 77, 952. [Google Scholar] [CrossRef]
- Polat, Z.; Besirli, G.; Sönmez, İ.; Yavuz, B. First report of Stemphylium leaf blight of garlic (Allium sativum) caused by Stemphylium vesicarium in Turkey. Australas. Plant Pathol. 2012, 25, 29. [Google Scholar]
- Latha, S.; Sathyanarayana, N.; Reddy, O. Interception of Embellisia allii in garlic bulbs imported from China. Indian Phytopathol. 2011, 60, 126–127. [Google Scholar]
- Lee, H.B.; Kim, C.J.; Yu, S.H. First report of bulb canker of garlic caused by Embellisia allii in Korea. Mycobiology 2002, 30, 240–243. [Google Scholar] [CrossRef]
- Lee, H.B.; Magan, N. The influence of environmental factors on growth and interactions between Embellisia allii and Fusarium oxysporum f. sp. cepae isolated from garlic. Int. J. Food Microbiol. 2010, 138, 238–242. [Google Scholar] [CrossRef] [Green Version]
- Delgado Ortiz, J.C.; Cema Chávez, E.; Ochoa Fuentes, Y.M.M.; Beltrán Beache, M. First report of Alternaria embellisia (syn. Embellisia allii) causing bulb canker or skin blotch on garlic in Mexico. Plant Dis. 2019, 103, 1031. [Google Scholar]
- Dugan, F.; Lupien, S.; Hellier, B. Infection by Fusarium proliferatum in aerial garlic bulbils is strongly reduced compared to rates in seed cloves when both originate from infected bulbs. Crop Prot. 2019, 116, 43–48. [Google Scholar] [CrossRef]
- Gálvez, L.; Marrero, M.D.; Llamas, D. In vitro and field efficacy of three fungicides against Fusarium bulb rot of garlic. Eur. J. Plant Pathol. 2017, 148, 321–328. [Google Scholar]
- Mondani, L.; Chiusa, G.; Battilani, P. Chemical and biological control of Fusarium species involved in garlic dry rot at early crop stages. Eur. J. Plant Pathol. 2021. [Google Scholar] [CrossRef]
- De Santis, D.; Garzoli, S.; Vettraino, A.M. Effect of gaseous ozone treatment on the aroma and clove rot by Fusarium proliferatum during garlic postharvest storage. Heliyon 2021, 7, e06634. [Google Scholar] [CrossRef]



| Symptom | n | Fungal Prevalence | Aspergillus | Embellisia | Fusarium | Penicillium | Rhizopus | Stemphylium |
|---|---|---|---|---|---|---|---|---|
| Dry Rot | 104 | 80.8 | 0.0 | 0.0 | 76 | 5.8 | 3.8 | 0 |
| Green Rot | 32 | 81.3 | 3.1 | 0.0 | 3.1 | 78.1 | 0.0 | 0 |
| Wound Damage | 14 | 100.0 | 0.0 | 14 | 28.6 | 21.4 | 0.0 | 42.9 |
| Symptom | Season | Garlic Cultivar | ||||
|---|---|---|---|---|---|---|
| 2013 | 2014 | Spanish Purple | Spanish White | Chinese Purple | Chinese White | |
| GR 1 | 90.3 * | 9.7 * | 9.7 | 35.5 * | 16.1 | 38.7 * |
| DR 2 | 40.1 | 59.9 | 21 | 24.9 | 29.3 | 24.8 |
| WD 3 | 58.3 | 41.7 | 0 | 7.5 | 25 | 67.5 |
| Isolate | Colony Diameter (mm) | Texture | Exudate | Diffusible Yellow Pigment | Concentric Furrows | Group |
|---|---|---|---|---|---|---|
| PA086 | 33.29 ± 1.21 | Granular | − | Low | Absent | 1 |
| PA088 | 17.63 ± 0.99 | Velvety | + | High | Present | 2 |
| PA089 | 31.97 ± 0.79 | Granular | + | Low | Present | 3 |
| PA090 | 32.74 ± 0.93 | Granular | − | Low | Absent | 1 |
| PA100 | 27.55 ± 0.50 | Velvety | − | Low | Present | 2 |
| PA102 | 30.54 ± 0.64 | Velvety | + | High | Present | 3 |
| PA112 | 27.72 ± 0.97 | Velvety | + | Low | Present | 3 |
| PA113 | 27.46 ± 0.34 | Velvety | + | Low | Present | 3 |
| PA114 | 30.83 ± 0.83 | Granular | + | High | Present | 3 |
| PA115 | 30.57 ± 0.46 | Granular | − | High | Present | 3 |
| PA130 | 21.75 ± 0.59 | Granular | + | Low | Absent | 1 |
| Isolate | FB1 | FB2 | FB3 | BEA | MON | Growth Rate | DSI |
|---|---|---|---|---|---|---|---|
| FPG05 | 749.59 | 144.82 | 45.05 | 146.09 | 57.62 | 8.34 | 1.42 |
| FPG08 | 2100.2 | 389.16 | 72.75 | 412.25 | 0.27 | 10.13 | 1.50 |
| FPG12 | 55.55 | 0.73 | 0.57 | nd | 0.98 | 5.09 | 2.17 |
| FPG16 | 8.64 | 1.36 | 0.43 | 188.48 | 11.59 | 8.09 | 1.00 |
| FPG20 | 766.74 | 127.79 | 13.58 | 429.78 | 0.44 | 9.21 | 2.09 |
| FPG21 | 2913.99 | 708.54 | 104.08 | 472.02 | 0.24 | 8.17 | 1.92 |
| FPG23 | 120.06 | 25.56 | 19.37 | 152.13 | 0.13 | 12.04 | 2.25 |
| FPG30 | 527.03 | 88.54 | 43.51 | 27.56 | 2.38 | 11.67 | 2.17 |
| FPG33 | 667.83 | 158.36 | 45.1 | 995.37 | nd | 7.80 | 2.25 |
| FPG34 | 1202.61 | 34.11 | 19.92 | 44.37 | 68.88 | 9.13 | 1.17 |
| FPG35 | 239.29 | 22.4 | 18.27 | 79.07 | 1.26 | 11.79 | 2.25 |
| FPG36 | 64.22 | 8.84 | 0.93 | nd | 4.71 | 9.13 | 0.92 |
| FPG45 | 493.51 | 62.28 | 47.07 | 187.25 | 0.37 | 12.29 | 2.21 |
| FPG58 | 1335.2 | 222.53 | 40.98 | 552.51 | 0.3 | 8.04 | 1.92 |
| FPG59 | 249.06 | 40.36 | 5.86 | 68.65 | 5.71 | 7.59 | 1.17 |
| FPG65 | 510.22 | 172.64 | 19.41 | 564.22 | 1.07 | 10.50 | 1.59 |
| FPG75 | 1.64 | 0.31 | 0.2 | 96.24 | 0.29 | 7.21 | 1.00 |
| FPG77 | 354.32 | 72.74 | 9.04 | 358.51 | 1.01 | 6.00 | 1.83 |
| FPG80 | 586.94 | 86.15 | 14.67 | 160.08 | 8.14 | 6.67 | 2.25 |
| FPG82 | 1730.23 | 216.28 | 15.96 | 596.94 | 1.87 | 9.84 | 1.42 |
| p valor * | 0.804 | 0.577 | 0.252 | 0.242 | 0.158 | 0.567 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gálvez, L.; Palmero, D. Incidence and Etiology of Postharvest Fungal Diseases Associated with Bulb Rot in Garlic (Alllium sativum) in Spain. Foods 2021, 10, 1063. https://doi.org/10.3390/foods10051063
Gálvez L, Palmero D. Incidence and Etiology of Postharvest Fungal Diseases Associated with Bulb Rot in Garlic (Alllium sativum) in Spain. Foods. 2021; 10(5):1063. https://doi.org/10.3390/foods10051063
Chicago/Turabian StyleGálvez, Laura, and Daniel Palmero. 2021. "Incidence and Etiology of Postharvest Fungal Diseases Associated with Bulb Rot in Garlic (Alllium sativum) in Spain" Foods 10, no. 5: 1063. https://doi.org/10.3390/foods10051063